4066
Intracranial atherosclerosis is associated with lower volume in the posterior body and tail of the hippocampus.1Biomedical Engineering, Illinois Institute of Technology, Chicago, IL, United States, 2Rush Alzheimer’s Disease Center, Rush University Medical Center, Chicago, IL, United States
Synopsis
Keywords: Dementia, Blood vessels, Atherosclerosis, Brain, Pathology, Ex-vivo applications, Hippocampus, Neurodegeneration, Vascular
Motivation: Intracranial atherosclerosis is a common and deleterious age-related neuropathology, however, its association with brain morphometry has not been investigated.
Goal(s): To investigate brain morphometric anomalies associated with intracranial atherosclerosis in a large number of community-based older adults.
Approach: Ex-vivo brain MRI and detailed neuropathologic examination were combined on 891 community-based older adults. Deformation-based morphometry (DBM) was used to investigate the association of intracranial atherosclerosis with brain morphometric characteristics voxel-wise.
Results: Intracranial atherosclerosis was associated with lower volume in the posterior body and tail of the hippocampus in linear regression models controlling for all other neuropathologies.
Impact: Intracranial atherosclerosis is the third age-related neuropathology after Alzheimer’s and limbic-predominant age-related TDP-43 encephalopathy (LATE) that is shown to be independently associated with lower volume of the hippocampus.
Introduction
Intracranial atherosclerosis is a common age-related neuropathology that has been linked to cognitive decline and dementia1–4. Black older adults have more severe atherosclerosis than white older adults5. Intracranial atherosclerosis is often mixed with Alzheimer’s and other neuropathologies increasing the odds of dementia6. Despite its prevalence, its negative impact on cognitive function and its role in aging and dementia, the association of atherosclerosis with brain morphometric abnormalities has not been explored. Deformation-based morphometry (DBM) is an approach that allows investigation of brain morphometry at the voxel level7,8. In this work, we combined DBM on ex-vivo MRI with detailed neuropathological examination in a large number of community-based older adults to investigate the association of intracranial atherosclerosis with brain morphometric anomalies.Methods
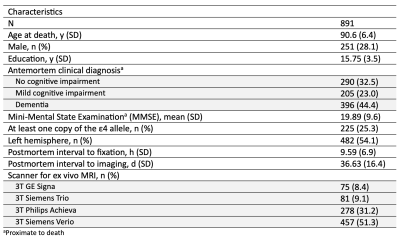
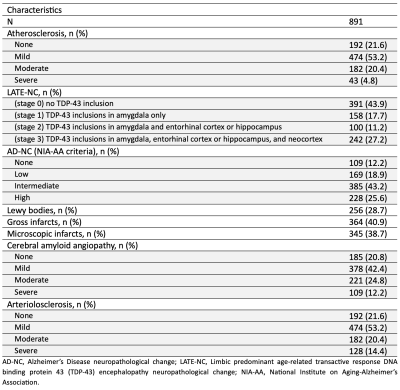
Participants, MRI, neuropathologic examination:This work included 891 community-based older adults participating in four cohort studies of aging: the Rush Memory and Aging Project, Religious Orders Study9, Minority Aging Research Study, and African American Clinical Core of the Rush Alzheimer’s Disease Research Center10. All participants came to autopsy. Hemispheres from all participants were submerged in 4% formaldehyde solution and imaged approximately one month postmortem on 3T clinical MRI scanners using a multi-echo spin-echo sequence with a voxel size=0.6×0.6×1.5 mm3. All images were non-linearly registered to an ex vivo brain hemisphere template using ANTs11. The logarithm of the Jacobian determinant of the deformation fields was calculated in each voxel and the resulting maps (LogJ) were smoothed using a Gaussian filter with a FWHM=4mm. Following ex-vivo MRI, all hemispheres underwent detailed neuropathologic examination. The assessed pathologies included atherosclerosis, arteriolosclerosis, cerebral amyloid angiopathy, gross and microscopic infarcts, Alzheimer's pathology, Lewy bodies, limbic-predominant age-related TDP-43 encephalopathy neuropathological change (LATE-NC), and hippocampal sclerosis.
Statistical Analyses:
Voxel wise linear regression was used to test the association of atherosclerosis with deformations shown in the smoothed LogJ maps, controlling for other age-related neuropathologies (Alzheimer’s pathology, Lewy bodies, limbic-predominant age-related TDP-43 encephalopathy (LATE), hippocampal sclerosis, arteriolosclerosis, cerebral amyloid angiopathy, gross infarcts, microscopic infarcts), demographics (age at death, sex, years of education), postmortem interval to fixation, postmortem interval to imaging, and scanner (Fig. 1,2). The FSL PALM tool with 1000 permutations, threshold-free cluster enhancement, and tail acceleration was used for the statistical analysis12. Associations were considered significant at p<0.05 after family-wise error rate (FWER) correction for multiple comparisons.
Results and Discussion
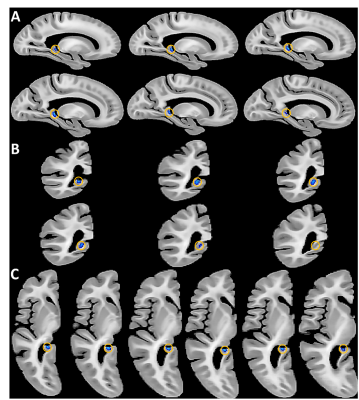
Voxel-wise linear regression showed that intracranial atherosclerosis was significantly associated with lower volume in the posterior body and tail of the hippocampus (p<0.05), independently of the effects of other age-related neurodegenerative and vascular pathologies (Fig.3). No part of the brain showed significantly higher volume with atherosclerosis. These findings suggest that intracranial atherosclerosis is associated with focal neurodegeneration of the posterior portion of the hippocampus, sparing other brain tissues.Conclusion
This work combined DBM on ex-vivo MRI with detailed neuropathologic examination in a large number of community-based older adults and demonstrated that intracranial atherosclerosis is associated with lower volume of the posterior body and tail of the hippocampus. This finding is of great interest due to the important role of the hippocampus in cognition. Furthermore, atherosclerosis is often mixed with Alzheimer’s pathology and/or LATE neuropathological change, which have previously shown independent associations with lower hippocampal volume. Therefore, hippocampal volume by itself cannot serve as a reliable marker of any of the three pathologies, but more localized metrics of hippocampal atrophy may potentially provide higher specificity.Acknowledgements
This study was supported by the following grants:
National Institute on Aging (NIA): R01AG064233, R01AG067482, R01AG017917, R01AG015819, P30AG010161, P30AG072975.
National Institute of Neurological Disorders and Stroke (NINDS): UF1NS100599.
References
1. Dearborn JL, Zhang Y, Qiao Y, et al. Intracranial atherosclerosis and dementia: The Atherosclerosis Risk in Communities (ARIC) Study. Neurology. 2017;88(16):1556-1563. doi:10.1212/WNL.0000000000003837.
2. Gorelick PB, Scuteri A, Black SE, et al. Vascular contributions to cognitive impairment and dementia: a statement for healthcare professionals from the american heart association/american stroke association. Stroke. 2011;42(9):2672-2713. doi:10.1161/STR.0b013e3182299496.
3. Lamar M, Leurgans S, Kapasi A, et al. Complex Profiles of Cerebrovascular Disease Pathologies in the Aging Brain and Their Relationship With Cognitive Decline. Stroke. 2022;53(1):218-227. doi:10.1161/STROKEAHA.121.034814.
4. Arvanitakis Z, Capuano AW, Leurgans SE, Bennett DA, Schneider JA. Relation of Cerebral Vessel Disease to Alzheimer’s Disease Dementia and Cognitive Function in Older Persons: A Cross-sectional Study. Lancet Neurol. 2016;15(9):934-943. doi:10.1016/S1474-4422(16)30029-1.
5. Barnes LL, Leurgans S, Aggarwal NT, et al. Mixed pathology is more likely in black than white decedents with Alzheimer dementia. Neurology. 2015;85(6):528-534. doi:10.1212/WNL.0000000000001834.
6. Kapasi A, DeCarli C, Schneider JA. Impact of multiple pathologies on the threshold for clinically overt dementia. Acta Neuropathol. 2017;134(2):171-186. doi:10.1007/s00401-017-1717-7.
7. Gaser C, Nenadic I, Buchsbaum BR, Hazlett EA, Buchsbaum MS. Deformation-based morphometry and its relation to conventional volumetry of brain lateral ventricles in MRI. Neuroimage. 2001;13(6 Pt 1):1140-1145. doi:10.1006/nimg.2001.0771.
8. Chung MK, Worsley KJ, Paus T, et al. A unified statistical approach to deformation-based morphometry. Neuroimage. 2001;14(3):595-606. doi:10.1006/nimg.2001.0862.
9. Bennett DA, Buchman AS, Boyle PA, Barnes LL, Wilson RS, Schneider JA. Religious Orders Study and Rush Memory and Aging Project. J Alzheimers Dis. 2018;64(s1):S161-S189. doi:10.3233/JAD-179939.
10. Barnes LL, Shah RC, Aggarwal NT, Bennett DA, Schneider JA. The Minority Aging Research Study: ongoing efforts to obtain brain donation in African Americans without dementia. Curr Alzheimer Res. 2012;9(6):734-745. doi:10.2174/156720512801322627.
11. Avants BB, Tustison NJ, Song G, Cook PA, Klein A, Gee JC. A reproducible evaluation of ANTs similarity metric performance in brain image registration. NeuroImage. 2011;54(3):2033-2044. doi:10.1016/j.neuroimage.2010.09.025.
12. Winkler AM, Ridgway GR, Webster MA, Smith SM, Nichols TE. Permutation inference for the general linear model. Neuroimage. 2014;92(100):381-397. doi:10.1016/j.neuroimage.2014.01.060.
Figures