2310
Neural Pathways of Applied Reflexology using Real-Time Task-Based and Resting-State fMRI in Healthy Controls and Patients with Stroke1University of New Mexico, Dept. of Neurology, Albuquerque, NM, United States, 2Physics and Astronomy, University of New Mexico, Albuquerque, NM, United States, 3Reflexology Research Project, Albuquerque, NM, United States, 4Division of Physical Therapy, Division of Rehabilitation Science, Department of Rehabilitation Medicine, Medical School, University of Minnesota, Minneapolis, MN, United States, 5Center for Magnetic Resonance Research, Department of Radiology, University of Minnesota, Minneapolis, MN, United States
Synopsis
Keywords: Stroke, Stroke, Reflexology, real-time, sensory, connectivity
Motivation: To characterize the biological substrates and mechanisms of reflexology.
Goal(s): (1) To characterize the somatotopic relationship between cortical activity and sensory stimulation of reflex areas. (2) To assess possible therapeutic effects of reflexology.
Approach: Real-time task-based and resting-state fMRI in 4 controls and 4 adults with stroke using multi-band multi-echo EPI.
Results: Deactivation of a bilateral network in superior-temporal gyrus, postcentral gyrus and insular cortex (depending on reflex area) in healthy controls and unilateral deactivation, which differed between left and right foot, in patients with stroke. Sensorimotor function in the hemiplegic hand improved (3 patients) and two-point discrimination increased (2 patients) after reflexology.
Impact: This preliminary real-time fMRI study demonstrates the potential to change the reflexologist’s technique application to focus on particular reflex areas for optimizing treatment results and to establish a neurobiological basis for predicting treatment outcomes in patients with neurological disorders.
INTRODUCTION
There is an urgent need to characterize the biological substrates and mechanisms of reflexology, and to establish a neurobiological basis for predicting treatment outcomes in patients with neurological disorders. Task-based fMRI studies demonstrated co-activation of the eloquent cortex beyond the foot area1, a lateralized projection of reflex areas to the primary somatosensory cortex that differs from that of the actual somatotopic representation of the body2, and selective decrease of signal changes in the retrosplenial/posterior cingulate during a cognitive task relative to the resting-state state3. Resting-state fMRI revealed changes in functional connectivity in the default-mode network, the sensorimotor network, and a newly discovered neural network correlates of pain4.In this study, we characterized the somatotopic relationship between cortical activity and sensory stimulation of reflex areas during the application of reflexology using real-time task-based and resting-state fMRI (tfMRI and rsfMRI) using highly accelerated fMRI. Four patients with stroke were scanned to assess disease-related functional reorganization of brain networks responsive to reflexology stimuli and possible therapeutic effects of reflexology.
METHODS
Real-time fMRI (5:27min per reflex area) data were acquired in 4 healthy controls (1M,3F, 43-71y) and in 4 patients with stroke (3M,1F, 52-63y, between 1-14 years after a right middle cerebral artery-ischemic stroke, resulting in severe left hemiplegia) while the reflexology practitioner applied reflexological stimuli (15 blocks of 8s stimulation and 12s rest). Informed written consent was obtained.Reflex areas of the right foot (pituitary, vagus, adrenal, eye, temporal) were stimulated in healthy controls. Bilateral reflex areas in patients (eye-ear, brain stem, vagus) were selected based on to the participant’s disability. Upper limb motor function (using MESUPES5) and upper limb sensory function (using a standard clinical battery6 of touch sensation, proprioception, two-point discrimination on the index finger, and stereognosis) were evaluated before and after reflexology.
Real-time fMRI was performed using multi-echo MB-EPI (TR400ms, TE1/TE2: 15/41ms or TE1/TE2/TE3: 14/39/64ms, multiband-acceleration: 8 or 10, flip-angle:30o, voxel-size:(3mm)3, 40slices) on a 3T scanner equipped with 32-channel head coil. Real-time tfMRI and rsfMRI analyses were performed using the TurboFIRE software tool7-10 on a laptop interfaced to the scanner. In patients, three RSNs were mapped using seed-based averaged sliding window (15s) correlation analyses with unilateral Brodmann area (BA) based seed regions (sensorimotor network (SMN): BA01-03, language networks (LAN): BA44,45 (Broca’s)) and a manually drawn seed region within bilateral precuneus (default mode network (DMN)).
RESULTS
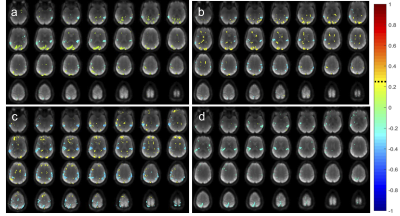
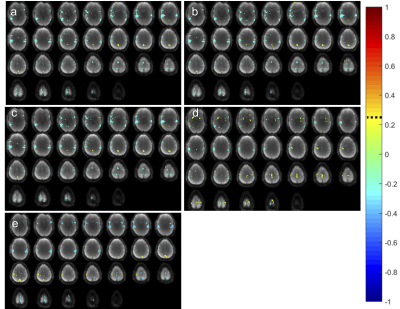
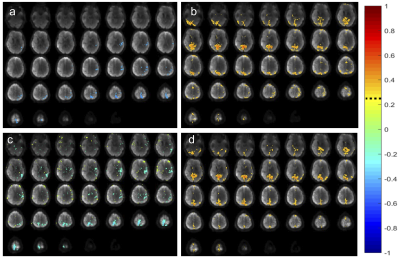
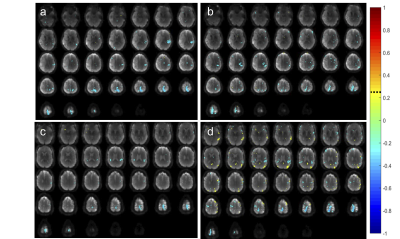
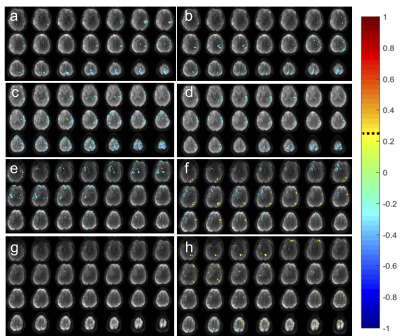
Bilateral networks in superior temporal gyrus, postcentral gyrus and insular cortex and basal nuclei (depending on reflex area) were deactivated in healthy controls (Figs. 1, 2). Reflexology related signal changes in the foot sensorimotor area were detectable in 3 controls: 6 scans showed activation, 6 scans showed deactivation, and 2 scans showed no fMRI signal change. In some of the scans that showed deactivation of the foot sensorimotor area, transient activation of the foot sensorimotor area was detectable in the beginning of the scan. In addition, we detected activation in a range of sensory areas, primarily in visual areas, which varied across subjects and reflex areas.Unilateral deactivation in healthy brain tissue was seen in patients which changed in intensity when stimulating the paralyzed foot (Figs. 3 - 5). In some cases, the deactivation pattern changed (Fig. 4a,b). Resting-state networks were robustly detected in healthy tissue.
After the reflexology sessions in the MRI scanner, sensorimotor function improved in the hemiplegic hand in three patients. Scores increased on the MESUPES arm function between 1 point (n=1) and 5 points (n=2). The patient who did not improve reported receiving regular bouts of botox in the upper limb every couple of months, while the other participants did not use botox. Two-point discrimination increased in two patients, from 6 to 2 mm and from 8 to 4 mm, respectively.
DISCUSSION
A surprising finding in this study was deactivation distant from the foot sensorimotor area in response to reflexology stimuli. This is different from activation patterns reported in previous studies that used a wooden stick for stimulation1,2. An unanticipated finding was the improvement of sensorimotor function in the hemiplegic hand. Improved MESUPES scores were related to increased relaxation of the hemiplegic hand during passive, assisted, or (if possible) active movements. Given that 2-3mm is a normal two-point discrimination for the index finger, the two-point discrimination improvements seen in patients is noteworthy. Further studies are needed to address limitations, including heterogeneity of the patients and reflex areas, and to substantiate these preliminary findings.CONCLUSIONS
This preliminary real-time fMRI study demonstrates the potential to change the reflexologist’s technique application to focus on reflex areas that optimize treatment results and to establish a neurobiological basis for predicting treatment outcomes in patients with neurological disorders.Acknowledgements
Supported by the University of New Mexico Foundation. We gratefully acknowledge Kevin Rosenberg for developing a performance enhanced version of the TurboFIRE software tool.References
[1] T. Nakamaru, N. Miura, A. Fukushima, and R. Kawashima, "Somatotopical relationships between cortical activity and reflex areas in reflexology: a functional magnetic resonance imaging study," Neurosci Lett, vol. 448, pp. 6-9, Dec 19 2008.
[2] N. Miura, Y. Akitsuki, A. Sekiguchi, and R. Kawashima, "Activity in the primary somatosensory cortex induced by reflexological stimulation is unaffected by pseudo-information: a functional magnetic resonance imaging study," BMC Complement Altern Med, vol. 13, p. 114, May 27 2013, PMC3668141.
[3] D. Sliz, A. Smith, C. Wiebking, G. Northoff, and S. Hayley, "Neural correlates of a single-session massage treatment," Brain Imaging Behav, vol. 6, pp. 77-87, Mar 2012, PMC3282900.
[4] E. Descamps, M. Boussac, K. Joineau, and P. Payoux, "Changes of cerebral functional connectivity induced by foot reflexology in a RCT," Sci Rep, vol. 13, p. 17139, Oct 10 2023, PMC10564852.
[5] A. Van de Winckel, H. Feys, S. van der Knaap, R. Messerli, F. Baronti, R. Lehmann, B. Van Hemelrijk, F. Pante, C. Perfetti, and W. De Weerdt, "Can quality of movement be measured? Rasch analysis and inter-rater reliability of the Motor Evaluation Scale for Upper Extremity in Stroke Patients (MESUPES)," Clin Rehabil, vol. 20, pp. 871-884, Oct 2006.
[6] K. Klingels, E. Jaspers, A. Van de Winckel, P. De Cock, G. Molenaers, and H. Feys, "A systematic review of arm activity measures for children with hemiplegic cerebral palsy," Clin Rehabil, vol. 24, pp. 887-900, Oct 2010.
[7] S. Posse, F. Binkofski, F. Schneider, D. Gembris, W. Frings, U. Habel, J. B. Salloum, K. Mathiak, S. Wiese, V. Kiselev, T. Graf, B. Elghahwagi, M. L. Grosse-Ruyken, and T. Eickermann, "A new approach to measure single-event related brain activity using real-time fMRI: Feasibility of sensory, motor, and higher cognitive tasks," Human Brain Mapping, vol. 12, pp. 25-41, Jan 2001.
[8] S. Posse, E. Ackley, R. Mutihac, T. Zhang, R. Hummatov, M. Akhtari, M. Chohan, B. Fisch, and H. Yonas, "High-speed real-time resting-state FMRI using multi-slab echo-volumar imaging," Front Hum Neurosci, vol. 7, p. 479, 2013, 3752525.
[9] K. Vakamudi, C. Trapp, K. Talaat, K. Gao, B. Sa De La Rocque Guimaraes, and S. Posse, "Real-Time Resting-State Functional Magnetic Resonance Imaging Using Averaged Sliding Windows with Partial Correlations and Regression of Confounding Signals," Brain Connect, vol. 10, pp. 448-463, Oct 2020, PMC7580636.
[10] K. Vakamudi, S. Posse, R. Jung, B. Cushnyr, and M. O. Chohan, "Real-time presurgical resting-state fMRI in patients with brain tumors: Quality control and comparison with task-fMRI and intraoperative mapping," Hum Brain Mapp, vol. 41, pp. 797-814, Feb 15 2020.
Figures