2216
Evaluating the Neuroprotective Effects of Caplacizumab in Thrombotic Thrombocytopenic Purpura with Quantitative MRI and CT1Medical Biophysics, University of Western Ontario, London, ON, Canada, 2Robarts Research Institute, London, ON, Canada, 3Lawson Health Research Institute, London, ON, Canada
Synopsis
Keywords: Blood Vessels, Neurodegeneration, Myelin Water Imaging
Motivation: Thrombotic Thrombocytopenic Purpura patients suffer from high risk of cognitive related complications post-remission that can decrease quality-of-life and in some cases death.
Goal(s): We investigate a new treatment drug Caplacizumab to determine whether it can reduce neurocognitive burden.
Approach: We use quantitative MRI and CT-perfusion to investigate patients that have received Caplacizumab to patients that have not. MRI and CT data look at a variety of different metrics to investigate brain health.
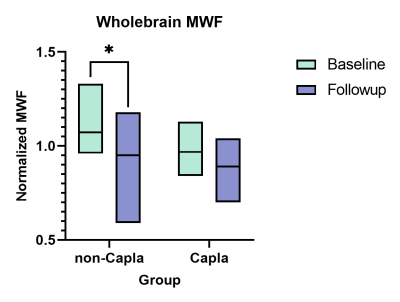
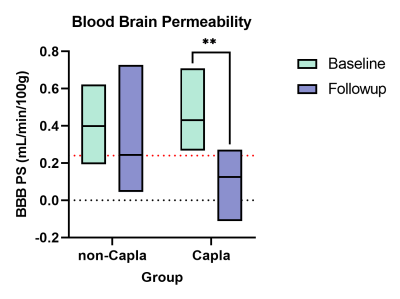
Results: Preliminary results from CT-perfusion indicate better blood brain barrier recovery in Caplacizumab group, which may decrease cognitive burden. MRI results show significant decrease of myelin in patients without Caplacizumab.
Impact: The results of this study can open the door for a new treatment for Thrombotic Thrombocytopenic Purpura patients that may ensure they do not suffer from cognitive decline or cerebrovascular disease related death.
Introduction
Immune-mediated thrombotic thrombocytopenic purpura (iTTP) is a rare life-threatening disease caused by enzyme inhibition of ADAMTS13 (A Disintegrin-like And Metalloprotease with Thrombospondin type 1 repeats 13), which leads to platelet aggregation in the microvasculature [1]. As a result, patients have reduced platelet levels, breakdown of red blood cells, small clots forming in small blood vessels, and organ failure. Modern methods of treatment include plasma exchange with or without immunosuppressants to restore ADAMTS13 activity back to healthy levels. Despite these effective treatments, more than 50% of patients in remission are prone to cerebrovascular disease, cognitive impairment, and depression [2,3]. The reasons for changes in brain tissue and cognitive performance in patients with iTTP are not well understood. However, a preliminary study using computed tomography (CT) perfusion done by our group found compromised blood brain barriers (BBB) over a six-month period along with elevated magnetic resonance imaging (MRI) T1 and T2 relaxation times that may indicate neuroinflammation [4]. Therefore, there is a need to better understand the pathophysiology in the brain of patients with iTTP and implement new treatments that reduce brain-related injuries and cognitive impairment. The exact reason for BBB disruption in iTTP remains undetermined however studies in other disease groups with thrombocytopenia find that platelets have a significant impact on the integrity and permeability of the BBB leading to alterations in endothelial cells [5]. Minimizing the BBB leakage may reduce long term brain related impairment. Caplacizumab is a new drug that expedites platelet count normalization which may reduce microthrombotic damage to the brain [6,7]. To date, the neuroprotective properties of Caplacizumab have not been studied in detail. The study is comprised of three objectives: (1) Determine the efficacy of Caplacizumab in reducing blood brain barrier leakage; (2) Study the extent of white matter injury using MRI post-remission; and (3) Determine neurocognitive changes and their association with compromised BBBs and white matter injury. We hypothesize that Caplacizumab’s innate ability to restore platelet levels faster will allow for less BBB disruption and faster recovery, thus reducing neurological complications.Methods
All imaging scans will be performed in London, Ontario. Imaging modalities include a variety of qualitative and quantitative MRI sequences using a 3 Tesla MRI (Siemens Biograph mMR) and a CT-perfusion scan (GE Healthcare Revolution CT) with iodine contrast injection. MRI sequences include 5 qualitative sequences modelled after a clinical MRI stroke protocol to assess gross pathology and 2 quantitative sequences to assess white matter integrity: myelin water imaging (MWI) and diffusion tensor imaging (DTI). MWI provides whole brain quantitative maps of relaxometry and myelin content which correlate with cognitive function. DTI provides maps of white matter tract bundles across a variety of metrics elucidating the underlying structure and integrity in different white matter regions of the brain. These metrics include fractional anisotropy (diffusion directionality), mean diffusivity (overall diffusion rate), axial diffusivity (axon integrity), and radial diffusivity (myelin integrity). Additionally, depression scores and a cognitive brain assessment using Cambridge Brain Sciences (CBS) will be assessed and correlated to imaging findings. CBS results are divided into 4 cognitive domains: reasoning, concentration, short-term memory, and verbal memory. All data is taken 30-days post remission for baseline and repeated 6-12 months for follow up.Preliminary Results
Preliminary results for 17 iTTP patients (6 on Caplacizumab) show a significant decrease in myelin during follow up MRI in patients that did not receive Caplacizumab (Figure.1). A small decrease in myelin was observed in the Caplacizumab group but not at a significant level. CT-perfusion scanning reveals a significant decrease in BBB permeability surface product in follow-up visits of Caplacizumab patients compared to standard treatment iTTP patients (Figure. 2).Discussion
Patients that received Caplacizumab had less blood brain permeability in follow-up visits, this may suggest that the faster platelet restoration provided by Caplacizumab may have aided in a faster recovery of the BBB. In comparison, while patients on standard treatment recovered blood brain permeability, it is still higher than healthy value. This could explain why this group suffered a significant decrease in myelin content compared to the Caplacizumab group, as a more compromised BBB could have resulted in further damage to brain tissue.Conclusion
This study will provide a thorough analysis of the use of Caplacizumab as a viable treatment for iTTP that not only aids in faster platelet normalization but could subsequently reduce the high risk of neurocognitive burden post remission. These preliminary results support the neuroprotective effect of Caplacizumab. Additionally, the neuroimaging used in this study will provide insight on the etiology of iTTP on the brain which remains insufficient to date.Acknowledgements
We thank our MRI technologists John Butler, Heather Biernaski, and Yvonne Huston for their help in acquiring all the imaging data; and Kerri Gallo for her help as a research coordinator.References
- Lancellotti S, Basso M, De Cristofaro R. Proteolytic processing of von Willebrand factor by adamts13 and leukocyte proteases. Mediterranean journal of hematology and infectious diseases. 2013;5(1).
- Deford CC, Reese JA, Schwartz LH, Perdue JJ, Kremer Hovinga JA, L¨ammle B, et al. Multiple major morbidities and increased mortality during long-term follow-up after recovery from thrombotic thrombo- cytopenic purpura. Blood, The Journal of the American Society of Hematology. 2013;122(12):2023–2029.
- Falter T, Schmitt V, Herold S, Weyer V, von Auer C, Wagner S, et al. Depression and cognitive deficits as long-term consequences of thrombotic thrombocytopenic purpura. Transfusion. 2017;57(5):1152–1162.
- Huang SHS, Pavenski K, Lee TY, Jurkiewicz MT, Bharatha A, Thiessen JD, et al. Blood–brain barrier permeability in survivors of immune-mediated thrombotic thrombocytopenic purpura: a pilot study. Blood Advances. 2021;5(20):4211–4218.
- Nair M, Maria JM, Agudelo M, Yndart A, Vargas-Rivera ME. Platelets contribute to BBB disruption induced by HIV and alcohol. Journal of alcoholism and drug dependence. 2015;3(1):182.
- Scully M, Kn¨obl P, Kentouche K, Rice L, Windyga J, Schneppenheim R, et al. Recombinant ADAMTS- 13: first-in-human pharmacokinetics and safety in congenital thrombotic thrombocytopenic purpura. Blood, The Journal of the American Society of Hematology. 2017;130(19):2055–2063.
- Peyvandi F, Cataland S, Scully M, Coppo P, Knoebl P, Kremer Hovinga JA, et al. Caplacizumab prevents refractoriness and mortality in acquired thrombotic thrombocytopenic purpura: integrated analysis. Blood advances. 2021;5(8):2137–2141.
Figures