2215
Associations between small vessel function and progressive white matter injury in CADASIL using advanced 7T MRI1Center for Image Sciences, University Medical Center Utrecht, Utrecht, Netherlands, 2Harvard Medical School, Massachusetts General Hospital, Boston, MA, United States, 3Institute for Stroke and Dementia Research (ISD), Ludwig Maximilian University of Munich, Munich, Germany, 4Medical Image Analysis Center (MIAC), University of Basel, Basel, Switzerland, 5Neurology, University Medical Center Utrecht, Utrecht, Netherlands
Synopsis
Keywords: Blood Vessels, White Matter, Small vessel disease, white matter hyperintensities, diffusion MRI, pulsatility, vascular reactivity, progressive white matter injury, CADASIL
Motivation: Impaired blood flow velocity and pulsatility on 7T MRI are associated with white matter injury in cerebral small vessel disease (cSVD) cross-sectionally, but it is unclear whether it relates to injury progression.
Goal(s): To assess if small vessel dysfunction is associated with markers of progressive white matter injury in monogenic cSVD.
Approach: Small vessel function was measured on 7T MRI in monogenic cSVD. White matter injury (white matter hyperintensities, peak-skeletonized mean diffusivity) was measured with 3T MRI on baseline and two-year follow-up.
Results: There were no associations between small vessel dysfunction and white matter injury progression in monogenic cSVD.
Impact: Whole-brain white matter injury progression cannot be explained by small vessel function. Local white matter injury progression might be linked with small vessel function and should be assessed in the future with voxelwise studies.
Introduction
Cerebral small vessel diseases (cSVDs) are a major cause of stroke and dementia and have been mostly studied through lesions in the brain parenchyma such as white matter hyperintensities (WMHs) or white matter integrity reflected by diffusion tensor imaging (DTI) metrics such as the peak width of the skeletonized white matter (PSMD)[1-3]. These markers are important in cSVDs because of their association with clinical outcome like stroke and cognitive decline. Small vessel function, measured with 7T MRI in terms of mean velocity and pulsatility of the perforating arteries, was found to be impaired in Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy (CADASIL), a monogenic form of cSVD[4]. Furthermore, small vessel dysfunction was associated with baseline cSVD disease burden in terms of WMHs and PSMD[5]. This suggests that small vessel function contributes to structural damage of the white matter. To better understand the role of small vessel function in the pathological pathway of cSVDs, it is important to assess if abnormal small vessel function on 7T MRI is also associated with progression of white matter injury in CADASIL.Methods
CADASIL patients were recruited through the ZOOM@SVDs study, a prospective observational cohort study with two-year follow-up[6]. CADASIL was confirmed by mutation of the NOTCH3 gene or by skin biopsy. Participants underwent a 3T brain MRI (Siemens Healthineers, Erlangen, Germany) at baseline and after two-year follow-up with a 64-channel head coil to quantify WMHs and PSMD. 7T brain MRI (Philips Healthcare, Best, The Netherlands) was performed at baseline with a 32-channel receive head coil to assess small vessel function (Figure 1). Acquisition details of the relevant sequences are presented in Table 1.The following aspects of small vessel function were assessed:
1. Blood flow velocity and pulsatility in perforating arteries at the level of the basal ganglia and centrum semioval using 2D phase-contrast angiography to assess arteriolar stiffness.
2. Reactivity to a visual stimulus was assessed in the visual cortex using the amplitude of the blood-oxygen-level dependent (BOLD) response, indicative of neurovascular coupling.
WMHs were normalized to intracranial volume. Change in WMHs and PSMD was defined as the difference between follow-up and baseline. Baseline and follow-up WMHs and PSMD were compared using a paired t-test. Small vessel function measures were related to baseline and change in WMHs and PSMD using linear regression.
Results
The characteristics of the 22 included CADASIL patients are shown in Table 2. The median follow-up time after baseline was 25 months. There was a significant increase in WMHs (p<0.001) and PSMD (p=0.03) after two-year follow-up (Figure 2).Mean blood flow velocity within the perforating arteries of the centrum semioval was significantly associated with baseline WMHs (β=-0.61; p=0.05) and PSMD (β=-0.84; p=0.04). None of the other small vessel function measures showed a significant association with baseline WMHs and PSMD (Table 3) on a whole-brain level.
No significant associations were found between any small vessel function measure and WMHs or PSMD change (all p>0.05).
Discussion
We presented the associations between cerebral small vessel dysfunction and white matter injury progression in terms of WMHs and PSMD change in CADASIL. We found a significant negative association between mean blood flow velocity in centrum semioval perforating arteries and baseline WMHs and PSMD, which was reported previously[5]. Though WMHs and PSMD increased significantly after two-year follow-up, we found no significant association with small vessel dysfunction.Other studies into vessel function and progressive white matter injury mainly targeted the larger intracranial arteries. Pulsatility in these large arteries was found not to be associated with whole-brain WMHs increase in community-dwelling cohorts[7,8]. To our knowledge, our study assessed this relation for the first time in the smaller vessels. Small vessel blood flow velocity and pulsatility were not related to progressive white matter injury in monogenic cSVD. However in the larger arteries in monogenic cSVD, acetazolamide-induced cerebrovascular reactivity (CVR) was associated with WMHs increase[9].
A limitation of the study was that we did not correct for the follow-up time in our analysis. Another limitation was that we only did whole-brain level analyses in this study. Voxelwise analyses using BOLD-CVR to a hypercapnic stimulus might increase the sensitivity to model white matter injury progression on a local level[4,10]. This warrants further studies into the association between voxelwise small vessel dysfunction and longitudinal alterations in white matter injury in cSVDs
Conclusion
No significant associations between small vessel dysfunction and whole-brain white matter injury progression were found. Further voxelwise studies are warranted to assess the association between small vessel dysfunction and white matter injury progression.Acknowledgements
We want to thank all study participants for participating in the ZOOM@SVDs study. Furthermore, we want to thank all partners of the SVDs@target collaborative program for their efforts in the field of SVDs. ZOOM@SVDs is part of SVDs@target that has received funding from the European Union’s Horizon 2020 research and innovative program under grant agreement No. 666881. JCWS, ND and SDTP are funded by the UMCU Brain Center Young Talent Fellowship 2019.References
1. Wardlaw JM, Smith C, Dichgans M. Small vessel disease: mechanisms and clinical implications. Lancet Neurol. 2019;18:684-696.
2. Duering M, Biessels GJ, Brodtmann A, et al. Neuroimaging standards for research into small vessel disease—advances since 2013. The Lancet Neurology, 22(7), 602-618.
3. Baykara E, Gesierich B, Adam R, et al. "A novel imaging marker for small vessel disease based on skeletonization of white matter tracts and diffusion histograms." Annals of neurology 80.4 (2016): 581-592.
4. van den Brink H, Kopczak A, Arts T, et al. CADASIL Affects Multiple Aspects of Cerebral Small Vessel Function on 7T-MRI. Ann Neurol. 2022
5. Vlegels N, van den Brink H, Kopczak A, et al. The relation between small vessel function and white matter microstructure in monogenic and sporadic small vessel disease - the ZOOM@SVDs study, Manuscript in preparation
6. van den Brink H, Kopczak A, Arts T, et al. Zooming in on cerebral small vessel function in small vessel diseases with 7T MRI: Rationale and design of the “ZOOM@SVDs” study. Cereb Circ - Cogn Behav. 2021;2:100013.
7. Kneihsl, M, Hofer, E, Enzinger, C, et al. Intracranial pulsatility in relation to severity and progression of cerebral white matter hyperintensities. Stroke (2020), 51(11), 3302-3309.
8. Vikner T, Karalija N, Eklund A, et al. 5-Year Associations among Cerebral Arterial Pulsatility, Perivascular Space Dilation, and White Matter Lesions. Ann Neurol. 2022 Nov;92(5):871-881. 9. Liem MK, Lesnik Oberstein SA, Haan J, et al. Cerebrovascular reactivity is a main determinant of white matter hyperintensity progression in CADASIL. AJNR Am J Neuroradiol. 2009 Jun;30(6):1244-7.
10. Pham S, Versluis D, van den Brink H, et al. Cerebrovascular reactivity on 7T MRI is associated with longitudinal white matter hyperintensity progression in small vessel disease on 7T MRI, Poster Presentation at ISMRM 2023, Toronto, 2023
Figures
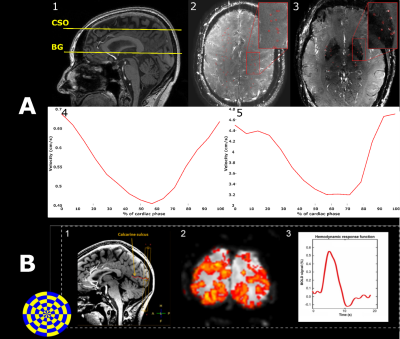
Figure 1 A) 2D-PC slice planning of the centrum semioval (CSO) and basal ganglia (BG) on a 3D T1-w image (1), with a magnitude image of the CSO (2) and of the BG (3). Arteries are marked in red. Average velocity trace over the cardiac cycle of the perforating arteries in the CSO (4) and the BG (5).
B) BOLD acquisition of the visual stimulus with examples of the (1) slice planning in the visual cortex on a 3D T1-w image, (2) voxelwise reactivity maps, and (3) BOLD signal curves over time for small vessel reactivity. The amplitude of signal was a measure for vascular reactivity.
Table 1 Acquisition parameters of relevant sequences on 3T and 7T MRI
BG = basal ganglia; BOLD = blood-oxygen level dependent; CSO = centrum semioval; FOV = field of view; GE = gradient echo; TE = echo time; TI = inversion time; TR = repetition time.*Total scan time for a heart rate of 80bpm.
Table 2 Characteristics of CADASIL patients
ICV = intracranial volume; IQR = inter-quartile range; PSMD = Peak width of the skeletonized mean diffusivity; SD = standard deviation; WMH = white matter hyperintensities * paired t-test (P<0.001) ** paired t-test (P=0.03)
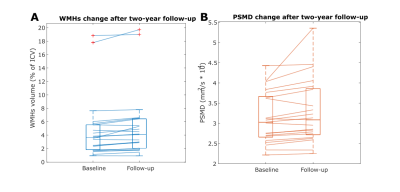
Table 3 Results of linear regression between small vessel dysfunction and white matter hyperintensities and the peak width of the skeletonized mean diffusivity for baseline and two-year change in CADASIL.
BG = basal ganglia; BOLD = blood-oxygen level-dependent; CSO = centrum semioval; HRF = hemodynamic response function; PSMD = Peak width of the skeletonized mean diffusivity; WMHs = white matter hyperintensities