2211
Signal changes of amide proton transfer weighted imaging in patients with moyamoya disease: a prospectively longitudinal study1Department of Radiology, West China Hospital, Sichuan University, Chengdu, China, 2Research Unit of Psychoradiology, Chinese Academy of Medical Sciences, Chengdu, China, 3Department of Neurosurgery, West China Hospital, Sichuan University, Chengdu, China, 4Philips Healthcare, Xi'an, China
Synopsis
Keywords: Blood Vessels, CEST & MT
Motivation: Identifying the dynamic changes of the brain microenvironment in patients with moyamoya disease (MMD) would improve disease management and treatment.
Goal(s): To explore the alteration of amide proton transfer weighted (APTw) signal in patients with MMD during the follow-up.
Approach: MMD patients who had not received revascularization were prospectively included. All patients underwent cranial computed tomography perfusion (CTP) and APTw imaging at baseline and at follow-up.
Results: APTw signal in bilateral internal carotid arteries blood supply area significantly increased in MMD patients without surgery during follow-up, and the change of APTw signal was more sensitive than that of CTP.
Impact: APTw imaging could reflect dynamic alterations of the brain microenvironment of patients with MMD over time, and the changes of APTw signal were more sensitive than CTP, which is helpful to improve clinical management.
Introduction
Moyamoya disease (MMD) is a chronic cerebral artery stenosis and occlusive disease, and patients with MMD are prone to stroke1. However, appropriate early intervention, such as revascularization or antiplatelet therapy, can prevent or delay stroke and improve the survival of MMD patients2. Amide proton transfer weighted (APTw) imaging is a non-invasive magnetic resonance imaging (MRI) technique that is highly sensitive to the PH changes of tissue3. At present, few studies have reported the application of APTw imaging in the field of MMD4. Therefore, in this prospectively longitudinal study, we used APTw imaging to investigate the changes of APTw signal in patients with MMD at follow-up.Methods
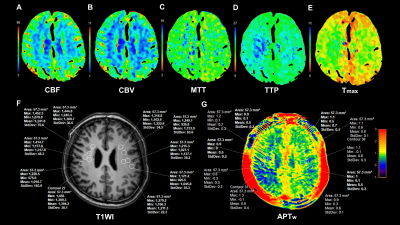
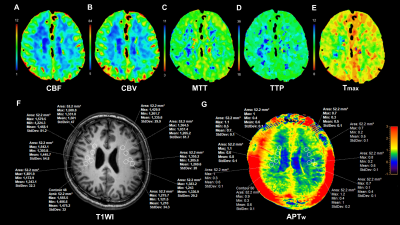
Six MMD patients (age: range 23-58 years) without surgical intervention were enrolled. All patients underwent cranial computed tomography perfusion (CTP) and APTw imaging at baseline and at follow-up. CTP was used to evaluate cerebral perfusion. MR examination was conducted on a 3.0T scanner (Ingenia Elition, Philips Healthcare, the Netherlands) with a 32-channel dedicated head coil. The structural images were acquired by using 3D-T1-TFE sequence. The APTw images were acquired by using a 3D TSE-DIXON sequence. To compare the signal changes of APTw, measurements were made at the same locations in both baseline and follow-up APTw images. For each patient, we placed multiple reference region of interests (ROIs) on the damaged perfusion area (DPA), the mirror site of the damaged perfusion area (mDPA), the normal-appearing white matter (NAWM) of the ipsilateral hemisphere of the damaged perfusion area, and the NAWM of the contralateral hemisphere. The NAWM particularly refers to the occipital white matter in the blood supply area of the posterior circulation. All the ROIs were copied from 3D-T1W to APTw images to ensure their locations were unchanged. Figures 1 and 2 show images of an MMD patient at baseline and follow-up. Two experienced neuroradiologists, with 6 and 8 years of diagnosis experience, manually drew ROIs (50-60 pixels each) independently to obtain APTw values. The intraclass correlation coefficient (ICC) was performed to evaluate the inter-observer consistency of APTw values. The paired t-test was used to compare the difference in APTw values between baseline and follow-up.Results
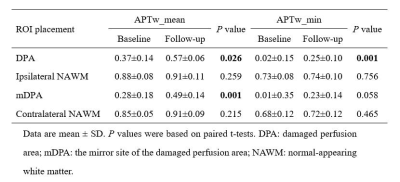
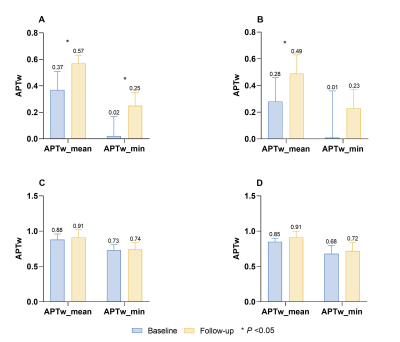
The ICC of the APTw value assessment was pretty good (ICC value>0.85). The demographic and clinical characteristics of enrolled patients with MMD are shown in Table 1. The median duration of follow-up for all patients was 12.5 months (range:11-15 months). CTP images of all patients showed unilateral impaired perfusion in the internal carotid artery supply area. Compared with the baseline APTw images, the APTw_mean and APTw_min values of DPA were significantly increased (P=0.026 and P=0.001, respectively). For the mDPA, the APTw_mean and APTw_min values of mDPA were all increased, but only the value of APTw_mean showed statistical significance at follow-up (P=0.001 and P=0.058, respectively). No significant changes were found in APTw_mean and APTw_min values of NAWM on the same side and opposite side of the DPA during the follow-up (P>0.05). Details are shown in Table 2 and Figure 3.Discussion
Our results showed that the APTw_mean and APTw_min values of the damaged perfusion area in MMD patients increased significantly with the extension of time. This indicates that without any intervention or under medical treatment, the acidosis of brain tissue in MMD patients with damaged perfusion area was reduced, and the acid-base metabolism disorder of the brain tissue was alleviated5. It's also means that PH changes in brain tissue of MMD patients are dynamic, and clinical efficacy can be reflected by APTw signals. Additionally, our results showed a significant increase in APTw_mean value of mDPA, while NAWM's APTw value remained stable. This suggests that despite normal CTP perfusion, there may be mild metabolic disturbances in the brain microenvironment of the internal carotid artery territory in MMD patients, with APTw changes being more sensitive than CTP6. These changes may result from long-term collateral circulation development, including ocular artery and leptomeningeal collateral anastomoses, which slowly improves blood supply and acid-base metabolism in both cerebral hemispheres7.While the stenosis or occlusion of vascular, the establishment of collateral circulation hardly affects the posterior circulation, which may account for the APTw signal of NAWM in both hemispheres had no significance during the follow-up.Conclusion
APTw imaging can reflect the dynamic changes of acid-base metabolism in the brain tissue of patients with MMD, and the changes of APTw signal are more sensitive than those of CTP, which is helpful to evaluate the therapeutic efficacy and improve clinical management.Acknowledgements
The authors thank the patients for participating in this study. This study was supported by grants from China Postdoctoral Science Foundation (2022M722270); the Youth Science Fund of the Natural Science Foundation of Sichuan Province, China (2022NSFSC1435); Sichuan University Postdoctoral Interdisciplinary Innovation Fund (JCXK2209); and Fund of the Beijing Medical Award Foundation (YXJL-2022-0665-0189).References
1. Ihara M, Yamamoto Y, Hattori Y, et al. Moyamoya disease: diagnosis and interventions. Lancet Neurol. 2022;21(8):747-758.
2. Seo WK, Kim JY, Choi EH, et al. Association of antiplatelet therapy, including cilostazol, with improved survival in patients with moyamoya disease in a nationwide study. J Am Heart Assoc. 2021;10(5):e017701.
3. Zhou J, Zaiss M, Knutsson L, et al. Review and consensus recommendations on clinical APT-weighted imaging approaches at 3T: application to brain tumors. Magn Reson Med. 2022;88(2):546-574.
4. Song G, Chen Y, Luo X, et al. Amide proton transfer-weighted MRI features of acute ischemic stroke subtypes.NMR Biomed. 2023;36(10):e4983.
5. Fujimura M, Tominaga T, Kuroda S, et al. 2021 Japanese Guidelines for the Management of Moyamoya Disease: Guidelines from the Research Committee on Moyamoya Disease and Japan Stroke Society. Neurol Med Chir (Tokyo). 2022;62(4):165-170.
6. Park JE, Jung SC, Kim HS, et al. Amide proton transfer-weighted MRI can detect tissue acidosis and monitor recovery in a transient middle cerebral artery occlusion model compared with a permanent occlusion model in rats. Eur Radiol. 2019;29(8):4096-4104.
7. Robert T, Cicciò G, Sylvestre P, et al. Anatomic and Angiographic Analyses of Ophthalmic Artery Collaterals in Moyamoya Disease. AJNR Am J Neuroradiol. 2018;39(6):1121-1126.
Figures