0340
Joint learning of optimal acquisition and high quality ADC mapping for low field diffusion-weighted PROPELLER MRI1Xiamen University, Xiamen, China, 2Foshan Ruijiatu Medical Technology Co., Ltd., Foshan, China, 3MSC Clinical & Technical Solutions, Philips Healthcare, Beijing, China
Synopsis
Keywords: Acquisition Methods, Low-Field MRI
Motivation: Adequate image signal-to-noise ratio (SNR) and resolution within a reasonable scan time is challenging for low-field diffusion quantitative MRI.
Goal(s): To present a PROPELLER-acquisition and ADC mapping joint learning neural network to accelerate DWI with improved image SNR and resolution.
Approach: Considering the similar anatomical structure between diffusion-weighted MR images, this work achieved DWI PROPELLER-acquisition optimization and reconstructed high quality ADC maps from data acquired by optimized acquisition using U-net.
Results: In vivo and simulation results demonstrate that our proposed method can generate high SNR and resolution ADC maps within 2 minutes scan time under 0.23T human scanner.
Impact: Joint learning acquisition and quantitative reconstruction based on PROPELLER sampling trajectory using neural network has successfully achieved fast ADC mapping, offering great possibility for quantitative analysis in low-field diffusion MRI.
Introduction
DWI is a powerful and widely applied technique in brain tumor, ischemia, and neurodegenerative disease assessment.1 DWI is vulnerable to the presence of noise, especially in low-field MRI. Diffusion-weighted PROPELLER MRI is robust to geometric distortion and motion artifacts but consumes long scan time.2-4 In recent years, the acquisition and reconstruction learning framework has shown superior performance in rapid imaging.5-8 To achieve more efficient acquisition and overcome the signal-to-noise ratio (SNR) limitation associated with low magnetic field strength, a novel framework jointly learning the PROPELLER acquisition optimization and fast ADC mapping reconstruction was proposed.Methods
Joint learning neural network: The PROPELLER-acquisition optimization network comprises three deep probabilistic subsampling (DPS) modules5. The retrospectively under-sampling scheme of fully-sampled b = 800 s/mm2 image with 25-blade was optimized at three diffusion gradient directions with non-overlapping blade position and comparable blade number. The k-space of b = 0 image was fully covered. The ADC mapping network was a modified U-net. It took a fully-sampled b = 0 image and a fully-sampled b = 800 s/mm2 diffusion-weighted image composed of three under-sampled images at three diffusion gradient directions as input. Finally, the trained ADC mapping network and optimized PROPELLER acquisition were used for real acquisition and reconstruction tasks. The joint learning neural network is shown in Figure 1.Synthetic data generation: The human brain data used for data synthesis were from Human Connectome Project Young Adult dataset, which contains b = 1000 s/mm2 diffusion-weighted images with 90 diffusion gradient directions and 18 b = 0 images acquired on a high field MRI scanner. In the process of data synthesis, we first averaged 18 b = 0 images as reference b = 0 image, and resampled the diffusion-weighted data into three predefined diffusion gradient directions (x, y, and z direction) using spherical harmonics to maintain consistency with in-vivo data. Then, the ADC map and b = 0 image were fitted from the resampled diffusion-weighted data. Finally, we synthesized diffusion-weighted PROPELLER k-space data using ADC physical model, during which the nonuniform fast Fourier transform (nuFFT) operator with PROPELLER trajectory, noise addition, zero-padding in k-space (to 256 × 256), and image normalization were also done. The number of synthetic image pairs used for training/validating/testing were 11346/1434/1419, respectively. Here, the diffusion-weighted PROPELLER k-space data are network input, and the network output is the reconstructed ADC map and b = 0 image.
In vivo experiments: In vivo data were acquired from four healthy volunteers on a 0.23T MRI scanner with an 8-channel head coil. The parameters for low SNR diffusion-weighted PROPELLER acquisition were as follows: echo time (TE) = 92 ms, repetition time (TR) = 2,414 ms, average number = 2, slice thickness = 7 mm, slice gap = 2 mm, FOV = 256 × 246 mm2, b = 0, 800 s/mm2 (3 diffusion directions for b = 800 s/mm2), 25 blades per b-value, echo train length (ETL) per shot = 8, an effective matrix diameter of 160. For acceleration, under-sampled diffusion-weighted PROPELLER was retrospectively sampled from low SNR diffusion-weighted PROPELLER. And high SNR diffusion-weighted PROPELLER with the same scan parameters but 100 blades per b-value was acquired for comparison. Healthy volunteers were employed to validate the accuracy and robustness of the trained model.
Results
Figure 2 illustrates the sampling schemes in the presented work. Uniform sampling is a uniform and non-repetitive undersampling in three diffusion directions. Figure 2a is the fully sampling scheme for b = 0 data, and Figure 2b is the uniform sampling for b = 800 s/mm2 data. Figure 2c shows that the learned sampling scheme eventually converges and the blades of each diffusion direction tend to cluster together as the training epoch increases.Figure 3 shows the results of the optimal sampling validation experiment done on synthetic testing data. Figure 3a is the visual results and Figure 3b gives the statistical results. The sampling scheme optimized by the proposed method offers ADC maps with better visual effect, lower normalized root-mean-square error (nRMSE), and higher structure similarity index measure (SSIM).
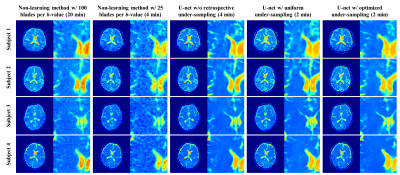
Figure 4 depicts the ADC maps of the in vivo human brain obtained from different sampling scheme and reconstruction algorithm combinations. The fifth column is the results of the proposed method. It is evident that apart from the primarily shortened acquisition time, our method also significantly reduces the image noise, eliminating image blurring and increasing image resolution.
Discussion and conclusion
The proposed method is able to reconstruct ADC maps with high image SNR and resolution from low-field PROPELLER DWI data within 2 minutes of acquisition time under 0.23T human scanner.Acknowledgements
No acknowledgement found.References
1. Baliyan V, Das CJ, Sharma R, et al. Diffusion weighted imaging: Technique and applications. World J Radiol. 2016;8(9):785-798.
2. Jeon JY, Chung HW, Lee MH, et al. Usefulness of diffusion-weighted MR imaging for differentiating between benign and malignant superficial soft tissue tumours and tumour-like lesions. Br J Radiol. 2016;89:20150929.
3. Attenberger UI, Runge VM, Stemmer A, et al. Diffusion weighted imaging: A comprehensive evaluation of a fast spin echo DWI sequence with BLADE (PROPELLER) k-space sampling at 3 T, using a 32-channel head coil in acute brain ischemia. Invest. Radiol. 2009;44:656-661.
4. Wang JC, Geng WH, Wu J, et al. Intravoxel incoherent motion magnetic resonance imaging reconstruction from highly under-sampled diffusion-weighted PROPELLER acquisition data via physics-informed residual feedback unrolled network. Phys Med Biol. 2023;68(17):175022.
5. Huijben IAM, Veeling BS, van Sloun RJG. Deep probabilistic subsampling for task-adaptive compressed sensing. In Proc In. Conf Learn Represent (ICLR). 2020;1-16.
6. van Gorp H, Huijben IAM, Veeling BS, et al. Active deep probabilistic subsampling. In Proc Int Conf Mach Learn (ICML). 2021;129:10509-10518.
7. Wang JC, Yang QQ, Yang QZ, et al. Joint optimization of Cartesian sampling patterns and reconstruction for single-contrast and multi-contrast fast magnetic resonance imaging. Comput Methods Programs Biomed. 2022;226:107150.
8. Peng W, Feng L, Zhao G, et al. Learning optimal k-space acquisition and reconstruction using physics-informed neural networks. In Proc IEEE/CVF Conf Comput Vis Pattern Recognit (CVPR). 2022;20762-20771.
Figures
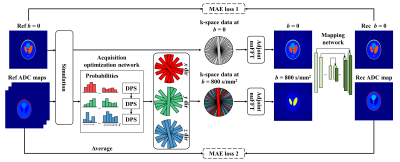
Figure 1. Framework of joint learning neural network. The synthetic PROPELLER k-space data with 25 blades per b-value were generated after simulation. The acquisition optimization network learns the optimal retrospective under-sampling scheme from 25 blades of each diffusion gradient direction. Then the mapping network (U-Net) is used to perform end-to-end ADC mapping. The total loss is the sum of MAE loss 1 and MAE loss 2.
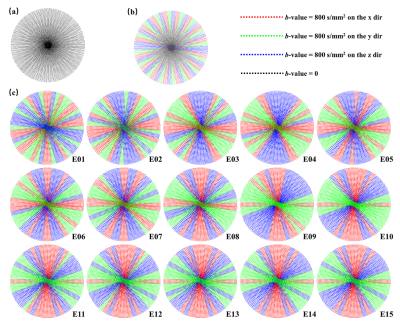
Figure 2. (a) 25-blade full sampling scheme for b = 0. (b) Uniform under-sampling for b = 800 s/mm2. (c) The learned under-sampling scheme for b = 800 s/mm2 in different training stages (E01 stands for 1st training epoch). With the number of training epoch increasing, the position of the sampling blade of each diffusion gradient direction keeps unchanged. Learned under-sampling scheme at E15 was used as the optimized under-sampling scheme for the proposed network.
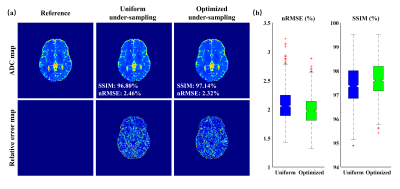
Figure 3. (a) ADC mapping results of one synthetic testing data. (b) Statistical analysis for the synthetic testing dataset. The ADC range is 0 to 4´10-3 s/mm2 and the relative error range is 50%.