3331
Altered brain function and structure in a transgenic mouse which overexpress MTH1 hydrolase following an oxidative stimulus by rs-fMRI and DTI1MRI Unit, Core Facilities, Istituto Superiore di Sanità, Rome, Italy, 2Department of Basic and Applied Sciences for Engineering, Sapienza University of Rome, Rome, Italy, 3University of Teramo, Teramo, Italy, 4Oncology and Molecular Medicine Dept., Istituto Superiore di Sanità, Rome, Italy, 5Environmental and Health Dept., Istituto Superiore di Sanità, Rome, Italy
Synopsis
Keywords: Brain Connectivity, Animals, DTI, oxidant agent
Oxidative stress is involved in the pathogenesis of cancer, neurodegeneration and aging. hMTH1 is a hydrolase able to protect cells by oxidative damage. Overexpression of hMTH1 in transgenic mice confers significant protection against oxidative damage. Our study showed alterations in the brain networks of hMTH1-Tg mice with respect to their controls. The chronical exposure to an oxidant agent (Paraquat, most widely used herbicide) causes differences in the brain function (measured by rs-fMRI) and structure (measured by DTI) of transgenic mice.
Introduction
The role of the oxidative stress is well known in the pathogenesis of cancer, neurodegeneration and aging. The Human MutT homologue (hMTH1) is a hydrolase able to protect nucleic acids from oxidative damage, by avoiding the incorporation of oxidized precursors in nuclear and mitochondrial DNA and RNA. Interestingly, transgenic mice, which overexpress the human MTH1 gene (hMTH1‐Tg) are protected from neurodegeneration and motor impairment1 and are characterized by a decreased oxidative DNA damage, a better mitochondrial functionality, a prolonged life-span and an enhanced exploratory behavior2.Aims
Aims of this study were to understand if the over-expression of hMTH1 is able to counteract the effects of a chronic exposure to an oxidant agent like the pesticide Paraquat.Methods
Male C57bl6 mice 5 month-old, wild-type (wt, n=8) and hMTH1-Tg (n=8), were analysed by rs-fMRI and DTI before, soon (acute phase) and one month (late phase) after a chronic treatment with Paraquat CL tetrahydrate (5 doses, 10 mg/kg i.p. on alternate days). Experiments were performed on a Pharmascan Bruker (Ettlingen, DE) system operating at 7T equipped with a cryo-probe.Mouse brain resting state fMRI exploiting the BOLD effect was studied by using a multi slice axial T2* EPI sequence (TR/TE=1000/13.77 ms, segments 2, bandwidth 298972 Hz, matrix 128 x 96, FOV = 20x20 mm,900 temporal points). Seed-based resting state fMRI connectivity maps for the two group of animals were generated as described in 3. In short, first voxel-wise correlation maps were obtained corresponding to a given seed (hippocampus and the pre-frontal cortex (PFC) in both hemispheres) for every session of every mouse. Then z-scores of connectivity are obtained from correlation values across all the sessions to compute significant connections at every voxel. This step is performed through a t-test in which the null hypothesis is that the connectivity is the same as a baseline value corresponding to the mean connectivity in the whole brain.
In the DTI study an EPI sequence with addition of the Stejskal-Tanner diffusion gradients was used. Diffusion gradients were applied along 30 spatial directions. Intensity, duration and diffusion time were set to 62.8 G/cm, 2.5 ms and 8.4 ms respectively, given a b-value of 1200 s/mm2. A field of view of 20 × 20 mm2 was sampled on a 128 × 128 cartesian grid. Multi-slice DT images were acquired (15 slices of 0.7 mm thickness) in the axial plane and TR/TE = 2000/18.75 ms. Using Paravision 360 software (Bruker), diffusivity values (fractional anisotropy, FA and mean diffusivity, MD) were derived from the tensor with a manual delineation of the regions of interest (Hip and PFC) on the FA and MD maps.
T-test and repeated ANOVA measurements were used for statistical comparisons (significance at p<0.05).
Results
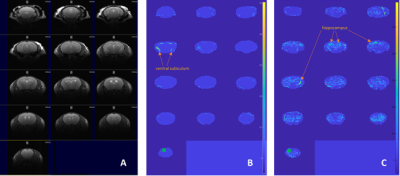
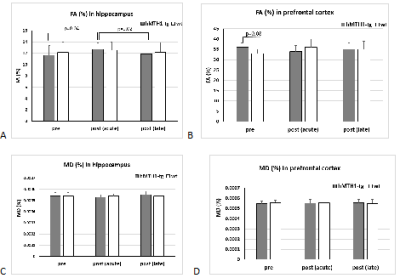
The basic finding in a typical connectivity study is that in resting subjects, spontaneous, slow (<0.1 Hz) fluctuations of the blood oxygen level dependent (BOLD) signal are temporally coherent within widely distributed functional networks. Our preliminary seed-based analysis found differences in the functional maps of networks between the wt and the hMTH1-Tg mice at baseline and after exposure to the antioxidant drug. Specifically, hippocampus (HIP) of the hMTH1-Tg mice shows diffuse increased connectivity with the cortex (mainly the auditory and somatosensory cortex) and the pre-frontal cortex (PFC) shows reduced connectivity with ventral subiculum region (See Figure 1A-B), which is the main subregion controlling the output connections from the HIP. It’s role in the mediation of hippocampal-cortical interaction is under investigation4. These results reveal different functional networks in hMTH1-tg compared to the wt mice. In the acute phase, we observed that differences in the value of connectivity with the HIP were spread all over the brain in both wt and hMTH1-tg. As an example, for the seed positioned in the PFC we detected differences in the reduction of the connectivity of hMTH1-Tg mice in ipsi- and contra-lateral HIP and other subcortical regions (Figure 1C). DTI analysis reveals structural differences, a trend of increase in FA in the PFC of the hMTH1-Tg mice compared to wt (see Figure 2). After the exposure to Paraquat we also observed an increase of FA together with a reduction of MD in the HIP of hMTH1-tg mice but not of the wt mice. No significant alteration was induced in the PFC showing a lower vulnerability of this bran region to the oxidant agent. Analysis on other relevant brain regions are currently in progress.Discussion and Conclusion
Taken together these results showed a region-dependent brain sensitivity of the transgenic mice to paraquat exposure, with HIP as the most involved area. Even if these are still preliminary results and we cannot still interpret the biological and functional significance of the detected differences, we show that our study could lead to clarify the role of oxidative damage on brain function and structure. In particular, given the pivotal role of mitochondria in brain metabolism and function, we can hypothesize that the structural and functional differences between hMTH1-Tg and wt could be linked to a diverse modulation of mitochondrial health and functionality.Acknowledgements
No acknowledgement found.References
1. De Luca G et al PLoS Genet 2008;4(11):e1000266.
2. De Luca G et al Aging Cell 2013;12(4):695-705.
3. Canese R et al Psychopharmacology (2015) 232:75–89.
4. Yu N, Song H et al, Neurosci Bull.2022
Figures