4931
Cerebral Amyloid Angiopathy (CAA) is associated with higher R2 relaxation rate: An ex-vivo MRI and pathology study1Department of Biomedical Engineering, Illinois Institute of Technology, Chicago, IL, United States, 2Rush Alzheimer's Disease Center, Rush University Medical Center, Chicago, IL, United States
Synopsis
Cerebral amyloid angiopathy (CAA) is characterized by deposition of amyloid-β protein in the walls of cortical and leptomemingeal small vessels. CAA is common in community-based older adults and has been associated with cognitive decline and dementia. To date, the association of CAA with the transverse relaxation rate, R2, remains unknown. This study in a large number of autopsied brains from community-based older adults showed for the first time that CAA is associated with higher R2, independent of other neuropathologies and demographic factors. The spatial pattern for this association derived from voxel-wise analysis included subcortical and frontal lobe structures.
INTRODUCTION
Cerebral amyloid angiopathy (CAA) is characterized by deposition of amyloid-β protein in the walls of cortical and leptomemingeal small vessels1. CAA is common in community-based older adults and has been associated with cognitive decline and dementia2-4. CAA has also been linked to intracerebral hemorrhage, microbleeds, microinfarcts, and cortical superficial siderosis5-7. However, in spite of its prevalence and deleterious effects, definitive diagnosis of CAA can only be conducted at autopsy. To date, the association of CAA with the transverse relaxation rate8,9, R2, one of the main sources of contrast in MRI, remains unknown. Therefore, the purpose of this study was to investigate the association of CAA with R2 values in a large number of autopsied brains from community-based older adults.METHODS
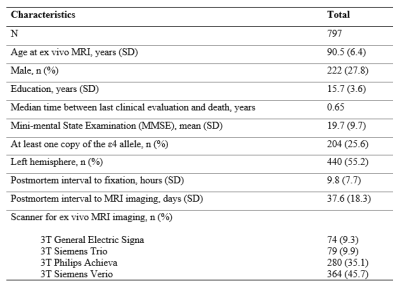
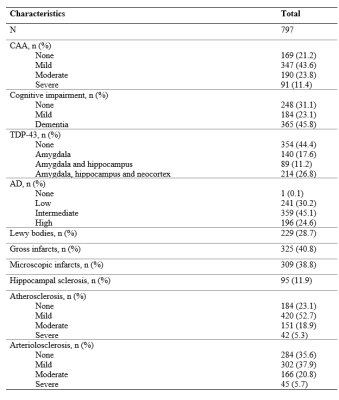
Cerebral hemispheres from 797 participants of three cohort studies of aging 10,11, the Rush Memory and Aging Project, the Minority Aging Research Study, and the Religious Orders Study (Fig. 1) were included in this work. All hemispheres were imaged ex-vivo at approximately 30 days postmortem while immersed in fixative solution at room temperature using 3T clinical MRI scanners8,9. Each scan included a multi-echo spin-echo sequence, and in each voxel, the signals from the different echoes were used to fit a mono-exponential T2 decay equation (R2 is the reciprocal of T2). The resulting R2 maps were registered to an ex-vivo brain hemisphere template using ANTS12. Following the MRI scan, each hemisphere underwent thorough neuropathologic evaluation (Fig. 2). CAA was examined in meningeal and parenchymal vessels from four brain regions: middle frontal, middle temporal, inferior parietal and calcarine cortices that were immunostained for β-amyloid. For each region, meningeal and parenchymal vessels were assessed for amyloid deposition and scored from 0 to 4. The score for each region was the maximum of the meningeal and parenchymal scores, and scores were averaged across regions and summarized as a continuous measure of CAA pathology. Other pathologies that were assessed included: Alzheimer’s pathology, LATE-NC, hippocampal sclerosis, Lewy bodies, arteriolosclerosis, atherosclerosis, gross and microscopic infarcts.Voxel-wise linear regression was conducted to test the association of R2 with CAA, controlling for all other neuropathologies listed above, demographics (age at death, sex, education), and other variables (postmortem interval to immersion in fixative, postmortem interval to ex-vivo MRI, scanners). Statistical analysis was performed using PALM (FMRIB, Oxford, UK)13 with tail-accelerated 1,000 permutations, threshold-free cluster enhancement, and family-wise error rate correction. Statistical significance was set at p<0.05.
RESULTS
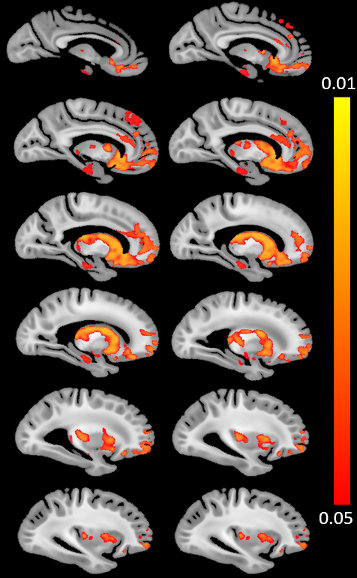
The voxel-wise analysis revealed a spatial pattern of higher R2 values for higher CAA burden (Fig.3). The pattern included subcortical structures such as the caudate, putamen, nucleus accumbens, hippocampus, as well as regions in the insula, anterior cingulate cortex, orbitofrontal cortex, and superior frontal cortex (Fig. 3). No voxel showed a negative association between R2 values and CAA burden.DISCUSSION
The present study demonstrated for the first time that CAA is independently associated with higher transverse relaxation rate R2 and extracted the spatial pattern of CAA-related R2 abnormalities, by combining ex-vivo MRI and detailed neuropathologic evaluation in brain autopsies from a large number of community-based older adults. The finding of higher R2 for higher CAA burden is particularly interesting since previous work has shown that several other vascular and neurodegenerative neuropathologies have the opposite relationship with R2, i.e. lower R2 for higher Alzheimer’s burden, more infarcts, or hippocampal sclerosis8. The interpretation of the lower R2 observed with those neuropathologies was that it was a result of neuronal dropout, astrocytosis and increased water content. In the case of CAA, however, we observed higher R2 which cannot be justified by the above factors. Considering that CAA has been linked to microbleeds, intracerebral hemorrhage, and cortical superficial siderosis (i.e. bleeding at microscopic or large scale), and that the hemosiderin deposited in these cases lowers T2 and T2* values (increases R2 values), we hypothesize that the association of CAA with higher R2 may be a result of increased levels of hemosiderin. Furthermore, the spatial pattern extracted in the present work included mainly subcortical and frontal lobe structures, sparing the parietal, occipital and temporal lobes. Neuropathology studies have shown that CAA is most prevalent in meningeal and parenchymal vessels in mainly posterior portions of the brain14. This suggests that what we observed in the present work is most likely a downstream effect linked to CAA and not a result of direct tissue damage in the vicinity of CAA pathology. Future work will focus on understanding the neurobiologic mechanisms supporting our findings. Finally, although the present study was conducted ex-vivo, we expect our ex-vivo results to hold in-vivo since we have previously shown that ex-vivo R2 is linearly related to in-vivo R2 for the same tissue preparation and imaging protocol used here8.CONCLUSION
The present work in autopsied brains from a large number of community-based older adults showed for the first time that CAA is associated with higher transverse relaxation rate R2, independent of other neuropathologies and demographic factors. The spatial pattern for this association derived from the voxel-wise analysis included subcortical and frontal lobe structures. These findings may potentially contribute towards in-vivo prediction of CAA.Acknowledgements
National Institute of Neurological Disorders and Stroke (NINDS) UH2-UH3NS100599
National Institute of Neurological Disorders and Stroke (NINDS) UF1NS100599
National Institute on Aging (NIA) R01AG064233
National Institute on Aging (NIA) R01AG067482
National Institute on Aging (NIA) R01AG017917
National Institute on Aging (NIA) R01AG015819
National Institute on Aging (NIA) RF1AG022018
National Institute on Aging (NIA) R01AG056405
National Institute on Aging (NIA) P30AG010161
National Institute on Aging (NIA) P30AG072975References
1. Vinters HV. Cerebral amyloid angiopathy. A critical review. Stroke. 1987;18(2):311–324.
2. Boyle PA, Yu L, Nag S, et al. Cerebral amyloid angiopathy and cognitive outcomes in community-based older persons. Neurol. 2015;85(22):1930–1936.
3. Kalaria RN, and Ballard C. Overlap between pathology of Alzheimer disease and vascular dementia. Alzheimer Dis Assoc Disord. 1999;13 Suppl 3:S115–S123.
4. Arvanitakis Z, Leurgans SE, Wang Z, et al. Cerebral amyloid angiopathy pathology and cognitive domains in older persons. Ann Neurol. 2011;69(2):320–327.
5. Greenberg SM, Al-Shahi Salman R, Biessels GJ, et al. Outcome markers for clinical trials in cerebral amyloid angiopathy. Lancet Neurol. 2014;13(4):419–428.
6. Chao CP, Kotsenas AL and Broderick DF. Cerebral Amyloid Angiopathy: CT and MR Imaging Findings. Radiographics. 2006;26(5):1517–1531.
7. van Veluw SJ, Charidimou A, van der Kouwe AJ, et al. Microbleed and microinfarct detection in amyloid angiopathy: a high-resolution MRI-histopathology study. Brain. 2016; 139(Pt 12):3151–3162.
8. Dawe RJ, Bennett DA, Schneider JA, et al. Ex vivo T2 relaxation: Associations with age-related neuropathology and cognition. Neurobiol Aging. 2014;35:1549–1561.
9. Yu L, Dawe RJ, Buchman AS, et al. Ex vivo MRI transverse relaxation in community based older persons with and without Alzheimer’s dementia. Behav Brain Res. 2017;322:233–240.
10. Barnes LL, Shah RC, Aggarwal NT, et al. The Minority Aging Research Study: Ongoing Efforts to Obtain Brain Donation in African Americans without Dementia. Curr Alzheimer Res. 2013;9:734–745.
11. Bennett DA, Buchman AS, Boyle PA, et al. Religious Orders Study and Rush Memory and Aging Project. J Alzheimer’s Dis. 2018;64:S161–S189.
12. Avants B, Tustison N, Song G. Advanced Normalization Tools (ANTS). Insight J 2009;1–35.
13. Winkler AM, Ridgway GR, Webster MA, et al. Permutation inference for the general linear model. Neuroimage. 2014;92:381–397.
14. Kövari E, Herrmann FR, Hof PR, et al. The relationship between cerebral amyloid angiopathy and cortical microinfarcts in brain ageing and Alzheimer's disease. Neuropathol Appl Neurobiol. 2013;39(5):498-509
Figures