4278
Lung cancer in multi-parametric PET/MRI: assessment of subtype and EGFR status based on amide proton transfer-weighted imaging and 18F-FDG PET1Department of Medical Imaging, Zhengzhou University People’s Hospital & Henan Provincial People’s Hospital, Zhengzhou, China, 2Department of Medical Imaging, Henan Provincial People’s Hospital, Zhengzhou, China, 3Department of Medical Imaging, Henan University People’s Hospital & Henan Provincial People’s Hospital, Zhengzhou, China, 4Department of Medical Imaging, Xinxiang Medical University Henan Provincial People’s Hospital, Zhengzhou, China, 5Central Research Institute, UIH Group, Shanghai, China, 6Beijing United Imaging Research Institute of Intelligent Imaging, UIH Group, Beijing, China, 7UIH America, Houston, TX, United States
Synopsis
18F-Fluorodeoxyglucose positron-emission tomography/magnetic resonance imaging (18F-FDG PET/MRI) allows multimodal quantitative MRI sequences to be scanned in parallel with PET imaging, providing a more multidimensional reflection of lesion information. Our results showed that amide proton transfer-weighted imaging (APTWI), and metabolism related parameters can be beneficial for the non-invasive assessment of subtype and EGFR status in patients with lung cancer.
Introduction
Accurate non-invasive assessment of histological subtypes and epidermal growth factor receptor (EGFR) expression status of lung cancer is of great significance for the diagnosis and prognosis of the patients involved [1, 2]. Multi-parametric positron-emission tomography / magnetic resonance imaging (PET/MRI), including 18F-FDG PET, diffusion-weighted imaging (DWI), and amide proton transfer- weighted imaging (APTWI), is sensitive to the glucose metabolism, water molecular diffusion, and mobile protein and peptide metabolism in biological tissues [3, 4, 5]. The purpose of this study was to use a hybrid PET/MRI imaging modality to investigate the diagnostic accuracy of APTWI-, DWI-, and 18F-FDG PET-related parameters in distinguishing small cell lung cancer (SCLC) from non-small cell lung cancer (NSCLC), EGFR mutation from EGFR wild-type NSCLC, in turn providing new insights for the clinical diagnosis and prognostic assessment of lung cancer patients.Material and Methods
A total of 99 enrolled subjects were were included in this study. A hybrid 3.0 T PET/MR system (uPMR 790, UIH, Shanghai, China) with a 12-channel phased-array body coil was was performed. The PET scan was initiated 60 minutes after injection of 18F-FDG (0.11 mCi/kg). A Dixon method-based water-fat imaging sequence was performed for attenuation correction and ordered subset maximum expected iteration methods were used to reconstruct the images. The DWI was performed by using two b values (0, 800 s/mm2). For APTWI: ETL = 39, B1 = 1.3 μT and 2.5 μT, Gaussian pulse, 100 ms duration, 10 repeats, Δ spanned from [-4.5 4.5] ppm in 31 steps, plus one S0 with no CEST saturation pulse for normalization; 11 low power B1 = 0.13 μT, Δ spanned from [-1.0 1.0] ppm images were collected as WASSAR images for B0 map correction. All images were performed with specialized software in the advanced-analysis toolkit of the UIH Workstation (uWS-MR: R005). SPSS 26.0 and R 3.53 were employed for statistical analyses. The Mann-Whitney U test or independent-sample t-test was utilized for data comparison. The area under the receiver operating characteristic (ROC) curve (AUC) was applied to describe the diagnostic efficacy, and the differences were compared using the DeLong test. Logistic regression analyses were performed to filter the best combination of parameters. A control model was established by the bootstrap (1000 samples), and its performance was verified with calibration curves and ROC curves.Results
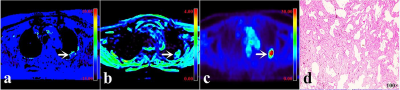
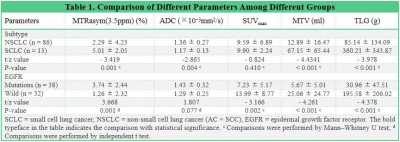
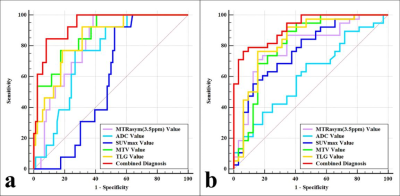
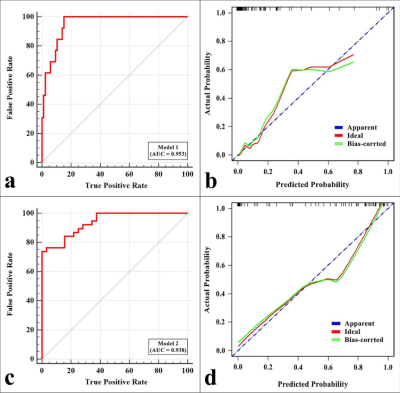
MTRasym (3.5 ppm), MTV, and TLG were lower and ADC was higher in NSCLC than in SCLC, MTRasym (3.5 ppm) was higher and SUVmax , MTV, and TLG were lower in EGFR mutation-type NSCLC than in EGFR wild-type NSCLC (all P < 0.05), and there was no significant difference in SUVmax between NSCLC and SCLC , and no significant difference in ADC between EGFR mutation-type NSCLC and EGFR wild-type NSCLC (Figure.1, Table 1). Logistic analysis showed that the combination of MTRasym (3.5 ppm), ADC and MTV, and the combination of MTRasym (3.5 ppm) and MTV had the strongest predictive value for the differentiation of SCLC and NSCLC, and the differentiation of EGFR mutation-type and wild-type NSCLC, respectively (AUC, 0.942, 0.919; sensitivity, 84.62 %, 76.32%; specificity, 93.02%, 93.75%; P all < 0.001, Figure.2). The ROC curve and calibration curve of verification model showed good accuracy (AUC = 0.953, 0.938) and consistency, respectively (Figure.3).Discussion
SCLC is a neuroendocrine tumor and is one of the most malignant lung cancer subtypes. Compared with NSCLC, it is more aggressive with more active cell proliferationa and a certain peptide hormone secretion ability [6, 7], which limits the diffusion of water molecules and simultaneously increases glucose metabolism and mobile protein and peptide content, so it has smaller ADC values and larger MTV, TLG and MTRasym (3.5 ppm) values. In the comparison EGFR mutation-type and wild-type NSCLC, SUVmax, MTV, and TLG all showed good diagnostic performance, and we suggest that the higher glucose transporter-1 (Glut-1) expression of EGFR wild-type NSCLC may be the main reasons for their increased these PET parameters compared with EGFR mutation-type NSCLC [8]. This study also found that the MTRasym (3.5 ppm) of the EGFR mutation-type was significantly higher than that of the wild-type NSCLC. We consider that the greater aggressiveness [9] and higher proportion of adenocarcinoma in EGFR mutation-type NSCLC may be potential reasons for this outcome.Conclusion
Subtype and EGFR status are important prognostic factors in lung cancer, the combination of MTRasym (3.5 ppm), ADC and MTV, and the combination of MTRasym (3.5 ppm) and MTV may be effective imaging markers for differentiating SCLC from NSCLC, and EGFR mutation-type from wild-type NSCLC, respectively. Based on these results, a personalised treatment plan can be developed for patients with lung cancer.Acknowledgements
This work was supported by the National Key R&D Program of China (2017YFE0103600), the National Natural Science Foundation of China (81720108021 and 31470047), the Zhongyuan Thousand Talents Plan Project - Basic Research Leader Talent (ZYQR201810117), the Zhengzhou Collaborative Innovation Major Project (20XTZX05015), and the Key Project of Henan Province Medical Science and Technology Project (LHGJ20210001, LHGJ20210005, LHGJ20190602), and the Henan provincial science and technology research projects (212102310689).References
1. Rudin CM, Brambilla E, Faivre-Finn C, Sage J. Small-cell lung cancer. Nat Rev Dis Primers. 2021;7(1):3.
2. Uchibori K, Inase N, Araki M, Kamada M, Sato S, Okuno Y, et al. Brigatinib combined with anti-EGFR antibody overcomes osimertinib resistance in EGFR-mutated non-small-cell lung cancer. Nat Commun. 2017;8:14768.
3. Madsen PH, Holdgaard PC, Christensen JB, Høilund-Carlsen PF. Clinical utility of F-18 FDG PET-CT in the initial evaluation of lung cancer. Eur J Nucl Med Mol Imaging. 2016;43(11):2084-2097.
4. Shen G, Jia Z, Deng H. Apparent diffusion coefficient values of diffusion-weighted imaging for distinguishing focal pulmonary lesions and characterizing the subtype of lung cancer: a meta-analysis. Eur Radiol. 2016;26(2):556-566.
5. Zhou J, Payen JF, Wilson DA, Traystman RJ, van Zijl PC. Using the amide proton signals of intracellular proteins and peptides to detect pH effects in MRI. Nat Med. 2003; 9(8): 1085-1090.
6. Togao O, Yoshiura T, Keupp J, et al. Amide proton transfer imaging of adult diffuse gliomas: correlation with histopathological grades. Neuro Oncol. 2014;16(3): 441-448.
7. Ohno Y, Yui M, Koyama H, et al. Chemical Exchange Saturation Transfer MR Imaging: Preliminary Results for Differentiation of Malignant and Benign Thoracic Lesions. Radiology. 2016;279(2):578-589.
8. Wang Y, Zhao N, Wu Z, et al. New insight on the correlation of metabolic status on 18F-FDG PET/CT with immune marker expression in patients with non-small cell lung cancer. Eur J Nucl Med Mol Imaging. 2020;47(5):1127-1136.
9. Shin DY, Na II, Kim CH, Park S, Baek H, Yang SH. EGFR mutation and brain metastasis in pulmonary adenocarcinomas. J Thorac Oncol. 2014;9(2):195-199.
Figures