2495
Quantitative evaluation of renal injury in the early chronic kidney disease with quantitative susceptibility mapping1Third Affiliated Hospital of Soochow University, Changzhou, China, China, 2Philips Healthcare, Beijing, China, 3Philips Healthcare, Shanghai, China
Synopsis
To explore the value of quantitative susceptibility mapping (QSM) in evaluating renal injury in patients with chronic kidney disease (CKD). 40 CKD patients were included in the study to evaluate the potential clinical value of QSM. We found that with the progress of CKD staging, renal medulla susceptibility values decreased significantly. Susceptibility value of the medulla was highly correlated with estimated glomerular filtration rate (eGFR). QSM could serve as a quantitative biomarker to assess the renal injury of early CKD.
Purpose
The prevalence of chronic kidney disease in the general population is increasing[1, 2], but there is a lack of sensitive and non-invasive detection methods, especially in the early stage. So far, a number of studies have reported that QSM could be used to measure pathologic deposits in basal ganglia in various neurological diseases[3], or as an imaging biomarker of hepatic iron overload[4, 5]. However, it remains unknown whether QSM could serve as a noninvasive biomarker to assess the renal injury. Therefore, the purpose of this study was to explore if QSM can assess the degree of renal injury in CKD and to associate susceptibility value with eGFR.Materials and Methods
Study populationFrom October 2019 to April 2021, 45 patients (32 males and 13 females, mean age 57 years) in the Third Affiliated Hospital of Soochow University were collected. The specific inclusion criteria were as follows: diagnosis of CKD; complete clinical data; eGFR>30ml•min-1·1.73m-2; no MRI examination contraindications. From this group, three cases were excluded due to the following reasons: (1) poor image quality (n = 2) and (2) large (>2 cm) or multiple (more than 3) simple kidney cysts or other lesions in the kidney (n = 3). At last, 40 cases were included in this study. This group was divided into the CKD1 group (eGFR≥90ml•min-1·1.73m-2, n=11),CKD2 group (60≤eGFR<90 ml•min-1·1.73m-2, n=18) and CKD3 group (30≤eGFR<60 ml•min-1·1.73m-2, n=11).
MRI protocol
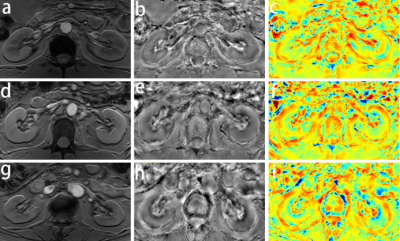
All patients were performed with a 3.0-T Ingenia MR scanner (Philips Healthcare, the Netherlands) using a 16 channel body coil. QSM data were acquired using an axial single-breath-hold 3D multi-echo gradient echo sequence with the following parameters: number of echoes = 5; TE1/ ΔTE/TR = 7.2/5.2/32 ms; flip angle = 17°; acquisition matrix = 268 × 254 mm2; voxel size = 1.3 × 1.5 × 5 mm3; bandwidth = 254.9 Hz/pixel; acquisition time 14s.
Statistical analysis
All the acquired images were post-processed on the software package STISuiteV3.0 in MATLAB(R2016b) to obtain the susceptibility map. The ImageJ software was used to manually draw regions of interest (ROIs) in cortex and medulla of bilateral kidneys and obtain the mean susceptibility value.
All statistical tests were performed using SPSS 26.0 software and MedCalc 15.2 software, and statistical significance was set at P < 0.05. The paired-sample t test was used to compare the difference of susceptibility between renal cortex and medulla and between left and right kidneys. The mean renal cortical and medullary susceptibility values among three groups were analyzed using one-way analysis of variance. Post-hoc multiple pairwise comparisons were performed with the least-significant difference test. Pearson correlation analysis was used to correlate the susceptibility value with eGFR. Receiver operating characteristic (ROC) curve was used to analyze the diagnostic efficacy of QSM in the staging of early CKD and the optimal cut-off-values were determined.
Results
The susceptibility values of the left renal cortex and medulla in patients with CKD were (9.78 ±11.03) ×10-3ppm and (-7.71±2.18) ×10-2ppm, respectively, and the difference was statistically significant (t = 23.278, P < 0.001). The susceptibility values of right renal cortex and medulla were (6.91±9.42) ×10-3ppm and (-7.32±2.15) ×10-2ppm, respectively, and the difference was statistically significant (t = 23.070, P < 0.001). In addition, there was no significant difference between left and right renal cortex (t = 1.917, P = 0.063), but there was significant difference between left and right renal medulla (t = -2.417, P = 0.020). The susceptibility values of renal cortex in CKD 1, 2 and 3 were (12.57±8.37) ×10-3ppm, (8.92±13.66) ×10-3ppm, (8.41±8.66) ×10-3ppm (left kidney) and (8.89±10.20) ×10-3ppm, (7.24±9.59) ×10-3ppm and (4.41±8.61) ×10-3ppm (right kidney) respectively. There was no significant difference among these three groups (P > 0.05). The susceptibility values of renal medulla in CKD 1, 2 and 3 were (-5.50±1.30) ×10-2ppm, (-7.64±0.88) ×10-2ppm, (-10.04±2.05) ×10-2ppm (left kidney) and (-5.01±1.39) ×10-2ppm, (-7.15±0.87) ×10-2ppm, (-9.90±1.24) ×10-2ppm (right kidney) respectively. There were significant differences among these three groups (P < 0.05). In addition, the susceptibility values of the left and right medulla were significantly correlated with eGFR (left: r=0.787 and right: r=0.871, respectively; P<0.001). For left renal medulla, a cut-off-value of -0.070 could be identified to detect CKD 1 and CKD (2 and 3) with a sensitivity of 90.91% and a specificity of 89.66%, while a cut-off-value of -0.096 could be identified to detect CKD (1 and 2) and CKD 3 with a sensitivity of 100.00% and a specificity of 72.73%. For right renal medulla, a cut-off-value of -0.068 could be identified to detect CKD 1 and CKD (2 and 3) with a sensitivity of 100.00% and a specificity of 82.76%, while a cut-off-value of -0.085 could be identified to detect CKD (1 and 2) and CKD 3 with a sensitivity of 96.55% and a specificity of 90.91%.Conclusion
We found that with the progress of CKD staging, renal medulla susceptibility values decreased significantly. Susceptibility value of the medulla was highly correlated with estimated glomerular filtration rate (eGFR). Renal susceptibility may be a promising marker for assessing the degree of renal injury. With further study, QSM can potentially be used as an alternative tool for the noninvasive detection and diagnosis of the severity of renal injury in patients with CKD.Acknowledgements
No acknowledgement found.References
[1] Glassock R J, Warnock D G, Delanaye P. The global burden of chronic kidney disease: estimates, variability and pitfalls[J]. Nat Rev Nephrol,2017,13(2):104-114.
[2] Mills K T, Xu Y, Zhang W, et al. A systematic analysis of worldwide population-based data on the global burden of chronic kidney disease in 2010[J]. Kidney Int,2015,88(5):950-957.
[3] Li D, Liu Y, Zeng X, et al. Quantitative Study of the Changes in Cerebral Blood Flow and Iron Deposition During Progression of Alzheimer's Disease[J]. J Alzheimers Dis,2020,78(1):439-452.
[4] Deh K, Zaman M, Vedvyas Y, et al. Validation of MRI quantitative susceptibility mapping of superparamagnetic iron oxide nanoparticles for hyperthermia applications in live subjects[J]. Sci Rep,2020,10(1):1171.
[5] Simchick G, Liu Z, Nagy T, et al. Assessment of MR-based R2* and quantitative susceptibility mapping for the quantification of liver iron concentration in a mouse model at 7T[J]. Magn Reson Med,2018,80(5):2081-2093.
Figures