1832
Quasiperiodic Patterns in resting state fMRI signal reflect dynamics of cortical slow rhythms1Emory University, Atlanta, GA, United States
Synopsis
In this study, we tested hypothesis advanced by some groups that brain slow rhythms serve as the neurophysiological basis of resting state fMRI (rsfMRI). Putative suppression of cortical rhythms with an established technique, led to significant reduction in the amplitude of rsfMRI quasi-periodic patterns (QPPs), and enhancement in the rsfMRI measures of intrinsic functional connectivity FC in canonical brain function networks in rats. The results indicate cortical slow rhythms serve as the genesis of only the vigilance dependent components (e.g., QPP) of rsfMRI signals. Further attenuation of these non-specific signals enhances delineation of brain function networks.
INTRODUCTION
The neurophysiological basis underlying resting state fMRI (rsfMRI) signals are not completely understood, impeding accurate interpretation of rsfMRI studies. A number of studies 1,2 point to slow rhythms (0.5-2 Hz) as the basis of rsfMRI. Slow rhythms exist in the absence of stimulation, propagate across the cortex 3, and are strongly modulated by vigilance 4 similar to parts of rsfMRI signals 5,6 like quasi-periodic patterns (QPPs). However, unlike slow rhythms, the strength of FC in brain function resting state networks (RSNs) decreases with reductions in vigilance and arousal levels 7,8. It is possible that slow rhythms provide the basis of only certain (e.g., vigilance dependent) components of the rsfMRI signals like QPPs, which are not specific to and can confound the accurate estimation of FC in RSNs.One mechanism for expression and maintenance of cortical slow rhythms in the brain is through a thalamocortical network of coupled oscillators driven by burst firing in thalamus induced by low-threshold T-type calcium (Ca2+) channels (TTCCs) 9,10. Systemic administration of the selective TTCC TTA-P2 11 suppresses cortical slow brain rhythms by up to 60% in anesthetized rats 12. In this study, preliminarily presented earlier, we examined the effects of TTA-P2 on rsfMRI signal in rats. We hypothesized that slow wave suppression would reduce the strength of QPPs, which in turn will enhance rsfMRI measures of FC in brain function networks.
METHODS
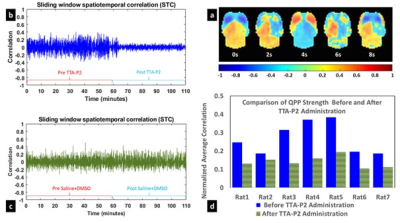
All experiments were conducted with protocols approved by IACUC. Seven adult Sprague-Dawley rats were administered subcutaneous injections of TTA-P2 (3-6 mg/kg dissolved in Vehicle (4% DMSO saline solution)), immediately after and before 40-90 min fMRI scans obtained under sedation induced by dexmedetomidine (which does not interfere with the action of TTA-P2 13). Five other rats were administered the Vehicle. MRI data were acquired on a 9.4 T Bruker animal MRI system with a custom-built surface coil. RsfMRI scans were obtained with a whole-brain respiration-gated gradient echo EPI (TR/TE/FA = 2000ms/25ms/90°, resolution = 0.5 mm isotropic voxels). Data was preprocessed with standard pipelines 14,15, band-passed filtered (0.01-0.20 Hz) and aligned to Paxinos atlas space 16,17. QPP templates were estimated with a well-established technique 14 from the pre-injection (Baseline) fMRI data. The changes in the strength of the expression of QPPs over time for each fMRI scan (Baseline and TTA-P2/vehicle) for each rat were estimated through the sliding window spatiotemporal correlation (STC) of the corresponding fMRI time-series with that rat’s QPP template. The effects of TTA-P2/vehicle on QPPs were assessed with between-session (TTA-P2 vs Baseline) paired t-tests (as well as Wilcoxon Signed Rank tests (WSRTs) on the mean of positive excursions of the STC curve above zero. We employed interhemispheric homotopic FC (IHFC) of 40 different cortical regions of interest (ROIs) defined in the Paxinos atlas to examine FC in RSNs. Group analysis was conducted through between-session t-tests (and WSRTs) on the z-transformed CC between homotopic cortical ROIs.RESULTS & DISCUSSION
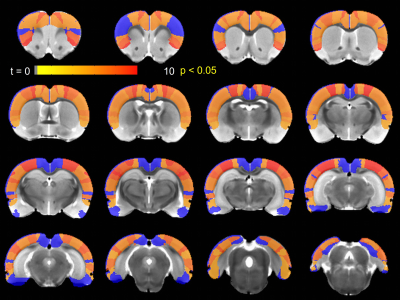
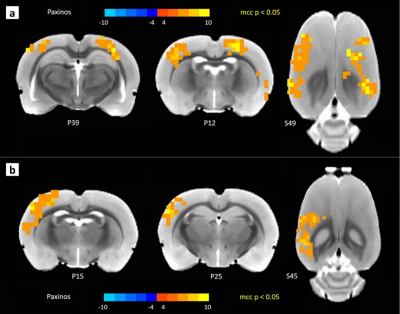
Fig.1.a shows a QPP with anterior to posterior propagation. The fluctuations in the strength of the QPP before and after injection of TTA-P2 or saline are shown for a representative rat administered TTA-P2 (Fig.1.b) and one given vehicle (Fig.1.c). TTA-P2 administration significantly (p < 0.01) reduced the strength (mean of positive STC values) of QPPs compared to Baseline. The amount of suppression of QPPs induced by TTA-P2 varied from 18-58% (mean 48%) across the rats (see Fig1.d). Vehicle did not alter the strength of the QPPs. Thus, suppression of cortical slow rhythms (putatively induced by TTA-P2 12) led to expected reduction in the strength of QPPs. This confirms our hypotheses that QPPs depend on/reflect the expression of cortical slow rhythms. Next, we examined IHFC of 40 cortical ROIs, defined according to the Paxinos atlas. Thirty-one of the forty ROIs exhibited (Fig.2) significantly (p < 0.05) increased IHFC after injection of TTA-P2. Most of the other 9 ROIs were in heteromodal regions (e.g., frontal and associative cortices), which generally do not exhibit strong IHFC in rsfMRI studies 18,19. The rest were very small in spatial extent and hence more susceptible to noise. The changes in the whole-brain FC maps obtained with sbCCA of two representative regions (right hemisphere rat barrel cortex (RS1-BF) and right auditiory cortex (RAud) are shown in Fig.3. TTA-P2 significantly increases the rsfMRI FC between RS1-BF (Fig.3a) and some areas in somatosensory, motor, auditory, visual, and parietal cortices, bilaterally consistent with increased in corresponding canonical brain function networks 20-23. RAud exhibited (Fig.3b) significantly increased FC to contralateral auditory, visual and somatosensory areas which enhanced the delineation of related brain circuits 22,24,25. Vehicle administration did not evoke appreciable changes in FC. Thus, TTA-P2 induced suppression of cortical slow rhythms enhances the detectability of FC in RSNs as expected due to the reduction of non-specific QPP signals.CONCLUSION
The results indicate that the vigilance dependent components of the rsfMRI signal (e.g. QPPs) reflect the dynamics of cortical slow rhythms. These fMRI signals are strongly attenuated by suppression slow rhythms, which enhances FC derived from rsfMRI in RSNs. These results have profound implications on the neurophysiological basis of rsfMRI signals. Future work would include simultaneous EEG recordings to directly examine cortical slow rhythms.Acknowledgements
This work was supported by Radiology Seed Grants from Department of Radiology & Imaging Sciences, Emory UniversityReferences
1. Chan RW, Leong ATL, Ho LC, et al. Low-frequency hippocampal-cortical activity drives brain-wide resting-state functional MRI connectivity. Proc Natl Acad Sci U S A 2017.
2. Matsui T, Murakami T, Ohki K. Transient neuronal coactivations embedded in globally propagating waves underlie resting-state functional connectivity. Proc Natl Acad Sci U S A 2016;113:6556-61.
3. Sheroziya M, Timofeev I. Global intracellular slow-wave dynamics of the thalamocortical system. J Neurosci 2014;34:8875-93.
4. Steriade M. Corticothalamic resonance, states of vigilance and mentation. Neuroscience 2000;101:243-76.
5. Billings JCW, Keilholz SD. The Not-So-Global BOLD Signal. Brain Connect 2018.
6. Thompson GJ, Magnuson ME, Merritt MD, et al. Short-time windows of correlation between large-scale functional brain networks predict vigilance intraindividually and interindividually. Hum Brain Mapp 2013;34:3280-98.
7. Hutchison RM, Hutchison M, Manning KY, Menon RS, Everling S. Isoflurane induces dose-dependent alterations in the cortical connectivity profiles and dynamic properties of the brain's functional architecture. Hum Brain Mapp 2014.
8. Larson-Prior LJ, Zempel JM, Nolan TS, Prior FW, Snyder AZ, Raichle ME. Cortical network functional connectivity in the descent to sleep. Proc Natl Acad Sci U S A 2009;106:4489-94.
9. Crunelli V, David F, Lorincz ML, Hughes SW. The thalamocortical network as a single slow wave-generating unit. Curr Opin Neurobiol 2015;31:72-80.
10. Crunelli V, Hughes SW. The slow (<1 Hz) rhythm of non-REM sleep: a dialogue between three cardinal oscillators. Nat Neurosci 2010;13:9-17.
11. Dreyfus FM, Tscherter A, Errington AC, et al. Selective T-type calcium channel block in thalamic neurons reveals channel redundancy and physiological impact of I(T)window. J Neurosci 2010;30:99-109.
12. David F, Schmiedt JT, Taylor HL, et al. Essential thalamic contribution to slow waves of natural sleep. J Neurosci 2013;33:19599-610.
13. Horvath G, Morvay Z, Kovacs M, Szilagyi A, Szikszay M. Drugs acting on calcium channels modulate the diuretic and micturition effects of dexmedetomidine in rats. Life Sci 1996;59:1247-57.
14. Majeed W, Magnuson M, Hasenkamp W, et al. Spatiotemporal dynamics of low frequency BOLD fluctuations in rats and humans. Neuroimage 2011;54:1140-50.
15. Pan WJ, Billings J, Nezafati M, Abbas A, Keilholz S. Resting State fMRI in Rodents. Curr Protoc Neurosci 2018;83:e45.
16. Paxinos G, Watson C. Paxino's and Watson's The rat brain in stereotaxic coordinates. Seventh edition. ed. Amsterdam ; Boston: Elsevier/AP, Academic Press is an imprint of Elsevier; 2014.
17. Valdes-Hernandez PA, Sumiyoshi A, Nonaka H, et al. An in vivo MRI Template Set for Morphometry, Tissue Segmentation, and fMRI Localization in Rats. Front Neuroinform 2011;5:26.
18. Stark DE, Margulies DS, Shehzad ZE, et al. Regional variation in interhemispheric coordination of intrinsic hemodynamic fluctuations. J Neurosci 2008;28:13754-64.
19. Shen K, Misic B, Cipollini BN, et al. Stable long-range interhemispheric coordination is supported by direct anatomical projections. Proc Natl Acad Sci U S A 2015;112:6473-8.
20. Rao RP, Mielke F, Bobrov E, Brecht M. Vocalization-whisking coordination and multisensory integration of social signals in rat auditory cortex. eLife 2014;3.
21. Frostig RD, Xiong Y, Chen-Bee CH, Kvasnak E, Stehberg J. Large-scale organization of rat sensorimotor cortex based on a motif of large activation spreads. J Neurosci 2008;28:13274-84.
22. Reid JM, Jacklin DL, Winters BD. Crossmodal object recognition in rats with and without multimodal object pre-exposure: no effect of hippocampal lesions. Neurobiol Learn Mem 2012;98:311-9.
23. Zakiewicz IM, Bjaalie JG, Leergaard TB. Brain-wide map of efferent projections from rat barrel cortex. Front Neuroinform 2014;8:5.
24. Schormans AL, Scott KE, Vo AM, et al. Audiovisual Temporal Processing and Synchrony Perception in the Rat. Frontiers in behavioral neuroscience 2016;10:246.
25. Thomas ME, Lane CP, Chaudron YMJ, Cisneros-Franco JM, de Villers-Sidani E. Modifying the Adult Rat Tonotopic Map with Sound Exposure Produces Frequency Discrimination Deficits That Are Recovered with Training. J Neurosci 2020;40:2259-68.
Figures