0025
Feasibility of single breath-hold CINE with combined Simultaneous Multi-Slice (SMS) and Region-Optimized Virtual (ROVir) coils1Department of Electrical and Computer Engineering, University of Southern California, Los Angeles, CA, United States, 2A. A. Martinos Center for Biomedical Imaging, Department of Radiology, Massachusetts General Hospital, Charlestown, MA, United States, 3Cardiovascular Research Center, Cardiology Division, Massachusetts General Hospital, Charlestown, MA, United States, 4Department of Medicine, Harvard Medical School, Boston, MA, United States, 5Department of Radiology, Massachusetts General Hospital, Charlestown, MA, United States, 6Department of Radiology, Harvard Medical School, Boston, MA, United States, 7Siemens Medical Solutions USA Inc., Chicago, IL, United States, 8Siemens Healthcare Pty Ltd, Melbourne, Australia, 9Siemens Healthcare GmbH, Erlangen, Germany, 10Siemens Medical Solutions USA Inc., Los Angeles, CA, United States
Synopsis
Conventional clinical cardiac MRI protocols use a large number (>20) of breath-holds for capturing cinemagraphic (CINE) scans of the heart in various views. We hypothesize simultaneous multi-slice (SMS) CINE can be further accelerated using a reduced FOV and a novel approach based on Region-Optimized Virtual (ROVir) coils, which can potentially achieve single breath-hold whole heart CINE. We demonstrated the feasibility of combining SMS and ROVir for highly accelerated CINE imaging (8-fold reduced scan time), enabling single breath-hold whole ventricular acquisition. Single breath-hold SMS+ROVir whole-heart CINE yielded cardiac function parameters with no significant bias when compared to SMS CINE.
Introduction
Conventional clinical cardiac MRI protocols use a large number (>20) of breath-holds for capturing cinemagraphic (CINE) scans of the heart in various views. Patients become fatigued from this kind of scan protocol, decreasing quality of breath-holds and consequently degrading image quality. Recent advances in accelerated techniques such as simultaneous multi-slice (SMS)1 and compressed sensing2 have been employed to drastically reduce CINE imaging scan time and the number of required breath-holds. One potential method capable of further accelerating CINE imaging is to drastically reduce the FOV to only include the heart, which could result in 4 to 5-fold acceleration3. However, naively reducing FOV will normally result in severe aliasing. In this work, we hypothesize that reduced-FOV SMS CINE can be achieved by using a novel beamforming approach, Region-Optimized VIRtual (ROVir) coils4, which can use the characteristics of a multi-channel receiver array to suppress signal from unwanted spatial regions. This technique can potentially achieve single breath-hold whole heart CINE. We test the proposed single breath-hold SMS+ROVir CINE against conventional multi- breath-hold CINE in normal volunteers with respect to contrast to noise ratio (CNR) and standard cardiac function parameters.Methods
Pulse Sequence Design and Image Reconstruction: SMS bSSFP CINE was achieved by implementing a gradient-controlled local Larmor adjustment (GC-LOLA 1) to restore the frequency response and stabilize banding artifacts across SMS acquired slices. GC-LOLA unbalances the slice gradient by a small constant and homogenizes RF phase cycles across all slices by adjusting the local Larmor frequency and aligning slice-specific frequency responses. This allows for predictable band placement across all slices and removes banding artifacts that might otherwise disrupt image quality (i.e. banding in blood pool). Prior to image reconstruction, virtual coils were obtained using ROVir, which linearly mixes the original receiver array channels in a way that optimally maximizes signal from the region of interest while also suppressing signal from unwanted spatial regions4 (Figure 1). SMS k-space data reconstruction from the ROVir coils was then performed with Split-Slice GRAPPA5.In vivo Study: Four healthy subjects were recruited with institutional IRB and scanned on a clinical 3T system (MAGNETOM Prisma, Siemens Healthcare, Erlangen, Germany). Full FOV (360mm x 360mm; matrix = 225 x 225) and 4-fold reduced phase encoding FOV (360mm x 90mm; matrix = 225 x 56) whole ventricular CINE were acquired with the prototype SMS (factor 2) using matching bSSFP CINE sequence parameters (TR = 3.1ms, TE = 1.8ms, alpha = 28º, SMS factor = 2, 1.6x1.6x10mm3, temporal resolution = 49.6ms, 8 slices, 25% gap). For full FOV measurements, 4 breath-holds were needed to cover the whole LV. A single cardiac phase from the full FOV measurement was used as calibration data for ROVir. For reduced FOV measurements, only a single breath-hold was needed. Noise scans were also performed by turning off the RF and collecting 200 samples of noise at each pixel with the same CINE sequence parameters above.
Image Analysis: CNR analysis was performed by first calculating the variance at each pixel from the noise scans. Afterwards, the absolute signal difference of the left ventricular myocardium and the blood were normalized by the noise estimates. Cardiac function parameters including end diastolic volume (EDV), end systolic volume (ESV), and ejection fraction (EF) were calculated with manual segmentation. Statistical comparisons were performed with Wilcoxon rank test to test for differences with a significance level of 0.05.
Results
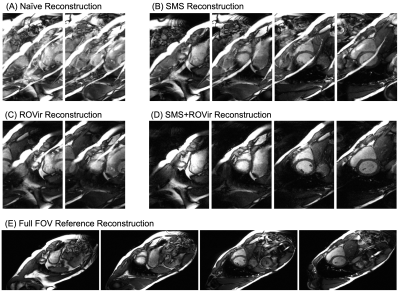
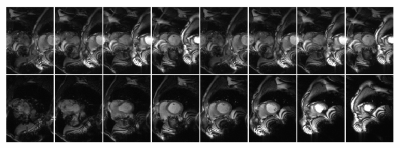
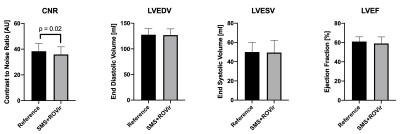
For reduced FOV CINE, reconstructing with either SMS only or ROVir only resulted in significant aliasing, severely degrading image quality when compared to the full FOV reference CINE (Figures 2 and 3). The proposed combination of SMS+ROVir substantially reduced aliasing (qualitatively) from both in-plane and through-plane sub-sampling. For all subjects, myocardial to blood CNR for single breath-hold SMS+ROVir CINE (35.8 ± 5.9) was significantly (p=0.02) decreased compared with reference full FOV CINE (38.4 ± 6.1). However, cardiac function parameters for single breath-hold SMS+ROVir CINE (EDV = 126.8 ± 12.2 ml; ESV = 49.5 ± 12.7 ml; EF = 61.3 ± 4.9 %) was not significantly different compared with full FOV SMS reference CINE (EDV = 127.5 ± 12.1 ml; ESV = 53.1 ± 9.7 ml; EF = 59.4 ± 6.8%) (Figure 4).Conclusion
We demonstrated the feasibility of combining SMS and ROVir reconstruction for highly accelerated CINE imaging (8-fold reduced scan time), thus enabling single breath-hold whole ventricular acquisition. Single breath-hold SMS+ROVir whole-heart CINE yielded cardiac function parameters with no significant bias when compared to SMS CINE. Future work will include combining SMS+ROVir with compressed sensing reconstructions for further acceleration and evaluating its potential utility in patients with a variety of cardiomyopathies.Acknowledgements
This work was partially funded by the National Institutes of Health awards R01HL135242 and R01HL151704.References
1. D. Stäb and P. Speier, “Gradient-controlled local Larmor adjustment (GC-LOLA) for simultaneous multislice bSSFP imaging with improved banding behavior,” Magn. Reson. Med., vol. 81, no. 1, pp. 129–139, 2019.
2. T. Kido et al., “Compressed sensing real-time cine cardiovascular magnetic resonance: Accurate assessment of left ventricular function in a single-breath-hold,” J. Cardiovasc. Magn. Reson., vol. 18, no. 1, pp. 1–11, 2016.
3. B. Madore, J. O. Fredrickson, M. T. Alley, and N. J. Pelc, “A reduced field-of-view method to increase temporal resolution or reduce scan time in cine MRI,” Magn. Reson. Med., vol. 43, no. 4, pp. 549–558, 2000.
4. "Region-Optimized Virtual (ROVir) Coils: Application of Sensor-Domain Beamforming for Localizing and/or Suppressing Spatial Regions.," ISMRM Proceedings 2021 (submitted).
5. F. Cauley, J. Polimeni, H. Bhat, L. Wald, K. Setsompop, "Interslice leakage artifact reduction technique for simultaneous multislice acquisitions.," Magn. Reson. Med. 72:93-102, 2014.
Figures
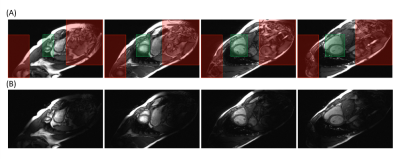
Figure 1. (A) Depictions of the regions of interest (green boxes) and the unwanted spatial regions (red boxes). (B) Coil-combined images from ROVir coils that optimally separate the region of interest from unwanted spatial regions corresponding to (A).