State-of-the-Art Echo-Planar BOLD Acquisition
1DZNE, Germany
Synopsis
This lecture reviews state-of-the-art 2D and 3D sequences with a focus on gradient echo EPI acceleration with controlled aliasing (CAIPIRINHA). The audience should learn which rapid EPI-based methods for BOLD fMRI are available and what to consider to minimize noise or artifacts due to strong parallel imaging, if needed, for the respective study goal.
BOLD and echo planar imaging
Due to its high sensitivity, the blood oxygen level-dependent contrast1 is the basis for most functional MRI (fMRI) studies. Although lacking spatial specificity (large draining veins), T2*-weighted echo planar imaging (EPI2) is most often used for fMRI. To maximize BOLD contrast-to-noise ratio, the echo time is usually selected in the order of 30-35/21-26ms (at 3T/7T, respectively). Even without parallel imaging, the whole brain can be imaged at a volume TR of 1-3s at typical spatial resolutions of 2-3mm isotropic. This is sufficient to sample the slow BOLD signal change, according to the average hemodynamic response function. Parallel imaging along the phase encode (PE) direction (R1>1) can be applied to increase the PE bandwidth by a factor of R1, and thus reduce susceptibility-induced artifacts (signal drop outs, geometric distortions, T2* blur, etc.). This is because the PE bandwidth is inversely proportional to the effective echo spacing (ESP), which is given by the actual ESP divided by the PE blip [measured in PE lines]. Large R1 can also be used to achieve higher spatial resolutions without increasing the EPI factor (the number of echoes in the EPI train = PE matrix size/PE blip). However, if spatial resolution is increased a lot, volume TRs can get very long.Simultaneous multi-slice (SMS) and 3D-EPI
Simultaneous multi-slice (SMS) acquisition3 can be employed to significantly speed up the imaging by acceleration. In slice-selective EPI, a stack of MB=R2>1 slices (MB: multiband factor) has to be excited simultaneously. The acquired signal is later disentangled using multiband parallel imaging principles4. Alternatively, 3D-EPI can be employed and reconstructed using conventional volumetric parallel imaging5,6. Apart from the excitation and second phase encoding, 3D-EPI and SMS-EPI are very similar sequence-wise. In terms of (parallel imaging) reconstruction, the equivalence between SMS- and 3D-EPI has been shown as well7-9.CAIPIRINHA, g-factor, temporal SNR and tSNR efficiency
Regardless of SMS- or 3D-EPI, care should be taken to condition the parallel imaging reconstruction as well as possible. Apart from optimal autocalibration acquisition (FLEET10, FLASH11, dual-polarity GRAPPA12, …), the undersampling pattern is very important. CAIPIRINHA sampling13,14,4 should be used to control the aliasing optimally and thus to avoid excessive noise increase. The latter is commonly expressed by the g-factor, g≥1, which depends on the number and the distribution of the receive-array coils and the (CAIPIRINHA) undersampling pattern. The g-factor can be found in the in the relation5SNR = SNR0/(g √R)
where R=R1R2 is the total parallel imaging undersampling factor and SNR0 is the (hypothetical) image SNR without parallel imaging and without physiological noise15. Note that, applying the respective Ernst angles, SNR0 is larger for 3D-EPI than for a corresponding 2D-EPI protocol16, and that SNR0 of SMS-EPI is somewhere in between17. On the other hand, more physiological noise adds to 3D-EPI than for SMS-EPI, when thermal noise does not dominate18,19 (e.g. large voxels). In practical terms, the combined effect of g, R and physiology – which is ultimately relevant for fMRI – can and should be compared in vivo by computing temporal SNR maps (voxel-wise mean/standard deviation along the time dimension) using the sequence and parameters under consideration. If one wants to compare between different temporal resolutions, it makes sense to convert tSNR maps into tSNR efficiency maps (tSNR per unit scan time), for instance: tSNR/√TRvol.
Blipped-CAIPI, shot-selective CAIPI and beyond
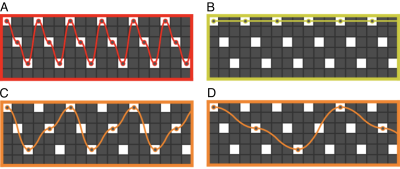
CAIPIRINHA sampling and EPI were first efficiently combined in SMS-EPI by adding alternating blips along the slice direction to the usual PE blips4. The same blipped-CAIPI sampling was applied later to 3D-EPI20. In SMS-k-space (or 2D PE k-space), the combined effect of blips is to follow a certain trajectory between samples of a selected 2D-CAIPIRINHA (short: CAIPI) pattern, according to14 expressed as R1xR2(D). D denotes the CAIPI shift along the second PE or slice direction (which corresponds to a certain PE FOV shift in SMS-EPI). Like without slice acceleration, the PE blip equals R1.An alternative shot-selective CAIPI EPI sampling was proposed for particularly high-resolution imaging, where large PE blips are required to keep the PE bandwidth high and EPI factor short21,22. Only those samples of the same CAIPI pattern are acquired that don’t require slice blips. One can also understand shot-selective CAIPI as interleaved multi-shot blipped-CAIPI with a segmentation factor S>1 (cf. Fig 1 A/B). The PE blip, and thus the PE bandwidth, is up to R2 times larger than with blipped-CAIPI, depending on the pattern. The shot-selective approach has recently been adopted in SMS-EPI23.
Note that the segmented blipped-CAIPI interpretation allows for further sampling variations of EPI (more possible PE blips with identical CAIPI pattern) (cf. Fig. 1 C/D)24,25.
Beyond parallel imaging
Since parallel imaging is limited by receive-coil design, it can make sense to consider alternative means to speed up imaging, and in turn consider reducing the total undersampling factor. A few suggestions:- Partial Fourier (PF) sampling is often applied along PE to maintain TE short despite a large EPI factor. If the latter is not a constrained, skipping late instead of early echoes may be a valuable option for shortening TRvol with reduced impact on the PSF (skipping only strongly decayed echoes).
- In 3D-EPI, partial Fourier can also be applied along the second PE direction16.
- Simultaneous PF along both PE directions should be used with care, as some of the skipped k-space information cannot be recovered.
- Generally, in T2*-weighted EPI, PF should be used with care, as the image phase at long TEs is far from homogeneous, and thus PF reconstruction is not as effective as in spin echo EPI (diffusion MRI).
- Usually, fat-saturation is used for fat suppression (~12ms per shot on Siemens systems). This prolongs TRvol considerably (~20%). At ultra-high fields this can often be omitted, as T2* of fat becomes very short26.
- With 3D-EPI, simple binomial water excitation (slab- or non-selective) can often be used and fat saturation can be omitted27.
- For 3D-EPI, semi-elliptical sampling has been proposed, where only late echoes outside an elliptical k-space mask are skipped with negligible consequences for the PSF28.
- It can make sense to acquire only one phase correction scan (per time series or per volume) external of imaging shots. Besides increased imaging efficiency, it also allows for shorter TE (or larger EPI factors).
DISCUSSION
Accelerated EPI is the state-of-the-art in BOLD imaging. Given the different options (some of which discussed above), it always makes sense to compare protocols in a study pilot. All the more as there is no definite “better” or “worse” in general. Before the piloting stage, one should consider, depending on the research question:- What spatial coverage is required? (whole-brain; reduced field-of-view or slab; single-slice)
- What temporal resolution is required? (may be different for resting-state or task fMRI; has consequences for temporal aliasing of physiological noise spectrum into the typical BOLD frequency range; check temporal correlation)
- What spatial resolution is required? (depends on structure of interest; check spatial specificity of BOLD)
- What the effect of physiological noise removal and temporal filtering is (motion, respiration, pulsation, derivatives, physiological noise model, highpass or bandpass filtering, …)
- Whether the region-of-interest or research question may benefit from multi-echo EPI (signal drop-outs; physiology; task or resting-state; …)
CONCLUSION
State-of-the-art echo-planar BOLD acquisition benefits largely from advanced parallel imaging techniques. Controlled aliasing (CAIPIRINHA) should always be applied for both SMS-EPI and 3D-EPI. This specifically includes a critical choice of the total undersampling factor (not too large) and an optimal aliasing pattern (minimal g-factor). At the study piloting stage, tSNR and tSNR efficiency should be criticially evaluated with and without physiological noise removal.Acknowledgements
No acknowledgement found.References
1. Ogawa S, Lee T ‐M, Nayak AS, Glynn P. Oxygenation‐sensitive contrast in magnetic resonance image of rodent brain at high magnetic fields. Magn. Reson. Med. 1990;14:68–78 doi: 10.1002/mrm.1910140108.
2. Mansfield P. Mutli-planar image formation using NMR spin echoes. J. Phys. C Solid State Phys. 1977;10:L55–L58 doi: citeulike-article-id:2605303.
3. Larkman DJ, Hajnal J V., Herlihy AH, Coutts GA, Young IR, Ehnholm G. Use of multicoil arrays for separation of signal from multiple slices simultaneously excited. J. Magn. Reson. Imaging 2001;13:313–317 doi: 10.1002/1522-2586(200102)13:2<313::AID-JMRI1045>3.0.CO;2-W.
4. Setsompop K, Gagoski BA, Polimeni JR, Witzel T, Wedeen VJ, Wald LL. Blipped-controlled aliasing in parallel imaging for simultaneous multislice echo planar imaging with reduced g-factor penalty. Magn. Reson. Med. 2012;67:1210–1224 doi: 10.1002/mrm.23097.
5. Pruessmann KP, Weiger M, Scheidegger MB, Boesiger P. SENSE: sensitivity encoding for fast MRI. Magn Reson Med 1999;42:952–62.
6. Griswold MA, Jakob PM, Heidemann RM, et al. Generalized autocalibrating partially parallel acquisitions (GRAPPA). Magn Reson Med 2002;47:1202–1210 doi: 10.1002/mrm.10171.
7. Zahneisen B, Poser BA, Ernst T, Stenger VA. Three-dimensional Fourier encoding of simultaneously excited slices: Generalized acquisition and reconstruction framework. Magn. Reson. Med. 2014;71:2071–2081 doi: 10.1002/mrm.24875.
8. Zahneisen B, Ernst T, Poser BA. SENSE and simultaneous multislice imaging. Magn. Reson. Med. 2015;74:1356–1362 doi: 10.1002/mrm.25519.
9. Poser BA, Setsompop K. Pulse sequences and parallel imaging for high spatiotemporal resolution MRI at ultra-high field. Neuroimage 2018;168:101–118 doi: 10.1016/j.neuroimage.2017.04.006.
10. Polimeni JR, Bhat H, Witzel T, et al. Reducing sensitivity losses due to respiration and motion in accelerated echo planar imaging by reordering the autocalibration data acquisition. Magn. Reson. Med. 2016;75:665–679 doi: 10.1002/mrm.25628.
11. Ivanov D, Barth M, Uludağ K, Poser BA. Robust ACS acquisition for 3D echo planar imaging. In: Proceedings of the International Society of Magnetic Resonance in Medicine. Vol. 23. ; 2015. p. 2059. doi: 10.1002/jmri.24551.
12. Hoge WS, Polimeni JR. Dual-polarity GRAPPA for simultaneous reconstruction and ghost correction of echo planar imaging data. Magn. Reson. Med. 2016;76:32–44 doi: 10.1002/mrm.25839.
13. Breuer F a, Blaimer M, Heidemann RM, Mueller MF, Griswold M a, Jakob PM. Controlled aliasing in parallel imaging results in higher acceleration (CAIPIRINHA) for multi-slice imaging. Magn. Reson. Med. 2005;53:684–91 doi: 10.1002/mrm.20401.
14. Breuer FA, Blaimer M, Mueller MF, et al. Controlled aliasing in volumetric parallel imaging (2D CAIPIRINHA). Magn. Reson. Med. 2006;55:549–556 doi: 10.1002/mrm.20787.
15. 1. Triantafyllou C, Hoge RD, Krueger G, et al. Comparison of physiological noise at 1.5 T, 3 T and 7 T and optimization of fMRI acquisition parameters. Neuroimage 2005;26:243–250 doi: 10.1016/j.neuroimage.2005.01.007.
16. Poser BA, Koopmans PJ, Witzel T, Wald LL, Barth M. Three dimensional echo-planar imaging at 7 Tesla. Neuroimage 2010;51:261–266 doi: 10.1016/j.neuroimage.2010.01.108.
17. Marques JP, Norris DG. How to choose the right MR sequence for your research question at 7 T and above? Neuroimage 2018;168:119–140 doi: 10.1016/j.neuroimage.2017.04.044.
18. van der Zwaag W, Marques JP, Kober T, Glover G, Gruetter R, Krueger G. Temporal SNR characteristics in segmented 3D-EPI at 7T. Magn. Reson. Med. 2012;67:344–352 doi: 10.1002/mrm.23007.
19. Blazejewska AI, Bhat H, Wald LL, Polimeni JR. Reduction of across-run variability of temporal SNR in accelerated EPI time-series data through FLEET-based robust autocalibration. Neuroimage 2017;152:348–359 doi: 10.1016/j.neuroimage.2017.02.029.
20. Narsude M, Gallichan D, Van Der Zwaag W, Gruetter R, Marques JP. Three-dimensional echo planar imaging with controlled aliasing: A sequence for high temporal resolution functional MRI. Magn. Reson. Med. 2016;75:2350–2361 doi: 10.1002/mrm.25835.
21. Poser BA, Ivanov D, Kannengiesser SA, Uludağ K, Barth M. Accelerated 3D EPI using 2D blipped-CAIPI for high temporal and/or spatial resolution. In: Proceedings of the International Society of Magnetic Resonance in Medicine. Vol. 22. ; 2014. p. 1506.
22. Hendriks AD, Agata FD, Raimondo L, et al. Pushing fMRI spatial and temporal resolution further: high density receive arrays combined with shot-selective 2D CAIPIRINHA for 3D EPI scans at 7T. In: Proceedings of the International Society of Magnetic Resonance in Medicine. ; 2019.
23. Eickel K, Porter DA, Söhner A, Maaß M, Lüdemann L, Günther M. Simultaneous multislice acquisition with multi-contrast segmented EPI for separation of signal contributions in dynamic contrast-enhanced imaging Degtyar VE, editor. PLoS One 2018;13:e0202673 doi: 10.1371/journal.pone.0202673.
24. Stirnberg R, Deistung A, Reichenbach J, Stöcker T. Accelerated quantitative susceptibility and R2* mapping with flexible k-t-segmented 3D-EPI. In: Proceedings of the International Society of Magnetic Resonance in Medicine. ; 2018.
25. Stirnberg R, and Stöcker T.; Unpublished Manuscript; 2020.
26. Speck O, Stadler J, Zaitsev M. High resolution single-shot EPI at 7T. Magn. Reson. Mater. Physics, Biol. Med. 2008;21:73–86 doi: 10.1007/s10334-007-0087-x.
27. Stirnberg R, Brenner D, Stöcker T, Shah NJ. Rapid fat suppression for three-dimensional echo planar imaging with minimized specific absorption rate. Magn. Reson. Med. 2016;76:1517–1523 doi: 10.1002/mrm.26063.
28. Stirnberg R, Huijbers W, Brenner D, Poser BA, Breteler M, Stöcker T. Rapid whole-brain resting-state fMRI at 3 Tesla: Efficiency-optimized three-dimensional EPI versus repetition time-matched simultaneous-multi-slice EPI. Neuroimage 2017;163:81–92 doi: 10.1016/j.neuroimage.2017.08.031.
29. Posse S, Wiese S, Gembris D, et al. Enhancement of BOLD-contrast sensitivity by single-shot multi-echo functional MR imaging. Magn. Reson. Med. 1999;42:87–97 doi: 10.1002/(SICI)1522-2594(199907)42:1<87::AID-MRM13>3.0.CO;2-O.
30. Poser B a, Norris DG. Investigating the benefits of multi-echo EPI for fMRI at 7 T. Neuroimage 2009;45:1162–72 doi: 10.1016/j.neuroimage.2009.01.007.
31. Kundu P, Inati SJ, Evans JW, Luh W-M, Bandettini P a. Differentiating BOLD and non-BOLD signals in fMRI time series using multi-echo EPI. Neuroimage 2012;60:1759–70 doi: 10.1016/j.neuroimage.2011.12.028.
Figures