4807
Deregulation of lipid metabolism in postmenopausal breast cancer patients using double quantum filtered correlation spectroscopy (DQF-COSY)1Aberdeen Biomedical Imaging Centre, University of Aberdeen, Aberdeen, United Kingdom, 2Breast Unit, Aberdeen Royal Infirmary, Aberdeen, United Kingdom, 3Clinical Radiology, Aberdeen Royal Infirmary, Aberdeen, United Kingdom, 4Radiology, Royal Marsden Hospital, London, United Kingdom
Synopsis
Deregulation of lipid metabolism has been shown in BRCA1/2 genetic mutation carriers. Mammary adipose tissues in postmenopausal women are the primary sites of oestrogen production linked to tumour initiation and progression. Therefore, lipid composition in postmenopausal breast plays a key role in breast cancer monitoring and subsequent development of prevention strategies. Previous studies focused on cell or animal models and invasive lipid extraction methods, while conventional MRS is inadequate in complete lipid composition measurement. We hypothesised that lipid composition in peri-tumoural breast adipose tissue is affected by the presence of tumour in postmenopausal women, using a non-invasive 2D MRS approach.
Introduction
Deregulation of lipid metabolism in breast has been shown in BRCA1/2 genetic mutation carriers1. Polyunsaturated fatty acid (PUFA) is depleted in tumour initiation, while free fatty acids (FA) are released from surrounding mammary adipocytes to support cancer growth2. Lipid composition is modulated by oestrogen linked to tumour initiation and progression3. While oestrogen is considered an endocrine hormone prior to menopause, the adipose tissue becomes the predominant source of oestrogens in postmenopausal women3. Therefore, lipid composition in postmenopausal women plays a key role in breast cancer initiation and subsequent development of prevention strategies4. Double quantum filtered correlation spectroscopy (DQF-COSY) provides accurate in vivo quantification of lipids5. We therefore hypothesised that lipid composition in the breasts of postmenopausal women is altered by the presence of adjacent tumour.Methods
Fifteen patients with invasive ductal carcinoma (age range, 53 – 71 years; mean: 63.1 years) and 15 age-matched healthy controls (age range, 54 – 76 years; mean: 65.1 years) participated in this study. Only patients undergoing wide local excision or mastectomy, with a tumour size larger than 1 cm on ultrasound were eligible. Patients with a previous history of cancer, having undergone neoadjuvant chemotherapy or hormonal therapy were not eligible. For healthy controls, women on hormone replacement therapy, having previous breast malignancy and family history of breast cancer were not eligible. The study was approved by the North of Scotland Research Ethics Committee (REC Ref: 16/NS/0077), and written informed consent was obtained from all the participants prior to the study.MRS Acquisition
All data were acquired on a 3.0 T whole body clinical MRI scanner (Achieva TX, Philips Healthcare, Best, Netherlands) using a body coil for uniform transmission and a 16-channel breast coil for high sensitivity detection. 2D MRS lipid spectra were acquired using DQF-COSY5 with TR of 552 ms, initial TE of 25 ms, a t1 increment of 1 ms, 256 increments, and voxel size of 20 × 20 × 20 mm3. In patients, a voxel (peri-tumoural) was positioned at 1 cm away from the tumour with a separate voxel (reference) in the contralateral healthy breast. In controls, a voxel was positioned in each breast. All voxels were positioned to contain primarily adipose tissue (Figure 1).
Data Processing
The spectral diagonal peaks of saturated FA (SFA) at (0.9,0.9) ppm, unsaturated FA (UFA) at (5.3,5.3) ppm, triglycerides (TRG) at (4.3,4.3) ppm, and off diagonal peaks of mono-UFA at (5.3,2.1) ppm and (2.1,5.3) ppm, PUFA at (5.3,2.8) ppm and (2.8,5.3) ppm were quantified in Felix software (v2007, Accelrys Inc., San Diego, USA)1. Peak ratios of degree of saturation (SFA/UFA), triacylglycerol pool (TRG/SFA), monounsaturation (MUFA/SFA) and polyunsaturation (PUFA/SFA) were subsequently computed for each breast. The disparities ([difference between the breasts] / [average of both breasts]) in the peak ratios were then calculated.
Histopathological Analysis
Standard routine histopathological examination was performed on excised breast tumours to determine tumour size, grade, lymph node metastasis and Nottingham Prognostic Index (NPI)6 in each patient.
Statistical Analysis
Statistical analysis was performed in SPSS software (Release 23.0, SPSS Inc., Chicago, IL, USA). Wilcoxon signed rank tests were used for non-normally distributed PUFA/SFA in patients and TRG/SFA in all participants. Paired-t and independent sample t-tests were performed on normally distributed SFA/UFA and MUFA/SFA. Pearson’s correlation tests were performed between disparities in degree of saturation and triacylglycerol pool against tumour size and NPI. A p value <0.05 was considered statistically significant.
Results
The characteristics of controls and patients are summarised in Table 1, and there were no significant differences between the two groups. There was a significantly higher disparity in degree of saturation (p = 0.0002) in patients (0.28 ± 0.10) compared to controls (0.13 ± 0.08) (Table 2, Figure 2a). There was a significantly higher disparity in triacylglycerol pool (p = 0.013) in patients (0.19 ± 0.15) compared to controls (0.08 ± 0.04) (Table 2, Figure 2b). There was no significant difference in disparity in degree of monounsaturation (p = 0.766) between controls and patients (Table 2, Figure 2c). There was no significant difference in disparity in degree of polyunsaturation (p = 0.294) between controls and patients (Table 2, Figure 2d).There were no significant correlations between disparities in the degree of saturation (p = 0.805) and triacylglycerol pool (p = 0.578) against tumour size (Figures 3a, 3b). There were no significant correlations between disparities in the degree of saturation (p = 0.500) and triacylglycerol pool (p = 0.150) against NPI (Figures 3c, 3d).
Discussion
We found that the differences in the degree of saturation and triacylglycerol pool between breast adipose tissue in patients were significantly higher than that in controls, indicating that the disparities in the degree of saturation and triacylglycerol pool could be two potential modifiable treatment targets critical for personalised medicine. Our work provided the first in vivo assessment of lipid deregulation in peri-tumoural adipose tissue, particularly triglycerides that cannot be resolved in 1D MRS, generating significant prognostic value.Conclusion
Our results showed that the degree of saturation and triacylglycerol pool in peri-tumoural breast adipose tissue are deranged as a result of the presence of tumour in postmenopausal women, offering potential treatment targets for breast cancer monitoring and prevention.Acknowledgements
The authors would like to thank Dr Matthew Clemence for clinical scientist support, Ms Angela Allan and Ms Vera Herd for nurse support, Ms Linda Lett, Ms Louisa Pirie, Ms Fiona Geddes, Ms Kate Shaw, Ms Sheila Ingram for patient recruitment support, Ms Kim Blake, Ms Shona Stuart, Ms Brenda Still, Ms Dawn Younie for logistic support, and Ms Beverly MacLennan, Ms Nichola Crouch, Ms Laura Reid and Mr Mike Hendry for radiographer support. The authors would also like to thank Ms Mairi Fuller, Mr Dionysios Koufoudakis, Ms Elizabeth Smyth and Ms Beatrice Elsberger for providing access to the patients. This project was funded by Friends of Aberdeen and North Centre for Haematology, Oncology and Radiotherapy (ANCHOR). Sai Man Cheung’s PhD study was jointly supported by Elphinstone scholarship, Roland Sutton Academic Trust and John Mallard scholarship and is currently funded by Cancer Research UK. Vasiliki Mallikourti’s PhD study is supported by Tenovus Scotland PhD studentship.
References
1. Ramadan S, Arm J, Silcock J, et al. Lipid and Metabolite Deregulation in the Breast Tissue of Women Carrying BRCA1 and BRCA2 Genetic Mutations. Radiology. 2015;275(3):675-682.
2. Wang YY, Attané C, Milhas D, et al. Mammary adipocytes stimulate breast cancer invasion through metabolic remodeling of tumor cells. JCI Insight. 2017;2(4):1-20.
3. Brown KA. Impact of Obesity on Mammary Gland Inflammation and Local Estrogen Production. J Mammary Gland Biol Neoplasia. 2014;19(2):183-189.
4. Freed M, Storey P, Lewin AA, et al. Evaluation of Breast Lipid Composition in Patients with Benign Tissue and Cancer by Using Multiple Gradient-Echo MR Imaging. Radiology. 2016;281(1):43-53.
5. Prescot AP, Dzik- Jurasz ASK, Leach MO, Sirohi B, Powles R, Collins DJ. Localized COSY and DQF-COSY 1H-MRS sequences for investigating human tibial bone marrow in vivo and initial application to patients with acute leukemia. J Magn Reson Imaging. 2005;22(4):541-548.
6. Elston CW, Ellis IO. Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology. 1991;19(5):403-410.
Figures
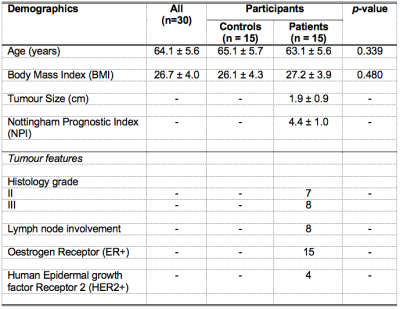
Table 1. Characteristics of study population.
Descriptive statistics of controls and breast cancer patients with histopathological findings are shown for each group and the entire cohort. Quantitative data were expressed as mean and standard deviation, while qualitative data expressed as number of positive cases.
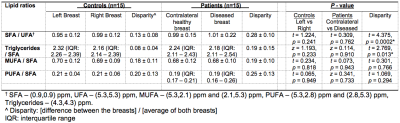
Table 2. Differences in lipid composition of breast adipose tissue between controls and patients after menopause.
Degree of saturation (saturated fatty acids (SFA) / unsaturated FA (UFA)), triacylglycerol pool (Triglycerides/SFA), degree of monounsaturation (MUFA/SFA) and degree of polyunsaturation (PUFA/SFA) measured in 2D double quantum filtered correlation spectroscopy (DQF-COSY) are shown. Significant findings are marked by ‘*’.
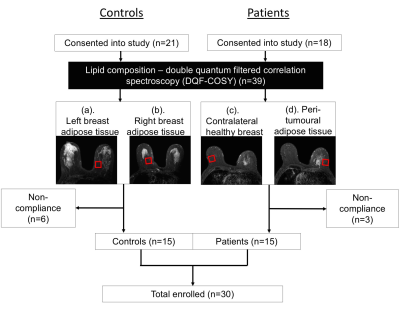
Figure 1. Study design.
A two-group cross sectional arrangement as shown in a flow chart. Twenty-one female healthy controls and 18 female breast cancer patients were eligible at initial screening and were consented into the study. All controls and patients were scanned on a clinical 3.0 T MRI scanner to assess the lipid composition in breasts (a, b: Controls; c, d: Patients) using double quantum filtered correlation spectroscopy (DQF-COSY). In total, 15 controls and 15 patients with invasive ductal carcinoma completed MR scans and participated in the study.
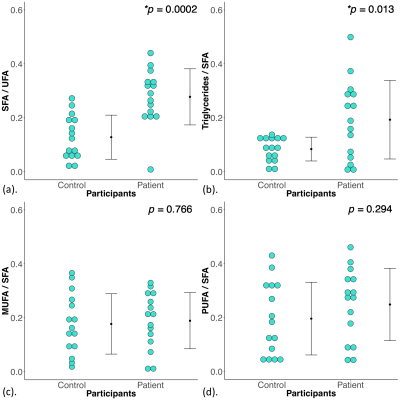
Figure 2. Disparity indices in the degree of saturation, triacylglycerol pool, degrees of monounsaturation and polyunsaturation.
The disparity ([difference between the breasts] / [average of both breasts]) in (a) degree of saturation (SFA/UFA), (b) triacylglycerol pool (Triglycerides/SFA), (c) degree of monounsaturation (MUFA/SFA) and (d) degree of polyunsaturation (PUFA/SFA) between controls and patients are shown in dot plots. The independent sample t-tests were performed between the groups and p value is shown. Statistically significant p values are marked by ‘*’.

Figure 3. Correlation results.
The disparities in the degree of saturation and triacylglycerol pool in patients were correlated with (a, b) tumour size and (c, d) Nottingham Prognostic Index (NPI). The corresponding Pearson’s correlation coefficients (r score) and p values are displayed. The disparities in the degree of saturation and triacylglycerol pool were not associated with tumour size and NPI from histology.