3326
The Investigation of Three-Dimensional MR elastography of the Brain in Multiple Sclerosis1Department of Radiology, the Third Affiliated Hospital, Sun Yat-sen University, Guangzhou, China, 2Department of Radiology, Mayo Clinic, Rochester, MN, United States, 3Department of Neurology, the Third Affiliated Hospital, Sun Yat-sen University, Guangzhou, China
Synopsis
Multiple sclerosis (MS) is a chronic inflammatory demyelinating disease of the central nervous system. MR Elastography (MRE) can quantitatively measure biomechanical tissue properties in vivo noninvasively. In this study, we investigated the potential value of MRE for the evaluation of centrum ovale viscoelasticity (including shear stiffness, storage modulus, shear loss modulus, and damping ratio) in Southern Chinese MS patients and to try to analyze its relevance to clinical manifestations. Our results showed that only the damping ratio of the centrum ovale was significantly decreased in MS patients and may provide a potential quantitative biomarker to evaluate MS.
Introduction
Multiple sclerosis (MS) is a chronic inflammatory disease of the central nervous system and the pathological process includes breakdown of the blood-brain barrier, multifocal inflammation, demyelination, oligodendrocyte loss, reactive gliosis, and axonal degeneration1. Magnetic resonance elastography (MRE), a noninvasive technique for quantifying tissue mechanical properties, may be sensitive to these pathological processes2-4. MRE characterization of animal models of MS revealed a clear correlation between viscoelastic tissue alteration and the magnitude of perivascular T cell infiltration as well as demyelination, and were reversible after remyelination2, 3. It has been reported that whole-brain viscoelasticity and stiffness decreased in subjects with MS in the West5, 6, but it is unclear if MRE can evaluate brain damage in the centrum ovale due to MS, one of the most affected sites in brain. The aim of this study is to assess the diagnostic performance of 3D MRE for the evaluation of MS in Southern China.Methods
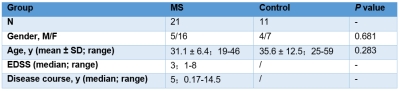
Following ethics committee approval with a waived informed consent requirement, 24 patients with MS and 16 healthy volunteers underwent conventional MRI and brain MRE scans. The MRE driver used was the pneumatic pillow driver developed by Mayo Clinic for brain MRE, and MRE scans with 60-Hz vibration frequency were performed. Three MS patients and five healthy controls were excluded due to unsuccessful MRE due to a loose or disconnected driver (n=8). Thus, the final study population comprised 21 patients and 11 controls. Clinical parameters including the expanded disability status scale (EDSS) and disease course (defined as years since diagnosis) were recorded. MRI scans including sagittal T2-weighted imaging (T2WI), axial T2WI, axial T2-weighted, fluid-attenuated inversion recovery (T2-FLAIR), T1-weighted imaging (T1WI) and diffusion-weighted imaging (DWI) were performed. The acquisition parameters for 3D MRE were as follows: TR/TE = 2000/62 ms, FOV = 24×24 cm; acquisition matrix = 96×96; number of excitations = 1; Bandwidth = 250 kHz; slices thickness = 3 mm with gap = 0 mm. The success of MRE was defined as visually detectable wave propagation in the whole brain. ROI-based stiffness, storage modulus and loss modulus measurements in the centrum ovale were obtained, and ROIs were drawn with reference to the MR images. The damping ratio (the loss modulus divided by two times the storage modulus) was also calculated. EDSS scores were obtained by two experienced neurologists for each patient in consensus. We compared the MRE parameters (including the stiffness, storage modulus, loss modulus and damping ratio) between the two groups using an unpaired t-test and related parameters were compared to the disease course and EDSS score with Pearson correlation. Statistical significance was defined as P<0.05.Results
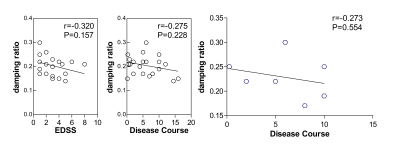
Demographic data and clinical characteristics are shown in Table 1 and the brain MRE images in MS are shown in Figure 1. In this study, only the mean damping ratio (0.21 ± 0.04 vs. 0.24 ± 0.04, P=0.036) of the centrum ovale was significantly decreased in MS patients compared with the control group, while there were no significant differences of the mean stiffness (2.29 ± 0.17 kPa vs. 2.35 ± 0.12 kPa, P=0.266), mean loss modulus (0.86 ± 0.20 kPa vs. 1.00 ± 0.17 kPa, P=0.058) or the mean storage modulus (2.09 ± 0.12 kPa vs. 2.09 ± 0.08 kPa, P=0.866) (Figure 2). There was a downward trend for the damping ratio with increasing EDSS and disease course (EDSS: r=-0.320, P=0.157; disease course: r=-0.275, P=0.228), but there were no statistical significances (Figure 3, left and middle). Damping ratio also had a significant correlation with disease course when EDSS score = 1 (r=-0.273, P=0.554) (Figure 3, right).Discussion
It has been shown that global stiffness and elasticity are reduced in MS5, 6. Our results showed significant decreased damping ratio of the centrum ovale in MS patients, while the stiffness, loss modulus and storage modulus did not decrease in this study. It has also been reported that damping ratio is associated with inflammation7. Maybe damping ratio in MS is a potential noninvasive biomarker to assess brain inflammatory damage due to MS. In addition, there was also a negative trend between damping ratio and EDSS and disease course, suggesting the potential of MRE to assess the clinical severity. However, due to the limited sample size, larger patient studies are needed to investigate this possibility.Conclusion
Damping ratio was decreased in the centrum ovale of MS patients and 3D MRE may be a potential method to evaluate brain tissue damage in MS.Acknowledgements
References
1. Trapp BD, Nave KA. Multiple sclerosis: an immune or neurodegenerative disorder? Annu Rev Neurosci 2008; 31:247-269.
2. Schregel K, Wuerfel E, Garteiser P, et al. Demyelination reduces brain parenchymal stiffness quantified in vivo by magnetic resonance elastography. Proc Natl Acad Sci U S A 2012; 109:6650-6655. 3. Riek K, Millward JM, Hamann I, et al. Magnetic resonance elastography reveals altered brain viscoelasticity in experimental autoimmune encephalomyelitis. Neuroimage Clin 2012; 1:81-90.
4. Fehlner A, Behrens JR, Streitberger KJ, et al. Higher-resolution MR elastography reveals early mechanical signatures of neuroinflammation in patients with clinically isolated syndrome. J Magn Reson Imaging 2016; 44:51-58.
5. Wuerfel J, Paul F, Beierbach B, et al. MR-elastography reveals degradation of tissue integrity in multiple sclerosis. Neuroimage 2010; 49:2520-2525.
6. Streitberger KJ, Sack I, Krefting D, et al. Brain viscoelasticity alteration in chronic-progressive multiple sclerosis. Plos One 2012; 7: e29888.
7. Yin M, Glaser KJ, Manduca A, et al. Distinguishing between Hepatic Inflammation and Fibrosis with MR Elastography. Radiology 2017; 284:694-705.
Figures