2163
Mechanical properties from CMR parameters in childhood acute lymphoblastic leukemia survivors.
Delphine Perie1, Egidie Uwase1, Tanguy Artz1, Marianna Gamba1, and Daniel Curnier2
1Mechanical Engineering, Polytechnique Montreal, Montreal, QC, Canada, 2Kinesiology, University of Montreal, Montreal, QC, Canada
1Mechanical Engineering, Polytechnique Montreal, Montreal, QC, Canada, 2Kinesiology, University of Montreal, Montreal, QC, Canada
Synopsis
Swine myocardium mechanical properties were found related to a combination of CMR relaxations times, magnetization transfer and diffusion parameters. Thus we assessed the relationships between mechanical properties and CMR parameters in childhood acute lymphoblastic leukemia survivors. The results showed that the pressure at diastasis, contractility and stiffness properties of the left ventricle can be partially predicted from the relaxation times T1 and T2 and the partition coefficient, reflecting cardiac health in childhood acute lymphoblastic leukemia. This indirect estimation of the mechanical behavior of the myocardium from multiparametric MRI could solve the challenging early cardiac sequelae detection.
Introduction
Doxorubicin-based chemotherapy entails dose-dependent cardiotoxicity [1] but both echocardiography and MRI investigations result in late detection, even when using strain quantification. Contrast enhanced T1 imaging is one of the most expected to early detect doxorubicin-induced cardiotoxicity [2,3]. Mechanical properties of the myocardium are considered as early biomarker of subtle changes in cardiomyopathy. Moreover, swine myocardium mechanical properties can be quantified from a linear combination of relaxations times, magnetization transfer and diffusion parameters [4]. The objective of this study was to investigate the relationships between mechanical properties computed from the CircAdapt model [5] and CMR parameters in childhood lymphoblastic leukemia survivors.Methods
Forty-seven acute lymphoblastic leukemia survivors (23±7 years old), treated with doxorubicin, were prospectively included in this study approved by our IRB. The CMR acquisitions were performed on a Siemens Skyra 3T MR system using a 18-channel phased array body matrix coil and included a MOLLI sequence for T1 mapping, a T2-prepared TrueFISP sequence for T2 mapping at apical, mid-ventricular and basal levels (pixel resolution 1.4mmx1.4mmx8.0mm), and an ECG-gated cine TruFISP sequence (14 slices in short axis and 5 slices in long axis, slice thickness 8mm, repetition time 34.6ms, effective echo time 1.2ms, flip angle 38°, iPAT factor 3, matrix 208x210 and in-plane pixel size 1.25x1.25 mm). The volunteers also underwent an exercise test using cardiac hemodynamic monitoring (PhysioFlow, Manatec Biomedical) to measure the arterial pressure, cardiac ouput and heart rate that were used as input data to the CircAdapt model [5]. The left ventricle stiffness (LVS) and contractility (LVC) were computed from the minimization of the differences between the left ventricle MRI volumes segmented using CIM (Figure 1, [6]) and the CircAdapt simulated volumes (Figure 2). Multiple linear regressions were investigated between the left ventricular pressure at diastasis (Pd), the left ventricular contractility or stiffness and the native relaxation times T1 and T2, the partition coefficient (PC), and the end-diastolic (EDV) and end-systolic (ESV) left ventricular volumes using SigmaStat (Systat Software, Inc.).Results
Low statistical power was obtained because CircAdapt optimizations succeeded only in 24 subjects over the 47 subjects (only mean differences between volumes less than 12mL were accepted). However, 46% of the left ventricular pressure at diastasis can be determined (r=0.68) from a linear combination of the CMR parameters (Equation 1) with a significant contribution of the partition coefficient (p=0.002). 23% of the left ventricular stiffness can be determined (r=0.47) from a linear combination of the CMR parameters (Equation 2) with a contribution of the partition coefficient (p=0.05). 36% of the left ventricular contractility can be determined (r=0.60) from a linear combination of the CMR parameters (Equation 3) with a significant contribution of the partition coefficient (p=0,02).Pd = -0,24 + (0.0001*T1) + (0.0007*T2) + (1.2*PC) Eq. 1
LVS = 0,01 + (0.0002*T1) - (0.007*T2) + (2.4*PC) Eq. 2
LVC = 4.3 - (0.001 T1) - (0.01*T2) - (2.9*PC) Eq. 3
Discussion
The partition coefficient of gadolinium is related to fibrosis [1], which should be associated to a stiffer behavior of the tissue due to more collagen fibers and less interstitial fluids. This is in agreement with the significant contribution of the partition coefficient in the determination of the left ventricular pressure at diastasis, the myocardium stiffness and the contractility. The variance inflation factors of T1, T2 and PC were always around 1 while preliminary analysis showed that the variance inflation factors of EDV and ESV were 10 times higher, suggesting that these parameters were likely candidates for elimination in the equation. However, based on our in-vitro study [4], T2 was expected to have a more significant contribution than the one we found. Magnetization transfer ratio and MRI diffusion parameters such as fraction of anisotropy and mean diffusivity should also be included to improve the relationship with the hyperelastic property of the myocardium.Conclusion
The pressure, contractility and stiffness properties in the left ventricle can be partially predicted from CMR parameters reflecting cardiac health in childhood acute lymphoblastic leukemia. This relationship can be improved with diffusion parameters, which in vivo acquisition remains challenging. This indirect estimation of the mechanical behavior of the myocardium from multiparametric MRI could solve the challenging early cardiac sequelae detection.Acknowledgements
NSERC and Polytechnique Montreal for the financial support, researchers from the PETALE study for the opportunity to do this complementary analysis on the cancer survivors.References
1- Aissiou M, Périé D, Cheriet F, Dahdah NS, Laverdière C, Curnier D. Imaging of early modification in cardiomyopathy: the doxorubicin-induced model. Int J Cardiovasc Imaging. 2013; 29(7):1459-76. 2- Perel RD, Slaughter RE, Strugnell WE. Subendocardial late gadolinium enhancement in two patients with anthracycline cardiotoxicity following treatment for Ewing's sarcoma. JCMR 2006;8(6):789-791. 3- Wassmuth R, Lentzsch S, Erdbruegger U, et al. Subclinical cardiotoxic effects of anthracyclines as assessed by magnetic resonance imaging-a pilot study. American heart journal 2001;141(6):1007-1013. 4- Périé D, Dahdah N, Foudis A, Curnier D. Multiparametric MRI as an indirect evaluation tool of the mechanical properties of in-vitro cardiac tissues. BMC Cardiovasc Disord. 2013 27; 13:24. 5- Arts T., et al., Adaptation to mechanical load determines shape and properties of heart and circulation: the CircAdapt model. Am J Physiol Heart Circ Physiol, 2005. 288(4):H1943-54. 6- Li B, Liu Y, Cowan BR, Young AA. (2010) In-line automated tracking for four dimensional ventricular function with magnetic resonance imaging. J Am Coll Cardiol Img 3, 860-866.Figures
Figure 1: Semi-automatic
segmentation from CIM (v8.1, University of Auckland, NZ, [6]). Six points are located by the operator at the apex
and base center on short axis images, at the right ventricular insertions on
short axis images and baseplane markers on long axis images. Manual correction
for mis-registration is then performed to obtain the endocardial and epicardial
contours in the short axis (right) and long axis (center) vues, as well as the
3D volumes (right).
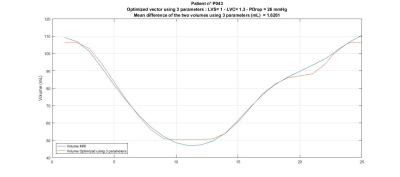
Figure 2: Volumes obtained from CIM
segmentation (orange line) and CircAdapt model (blue line) after left
ventricular stiffness and contractility optimization.