1345
Assessing the origin of human alpha oscillations using laminar layer 7T fMRI-EEG1Sir Peter Mansfield Imaging Centre, School of Physics and Astronomy, University of Nottingham, Nottingham, United Kingdom, 2Centre for Human Brain Health, School of Psychology, University of Birmingham, Birmingham, United Kingdom, 3Turner Institute for Brain and Mental Health, Monash University, Melbourne, Australia
Synopsis
EEG alpha (8-13Hz) oscillations occur throughout the cortex but the generating mechanisms are poorly understood. Opinion is divided between alpha being driven by bottom-up, top-down or both these processes. Here we use simultaneous 7T-fMRI-EEG during periods of eyes open/closed to assess the generator of alpha by determining the strongest BOLD-alpha negative layer correlations. We show the feasibility of using high spatial resolution 7T-fMRI with EEG to understand the origin of oscillations. Preliminary analysis shows BOLD-alpha correlations peak in middle layers of V1 (but not in V2/V3) providing suggestion that the alpha oscillations investigated are driven by bottom-up processing.
Introduction
EEG alpha (8-13Hz) oscillations occur throughout the cortex. Alpha power modulation is associated with performance of many cognitive tasks and opening/closing the eyes[1-5]. Changes in alpha power reflect a neural mechanism for gating of sensory input to the cortex[6-8]. The generating mechanisms of alpha are poorly understood, with opinion divided between whether alpha is primarily driven by: (i) bottom-up thalamic sources[9, 10]; (ii) top-down cortical sources[11-13]; or (iii) both processes[1, 7].Simultaneous EEG-fMRI using high spatial resolution fMRI at ultra-high field (7T) provides a unique method to study bottom-up and top-down mechanisms to provide new insight into the origin of alpha oscillations. Here we investigate EEG-fMRI correlations during periods of eyes open/closed to modulate resting-state activity.
Hypothesis
Increases in oscillatory alpha power during eyes closed are generated by bottom-up thalamocortical loops which primarily induce decreases in BOLD fMRI responses which are strongest in layer 4 of V1.Methods: Data Acquisition
EEG-fMRI data were acquired using MRplus EEG amplifiers and 64-channel EEG cap (Brain Products) in a 7T Philips Achieva MR scanner with MR-EEG clocks synchronised.fMRI data were collected using two high‐density 16‐channel receive arrays (MR Coils BV, Netherlands) positioned over the visual cortex and a 3D-EPI acquisition (88x168 matrix, 0.8mm isotropic resolution, TE/TR/TRvolume= 32/76/3800ms, SENSE=3.45/1.7 (P/S), 44 axial slices). Additional scans (same orientation with matched shim settings) included a B0field map (TR/TE=20/5.92ms, ΔTE=1ms, voxels=4mm3, 40 slices), partial head T1-weighted anatomical (PSIR, voxels=0.7mm3), and multi-TE 3D-EPI data (TE=30, 35, 40, 50ms). VCG and respiratory recordings aided EEG and MR physiological correction. Digitisation of electrode locations (Polhemus Fastrak) allowed EEG source localisation. In a separate session, using a 32-channel receive coil (Nova-Medical), a whole head PSIR (0.7mm3) and visual retinotopic mapping data[14](2D-EPI, 1.5mm3) were acquired.
Data were acquired on 10 healthy volunteers (4F, 28±5yrs) during a paradigm of alternating 30s blocks of eyes open/closed, with 4 blocks/run and 4-5 runs/subject.
Methods: Analysis
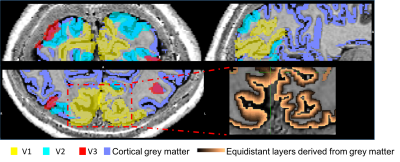
EEG: Data were gradient and pulse artefact corrected, filtered 0.1-40Hz and downsampled to 500Hz (BrainVision Analyzer2) then exported to FieldTrip[15]. The data were then demeaned per block. Noisy channels, blocks and eye blinks (ICA) were removed. Data were average-referenced then filtered into the alpha frequency (8-13Hz). Source localisation was performed using an LCMV beamformer. Pseudo-T-statistic (T-stat) maps were calculated showing task-related changes in oscillatory alpha-power between eyes open/closed. A virtual electrode (VE) time-course of alpha power (Hilbert envelop of the response) was derived from the site of maximum alpha power change in occipital cortex for each run during eyes open/closed.fMRI: 3D-EPI data were pre-processed as follows: physiological noise correction (RETROICOR[16]), distortion correction using the B0 field map, transforms for motion correction between and within runs were concatenated to the central image of the entire session and applied to the data with spline interpolation (MCFLIRT&FLIRT, FSL). The EEG alpha power VE-time-course was convolved with a HRF (double gamma, time-to-peak=6s) and downsampled to the fMRI TRvolume and along with motion realignment parameters was input as regressors into an individual subject fixed effects GLM (FEAT, FSL). PSIR data acquired in the same session were used to manually define a grey matter mask which was combined with V1, V2 and V3 regions of interest (ROIs) defined from retinotopic mapping data analysed in mrTools[17]. The grey matter mask was used to define 12 layers across the cortex using the equidistance algorithm implemented in LAYNII[18]. Z-statistics corresponding to the negative BOLD-alpha correlation were found across layers in V1-V3 and averaged within layers for different thresholds.
Results
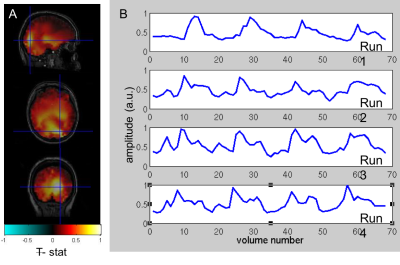
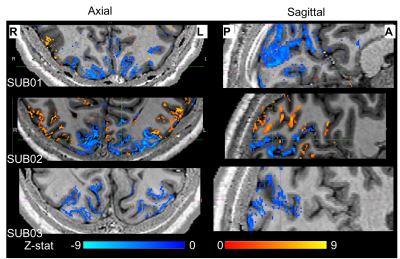
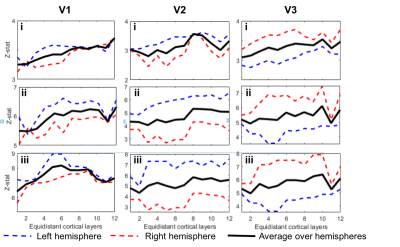
To date, analysis is completed in 3 subjects. Figure 1 shows clear localisation of alpha power to the visual cortex and modulation of alpha power with eyes open/closed. Figure 2 shows an example of the definition of the ROIs and layers on the PSIR image. Figure 3 illustrates excellent spatial agreement of the BOLD modulations across all runs within grey matter, and the expected negative correlation between BOLD and alpha power[19]. Figure 4 shows the variation in negative alpha-BOLD correlation across layers for V1, V2 and V3. A peak within the mid-layers is revealed in V1 when considering the top 5% or maximum Z-stat within each layer. Whilst in V2 and in V3 a relatively flat profile across layers is seen regardless of threshold.Discussion and Conclusion
Here we have shown the feasibility of using high spatial resolution 7T fMRI in combination with EEG to further our understanding of the origin of neural oscillations. We show with careful experimental setup it is possible to simultaneously acquire high quality oscillatory alpha (Fig 1) and BOLD responses. These responses are well correlated and highly spatially specific to the grey matter (Fig. 3). The variation of activation profile across layers indicates a peak in the middle layers of V1 (Fig 4A), although pial vein effects remain which will be assessed in further analysis using the T2*-maps[20]. A flat profile in V2&V3 is seen, where bottom-up inputs project to all layers[21]. These preliminary findings agree with our hypothesis and suggest that in resting state, alpha is generated by bottom-up processes, in contrast to [13] who showed, using 3T MRI, alpha was driven by top-down processes during visual spatial attention.Acknowledgements
This work was funded by the Leverhulme Trust [grant number RPG-2014-369], and the Engineering and Physical Sciences Research Council (EPSRC) and Medical Research Council (MRC) [grant number EP/L016052/1].
References
1. Hanslmayr, S., et al., The role of alpha oscillations in temporal attention. Brain Res Rev, 2011. 67(1-2): p. 331-43.
2. Jensen, O., et al., Oscillations in the alpha band (9-12 Hz) increase with memory load during retention in a short-term memory task. Cereb Cortex, 2002. 12(8): p. 877-82.
3. Klimesch, W., et al., 'Paradoxical' alpha synchronization in a memory task. Brain Res Cogn Brain Res, 1999. 7(4): p. 493-501.
4. Romei, V., J. Gross, and G. Thut, On the role of prestimulus alpha rhythms over occipito-parietal areas in visual input regulation: correlation or causation? J Neurosci, 2010. 30(25): p. 8692-7.
5. Berger, H., Uber das elektroenkephalogram des menschen. Arch Psychiatr Nevenkr, 1929. 87: p. 527-570.
6. Jensen, O. and A. Mazaheri, Shaping functional architecture by oscillatory alpha activity: gating by inhibition.Front Hum Neurosci, 2010.4: p. 186.
7. Klimesch, W., P. Sauseng, and S. Hanslmayr, EEG alpha oscillations: the inhibition-timing hypothesis.Brain Res Rev, 2007. 53(1): p. 63-88.
8. Rihs, T.A., C.M. Michel, and G. Thut, Mechanisms of selective inhibition in visual spatial attention are indexed by alpha-band EEG synchronisation. Eur J Neurosci, 2007. 25(2): p. 603-10.
9. Hughes, S.W., et al., Synchronized oscillations at alpha and theta frequencies in the lateral geniculate nucleus. Neuron, 2004. 42(2): p. 253-68.
10. Lopes da Silva, F., Neural mechanisms underlying brain waves: from neural membranes to networks. Electroencephalogr Clin Neurophysiol, 1991. 79(2): p. 81-93.
11. Buffalo, E.A., et al., Laminar differences in gamma and alpha coherence in the ventral stream. Proc Natl Acad Sci U S A, 2011. 108(27): p. 11262-7.
12. von Stein, A., C. Chiang, and P. Konig, Top-down processing mediated by interareal synchronisation. Proc Natl Acad Sci U S A, 2000. 97(26): p. 14748-53.
13. Scheeringa, R., et al., The relationship between oscillatory EEG activity and the laminar-specific BOLD signal. Proceedings of the National Academy of Sciences of the United States of America, 2016. 113(24): p. 6761-6766.
14. Wandell, B.A., S.O. Dumoulin, and A.A. Brewer, Visual field maps in human cortex. Neuron, 2007. 56(2): p. 366-383.
15. Oostenveld, R., et al., FieldTrip: Open source software for advanced analysis of MEG, EEG, and invasive electrophysiological data. Comput Intell Neurosci, 2011. 2011: p. 156869.
16. Glover, G.H., T.Q. Li, and D. Ress, Image-based method for retrospective correction of physiological motion effects in fMRI: RETROICOR. Magn Reson Med, 2000. 44(1): p. 162-7.
17. Gardner, J. L., et al, mrTools: Analysis and visualization package for functional magnetic resonance imaging data. (Zenodo, 2018). 10.5281/zenodo.1299483.
18. Huber, L., layerfMRI/LAYNII: LAYNII (v1.0.0) [Computer Software]. Retrieved from:https://layerfmri.com/category/code/
19. Goldman, R.I., et al., Simultaneous EEG and fMRI of the alpha rhythm.Neuroreport, 2002. 13(18): p. 2487-92.
20. Shmuel, A., et al., Spatio-temporal point-spread function of fMRI signal in human gray matter at 7 Tesla.Neuroimage, 2007. 35(2): p. 539-552.
21. Felleman, D.J. and D.C. Van Essen, Distributed hierarchical processing in the primate cerebral cortex.Cereb Cortex, 1991. 1(1): p. 1-47.
Figures