0701
In Vivo Spectroscopic Detection of Glutaminase Enzyme Activity with Hyperpolarized [5-13C,4-2H2,5-15N]-L-Glutamine1Radiology, Memorial Sloan Kettering Cancer Center, New York, NY, United States, 2Cancer Biology & Genetics, Memorial Sloan Kettering Cancer Center, New York, NY, United States
Synopsis
Aberrations in glutaminase enzyme expression are associated with a variety of pathologies, and an in vivo probe to quantify flux through this pathway may provide a new layer of information. We developed a custom-synthesized compound, [5-13C,4-2H2,5-15N]-L-Glutamine, as a hyperpolarized MRI probe for glutaminase activity. Triple labeling of glutamine and D2O solvation reduces quadrupolar relaxation and extends both T1 and T2, facilitating in vivo imaging. We were able to acquire 13C spectroscopic data on a subcutaneous RCC xenograft murine model and detect in vivo conversion of hyperpolarized glutamine to glutamate, which permits further exploration of this imaging probe in the future.
Introduction
Glutamine is highly consumed by fast- growing cells, and its main cellular role is providing carbon and nitrogen through various metabolic pathways. Glutaminase utilizes glutamine as a substrate and produces glutamate and ammonia. Quantification of glutamine metabolism appears to be crucial since it is involved in multiple diseases including cancer and diabetes.1 Due to the global relevance of glutaminase in a variety of diseases, we seek to develop an in vivo metabolic probe. In this study, a xenograft murine model of renal cell carcinoma as a glutamine-avid tumor has been used. The goal is to develop a probe for real-time flux in glutamine metabolism.Hyperpolarized magnetic resonance provides high transient spin-lattice signal. Fast acquisition sequences in magnetic resonance enable us to obtain dynamics fast biochemical processes. Prior studies using commercial [5-13C]-L-glutamine confirmed that detecting the carbon-5 resonance of [5-13C]-L-glutamate with 13C nuclear magnetic resonance (NMR) could be used to monitor dynamics of the reaction in rat model(Fig.1). However, directly-bonded quadrupolar 14N-nuclei increase signal decay outside the polarizer due to quadrupolar effect at low magnetic field.2 We also learned that T1 can be increased with deuteration of adjacent carbons.3 Here, we provide a modular synthetic strategy for a series of 13C-, 2H(D)-, 15N-isotope-labeled glutamine in high yield. Enriching with 15N decreases low field quadrupolar relaxation. The presence of deuterium next to side chain carbonyl increases T1 by eliminating dipole-dipole relaxation. In addition, we discovered the presence of 15N increases T2.4 Following up our on previous studies, we used D2O phosphate buffer for dissolution resulting in the exchange of protons with deuterium, which increased T1 and T2 relaxations. We have characterized the physical chemical properties of these compounds and have showed triple labeling [5-13C,4-2H2,5-15N]-L-Glutamine and dissolving in D2O improved SNR and enabled detection of in vivo glutaminase activity and performed Magnetic resonance spectroscopy with two-dimensional resolution.5
Methods
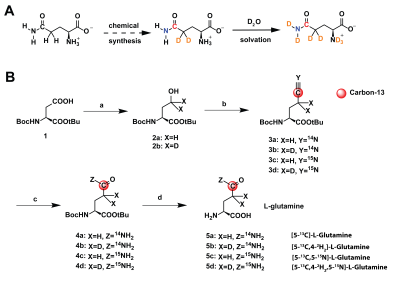
Four labeling patterns of glutamine were synthesized using a four-step synthesis (Fig.2B) adapted from previous work on arginine.4 For hyperpolarized experiments, glutamine was polarized in a SPINLab (GE Healthcare) for >2h (5.0T, 0.8K, 139.960GHz). Inversion-recovery and Carr-Purcell-Meiboom-Gill acquisitions for T1 and T2 measurements of the carbon-5 resonance, centered at 178ppm, were performed on a Bruker 14.1T NMR. UOK262 cells (gift from Dr. W.M. Linehan at NIH) were cultured in high-glucose DMEM. 5 million cells were xenografted in female athymic nude mouse and imaged when the tumors were approximately 200-300mm3. Hyperpolarized T1 were measured at low field on a 1T 13C-NMR. For in vivo 13C spectroscopy, xenografted mouse was infused with 30mM [5-13C,4-2H2,5-15N]-L-Glutamine via tail vein catheter and imaged on a Bruker 3T MRI following an IACUC-approved protocol. A dual-tuned 1H/13C coil was used to acquire anatomic reference images and spectroscopic data. T2-weighted fast-spin-echo (FSE) images were acquired for anatomic localization, covering the whole tumor (40x40x24-mm FOV, 128x128x12 matrix, TE/TR=69/1800ms). A chemical-shift imaging sequence (2DCSI) was used to acquire HP spectra (40x40x10-mm FOV, 12x12x1 matrix, 5kHz sweep width, 0.1ms dwell time, 320 spectral points, TE/TR 5/70ms, 20° excitation), 25s post-injection derived from the previous dynamic studies.Results
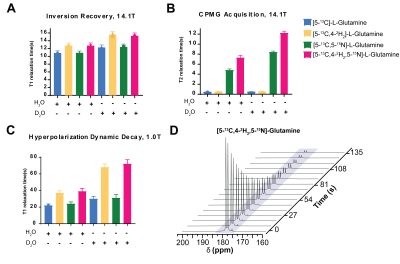
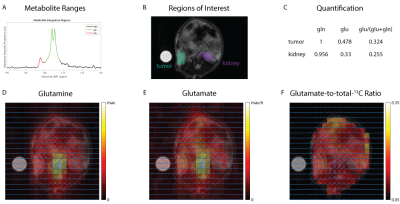
At 14.1T, the T1 of the glutamine carbon-5 resonance increased with 2H-enrichment: [5-13C]-L-Glutamine T1=10.8±0.55s and [5-13C,4-2H2]-L-Glutamine T1=12.74±0.69s. 15N-enrichment provided minimal increase: [5-13C,5-15N]-L-Glutamine T1=10.9±0.5s. Triple-labeling resulted in the largest increase: [5-13C,4-2H2,5-15N]-L-Glutamine T1=12.74±0.69s) (Fig.2B). Dissolution in D2O increased T1 in all species, e.g., [5-13C,4-2H2,5-15N]-L-Glutamine T1=15.58±0.7s. Hyperpolarization decay at 1T showed similar trends: [5-13C,4-2H2,5-15N]-L-Glutamine relaxation in phosphate buffer in D2O decreased ~3.3-fold resulting in a T1=72±5s. (Fig.3C) Interestingly, by using slightly acidic phosphate buffer (pH= 3.1), accounting for the NaOH in the prep formulation, no side production of pyroglutamate or glutamate were detected in the final dissolution as highlighted in the hyperpolarized 13C dynamic decay (Fig.3D). At 14.1T, the T2 of the carbon-5 resonance increased with 15N-enrichment: [5-13C]-L-Glutamine T2=0.474±0.029s and [5-13C, 5-15N]-L-Glutamine T2=4.82±0.23s. 2H-enrichment provided a minimal increase: [5-13C,4-2H2]-L-Glutamine T2=0.521±0.048s. Triple labeling provided the largest increase: [5-13C,4-2H2,5-15N]-L-Glutamine T2=7.28±0.48s. Dissolution in D2O increased the T2 in all species, e.g., [5-13C,4-2H2,5-15N]-L-Glutamine T2=12.23±0.36s, ~26-fold enhancement (Fig.3B). Axial 2D CSI 13C HP MRS in a mouse injected with hyperpolarized [5-13C,4-2H2,5-15N]-L-Glutamine revealed in vivo conversion to hyperpolarized glutamate (Fig.4D,E). The ratio of conversion in tumor voxels (0.324) was higher than in kidney voxels (0.255) (Fig 4C,F).Discussion
2H-enricment has a substantial impact on extending glutamine carbon-5 resonance T1 at high or low field. 15N-enrichment has an insignificant effect on thermal T1, but reduces quadrupolar relaxation and alleviates rapid depolarization following dissolution when the sample is removed from a strong magnetic field. 15N-enrichment increased T2 at all field strengths, thereby improving imaging quality. Conversion of multi-labeled hyperpolarized glutamine can be used for glutaminase activity in vivo. 13C spectroscopic data in a xenograft model with hyperpolarized glutamine demonstrated detection of higher metabolic flux in the tumor as compared to neighboring normal kidney tissue, suggesting delivery and metabolic conversion occur rapidly and further hyperpolarized studies should be pursued.Conclusions
The quantification of glutamine flux through conversion of hyperpolarized [5-13C,4-2H2,5-15N]-L-Glutamine to [5-13C,4-2H2]-L-glutamate was detected in vivo in a xenograft model, and it is a promising first step towards using this probe to study glutaminolysis in more disease models. Moving forward, we aim to further optimize our acquisition scheme for higher resolution spectroscopy and rapid imagingAcknowledgements
This work was supported by NIH/NCI Cancer Center Support Grant P30 CA008748, NIH S10 OD016422 as well as Tow Foundation, Thompson Family Foundation.References
1. Pavlova, N.N, et al. 2016. The Emerging Hallmarks of Cancer Metabolism. Cell Metab, 23. p.27-47.
2. Cabella, C, et al, 2013. In vivo and in vitro liver cancer metabolism observed with hyperpolarized [5-13C]glutamine. J Magn Reson, 232. p.45-52
3. Qu, W., et al. 2011. Facile synthesis [5-13C-4-2H2]-L-glutamine for hyperpolarized MRS imaging of cancer cell metabolism. Acad Radiol,18,9. p932-9.
4. Cho, A. et al. 2019. Hyperpolarized [6-13C,15N3]-Arginine as a Probe for in Vivo Arginase Activity. ACS Chem Biol, 14,4. p.665-673
5. Cho, A. et al. 2018. A non-synthetic approach to extending the lifetime of hyperpolarized molecules using D2O solvation. J Magn Reson, 295.p.57-62.
Figures