0049
Quantitative Myelin Mapping in Human Brain Using a Short TR Adiabatic Inversion Recovery Prepared Ultrashort Echo Time (STAIR-UTE) Sequence1UC San Diego, San Diego, CA, United States, 2VA health system, San Diego, CA, United States
Synopsis
To quantitatively image myelin on clinical scanners, we propose a Short TR Adiabatic Inversion Recovery prepared UTE (STAIR-UTE) sequence for in vivo brain imaging. With STAIR technique, long T2 tissues with a broad range of T1s can be sufficiently suppressed. Healthy volunteer has a higher myelin proton density in white matter than that in patient with multiple sclerosis.
Introduction
UTE sequences with echo times less than 50 µs can directly detect signals from myelin protons (1-4). However, over 90% of the UTE signal originates from long T2 water protons, even in myelin-rich white matter. Sufficient long T2 suppression is crucial for accurate quantitative myelin imaging. Adiabatic inversion recovery prepared UTE (IR-UTE) sequences have been proposed for imaging myelin (4-6). However, long T2 suppression is sensitive to the selection of TI in currently available IR-UTE techniques (3-7). A slight TI offset may cause significant long T2 contamination, leading to inaccurate short T2 myelin quantification. It is well-recognized that T1 relaxation may change with both age and pathology in the brain (8,9). This makes the corresponding TI determination more challenging. To address the aforementioned problems in quantitative myelin imaging, we propose a Short TR Adiabatic Inversion Recovery prepared UTE (STAIR-UTE) technique for more robust long T2 suppression regardless of T1 variations in white matter and which, therefore, provides more accurate quantitative myelin imaging.Methods
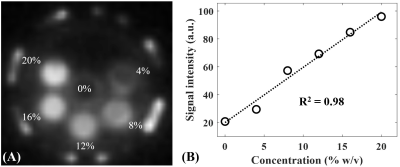
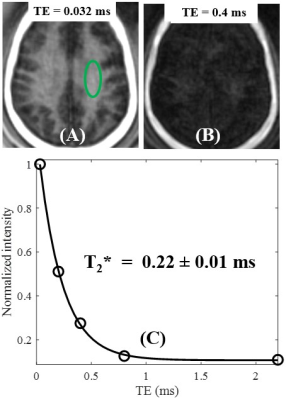
Figure 1A shows features of the 3D STAIR-UTE sequence (10,11). Following an adiabatic IR pulse, Nsp separate k-space spokes with an identical time interval, τ, are used for fast data acquisition. Figure 1B shows the contrast mechanism of myelin imaging. The myelin magnetization recovers from zero, and pure myelin signals can be acquired at a specific TI when the long T2 components are nulled (12,13). Numerical simulation was performed to investigate the efficiency of long T2 suppression for the STAIR scheme with different TRs. The simulated T1 values of the long T2 components ranged from 20 to 2500 ms, which cover T1 values for both white matter and gray matter. The TIs were determined by Eq. [8] in Ma et al. (11) in order to minimize the average signals from the long T2 components with T1 values ranging from 500 to 1500 ms.A myelin-D2O phantom was made by compounding myelin lipid powder with D2O in 1-mL syringes. A total of six myelin-D2O tubes with myelin concentrations of 0, 4, 8, 12, 16, and 20% weight/volume (w/v) were prepared. The myelin phantoms were simultaneously imaged in a homemade T/R birdcage coil using the STAIR-UTE sequence with the following parameters: FOV=10×10×6.4cm3, matrix=128×128×16, TR/TI=150/71ms, TE=32μs, Nsp=3, τ=5ms, FA=20°, bandwidth=62.kHz. T2* was also measured using the conventional 3D UTE sequence with TEs of 0.032, 0.2, 0.4, 0.8, and 2.2 ms.
In vivo brain imaging was performed on ten healthy volunteers (19-45 years of age) and ten patients with multiple sclerosis (MS) (35-70 years of age). Informed consent was obtained from all subjects in accordance with guidelines of the institutional review board. The sequence parameters are as follows: FOV=22×22×30 cm3, matrix=140×140×60, TR/TI=140/61ms, TE=3μs, Nsp=5, τ=3.2ms, FA=2°, bandwidth=125kHz, scan time=9.5 min. A rubber band with a short T2 (i.e., T2 around 0.3ms) was placed between each subject’s head and the coil to serve as a reference to calibrate the proton density of myelin using Eq. [9] in Ma et al. (11). Clinical T2-FlAIR sequence was used for comparison.
The signal intensities in both the normal-appearing white matter (NAWM) in MS patients and normal white matter (NWM) in volunteers were measured for comparison. ANOVA analysis was performed to evaluate the signal difference between lesion-containing and normal white matter.
Results and Discussion
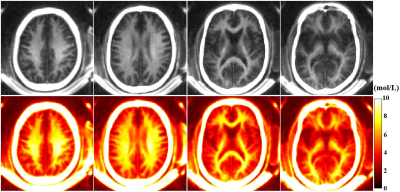
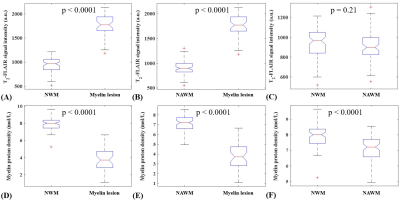
The mean T2* value of the six myelin-D2O phantoms is 0.25±0.03ms. Figure 2 shows an excellent correlation (R2=0.98) between STAIR-UTE signal intensities and myelin concentrations for these six phantoms. The STAIR-UTE image from a healthy brain in Figure 3 with TE=32 μs shows a high signal intensity in the white matter region. Exponential fitting of the STAIR-UTE signals with different TEs demonstrates a short T2* of 0.22±0.01 ms. The measured short T2* value is very close to that of myelin-D2O phantoms (T2*=0.25±0.03 ms), demonstrating that STAIR technique allows sufficiently nulling of long T2 components in white matter.Figure 4 shows morphological images and corresponding proton density maps from a healthy volunteer. Both T2-FLAIR and STAIR-UTE measures show significant differences in MS lesions between NWM of healthy volunteers and NAWM of MS patients (p<0.0001) (A, B, D, and E in Figure 5). This demonstrates the feasibility of the proposed STAIR-UTE in quantitatively detecting MS lesions. In addition, the STAIR-UTE measures show significant difference between NWM of healthy volunteers and NAWM of MS patients (p<0.0001, see Figure 5F). In contrast, there is no significant difference found in T2-FLAIR images between these two groups (p=0.21, see Figure 5C). Those results suggest that the proton density quantified by the STAIR-UTE technique may be a useful biomarker in differentiating the myelin content in MS lesions, NAWM, and NWM.
Conclusion
The 3D STAIR-UTE sequence with a short TR/TI combination provides robust suppression of long T2 components in white matter, allowing accurate volumetric mapping of myelin proton density in brain.Acknowledgements
The authors acknowledge grant support from NIH (1R01NS092650) and VA Clinical Science and Rehabilitation R&D Awards (I01CX001388 and I01RX002604).References
1. Waldman A, Rees JH, Brock CS, Robson MD, Gatehouse PD, Bydder GM. MRI of the brain with ultra-short echo-time pulse sequences. Neuroradiology 2003; 45:887–892.
2. Horch AR, Gore JC, Does MD. Origins of the ultrashort-T2 1H NMR signals in myelinated nerve: a direct measure of myelin content? Magn Reson Med 2011; 66:24–31.
3. Wilhelm MJ, Ong HH, Wehrli SL, Li C, Tsai PH, Hackney DB, Wehrli FW. Direct magnetic resonance detection of myelin and prospects for quantitative imaging of myelin density. Proc Natl Acad Sci USA 2012; 109:9605–9610.
4. Du J, Ma G, Li S, Carl M, Szeverenyi NM, VandenBerg S, Corey-Bloom J, Bydder GM. Ultrashort echo time (UTE) magnetic resonance imaging of the short T2 components in white matter of the brain using a clinical 3T scanner. Neuroimage 2014;15;87:32-41.
5. Sheth V, Shao H , Chen J, Vandenberg S, Corey-Bloom J. Bydder GM, Du J. Magnetic resonance imaging of myelin using ultrashort Echo time (UTE) pulse sequences: phantom, specimen, volunteer and multiple sclerosis patient studies. Neuroimage 2016; 136, 37–44.
6. Seifert AC, Li C, Wilhelm MJ, Wehrli SL, Wehrli FW. Towards quantification of myelin by solid-state MRI of the lipid matrix protons. NeuroImage 2017;163:358-67.
7. Fan S-J, Ma Y, Zhu Y, et al. Yet more evidence that myelin protons can be directly imaged with UTE sequences on a clinical 3T scanner: Bicomponent T2* analysis of native and deuterated ovine brain specimens. Magn. Reson. Med. 2018;80:538–547
8. Suzuki S, Sakai O, Jara H. Combined volumetric T1, T2 and secular-T2 quantitative MRI of the brain: age-related global changes (preliminary results). Magn. Reson. Imaging 2006;24:877–887.
9. Margaret Cheng H-L, Stikov N, Ghugre NR, Wright GA. Practical medical applications of quantitative MR relaxometry. J. Magn. Reson. Imaging 2012;36:805–824.
10. Carl M, Bydder GM, Du J. UTE imaging with simultaneous water and fat signal suppression using a time-efficient multispoke inversion recovery pulse sequence. Magn. Reson. Med. 2016;76:577–582.
11. Ma YJ, Chen Y, Li L, Cai Z, Wei Z, Jerban S, Jang H, Chang EY, Du J. Trabecular bone imaging using a 3D adiabatic inversion recovery prepared ultrashort TE Cones sequence at 3T. Magn. Reson. Med. 2019; doi: 10.1002/mrm.28027.
12. Horch RA, Gochberg DF, Nyman JS, Does MD. Clinically compatible MRI strategies for discriminating bound and pore water in cortical bone. Magn. Reson. Med. 2012;68:1774–1784.
13. Du J, Bydder GM. Qualitative and quantitative ultrashort-TE MRI of cortical bone. NMR Biomed. 2013;26:489–506.
Figures