4266
18F-Fluoroethyltyrosine PET and Multiple Quantum Filtered Sodium MRI in Cerebral Gliomas1Institute of Neuroscience and Medicine - 4, Forschungszentrum Jülich GmbH, Jülich, Germany, 2Institute of Neuroscience and Medicine - 11, Forschungszentrum Jülich GmbH, Jülich, Germany, 3Department of Nuclear Medicine, RWTH Aachen University, Aachen, Germany, 4Jülich-Aachen Research Alliance (JARA) - Section JARA-Brain, Aachen, Germany, 5Department of Neurology, RWTH Aachen University, Aachen, Germany
Synopsis
[18F]-FET-PET and sodium MRI were carried out on a cohort of patients with untreated cerebral gliomas. It is shown that the results from multiple quantum filtered sodium MRI using the enhanced SISTINA sequence are related to IDH mutational status in contrast to [18F]-FET-PET, thus providing the diagnostic potential to predict the genetic profile of gliomas.
Introduction
The diagnosis of cerebral gliomas benefits from the use of O-(2-[18F]fluoroethyl)-L-tyrosine ([18F]-FET) based PET1-3. This study aims to complement [18F]-FET-PET with sodium MRI as a means to observe and evaluate tumour metabolism. The relationship between [18F]-FET-PET and sodium MRI in patients with cerebral gliomas in terms of the mutational status of the enzyme isocitrate dehydrogenase (IDH) is explored.Methods
Ten patients with untreated cerebral gliomas underwent dynamic [18F]-FET-PET and sodium MRI using an enhanced SISTINA sequence4 to estimate sodium concentrations: total sodium concentration (NaT), weighted non-restricted sodium concentration (NaNR) and restricted sodium concentration (NaR), in tumours and normal brain tissue. NaNR is assumed to relate mainly to sodium in extracellular space, whereas NaR is alleged to indicate mostly intracellular space. Sodium and [18F]-FET uptake are compared for tumours with different IDH mutational status. Following biopsy or resection, histology and the IDH mutational status were determined neuropathologically. Informed consent was granted by all subjects.
Sodium MRI experiments were carried out on a 4T scanner with a Siemens console and a dual tuned Na/H birdcage coil (Rapid Biomedical, Germany), using a 320x240x160mm3 field of view with 10mm isotropic resolution. A 12 step, multiplex phase cycle was implemented5 to separate single quantum (SQ) and triple quantum (TQ) signals. A repetition time of TR=150ms led to a total acquisition time of approximately 8min. A preparatory time τ=7ms allowed for a readout train consisting of 5 radial acquisitions with a base resolution of 22 and bandwidth of 1kHz/pixel sampled at TEute= 0.36ms; 1.75ms; 3.14ms; 4.53ms; 5.92ms after the first RF pulse. DISCOBALL6 was used as the radial acquisition scheme, yielding a full-width-half-maximum of the point spread function of approximately 6mm. The mixing time δ was 40µs. Following the third RF pulse multiple quantum filtered images were acquired using Cartesian sampling of monopolar gradient echoes (mGRE) at 5.92ms; 15.10ms; 25.32ms; 34.48ms; 43.64ms; and 52.80ms, using a bandwidth of 120Hz/pixel.
FET-PET Dynamic PET data were acquired on an ECAT EXACT HR+ scanner (Siemens Medical Systems, Inc.) in 3-dimensional mode (32 rings; axial field of view of 155mm) up to 3000s after intravenous injection of ~200MBq [18F]-FET. The reconstructed dynamic dataset consisted of 16 time frames (5×60s; 5×180s; 6×300s). A 10min transmission scan measured with three rotating line sources (68Ge/68Ga) served as the basis for attenuation correction. Random and scattered coincidences, as well as dead time, was accounted for prior to the iterative reconstruction of 63 image planes using the OSEM algorithm (16 subsets, 6 iterations). The reconstructed image resolution7 is approximately 5.5mm. Tumour-to-brain ratios (TBR) were calculated from [18F]-FET-PET images taken 1200-2400s post injection by dividing the mean and maximal ROI value of the lesion by the mean ROI value of normal brain tissue. If the maximum TBR exceeded a threshold of 1.6, the lesion was considered to be [18F]-FET positive8.
Results
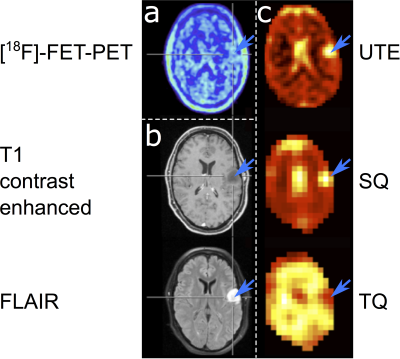
IDH mutated gliomas show significantly elevated NaT (p=0.05), TBR of NaT (p=0.02), NaNR (p=0.003), and NaT/NaR (p<0.001) as well as a significantly reduced NaR (p=0.01) compared to IDH wildtype gliomas (n=5 patients each). [18F]-FET parameters (TBR and time-to-peak) were not predictive for IDH status in these patients (see figure 1 for sample data from one subject). There was no notable relationship between sodium distribution and [18F]-FET uptake.Conclusions
Sodium MRI seems to be more strongly associated with the IDH mutational status of gliomas than [18F]-FET-PET, thus sodium MRI has the potential to predict the IDH mutational status of gliomas.Acknowledgements
The authors wish to express their since gratitude to Prof. Dr. Norbert Galldiks, Prof. Dr. Bernd Neumaier, Dr. Johannes Lindemeyer, Dr. Gabrielle Stoffels, Petra Engels, Elke Bechholz, Anita Köth and Claire Rick.References
1. Kunz M et al. Dynamic 18F-FET PET is a powerful imaging biomarker in gadolinium-negative gliomas. Neuro-oncology. 2018.
2. Suchorska B et al. Identification of time-to-peak on dynamic 18F-FET-PET as a prognostic marker specifically in IDH1/2 mutant diffuse astrocytoma. Neuro-oncology. 2018;20:279-88.
3. Verger A et al. Static and dynamic (18)F-FET PET for the characterization of gliomas defined by IDH and 1p/19q status. European journal of nuclear medicine and molecular imaging. 2018;45:443-51.
4. Worthoff WA, Shymanskaya A, Shah NJ. Relaxometry and quantification in simultaneously acquired single and triple quantum filtered sodium MRI. Magn Reson Med. 2018;00:1-13.
5. Fiege DP et al. Simultaneous single-quantum and triple-quantum-filtered MRI of 23Na (SISTINA). Magn Reson Med. 2013;69:1691-6.
6. Stirnberg R et al. A new and versatile gradient encoding scheme for DTI: a direct comparison with the Jones scheme. Proc Int Soc Mag Reson Med. 2009; 17:3574.
7. Herzog H et al. NEMA NU2-2001 guided performance evaluation of four siemens ECAT PET scanners. Ieee T Nucl Sci. 2004;51:2662-9.
8. Pauleit D et al. O-(2-[18F]fluoroethyl)-L-tyrosine PET combined with MRI improves the diagnostic assessment of cerebral gliomas. Brain : a journal of neurology. 2005;128:678-87.
Figures