3896
Increased Functional Connectivity of Insular Subregions Imporves Smoking Cessation Likelihood1Radiology, the Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, China
Synopsis
Most smoking cessation attempts result in failure. Damage to the insular cortex disrupts smoking behavior in stroke patients. Neuroimaging studies have shown that chronic smokers had altered functional connectivity (FC) of insular subregions. Here, we further investigated the different FC changes of insular subregions between smokers who quitted successfully and those who relapsed after smoking cessation treatment. We found that smokers who quitted successfully had increased FC of anterior insula, while those who relapse had decreased FC of insular subregions. These novel findings suggest that increased connectivity of insular subregions could imporve likelihood of cessation.
Purpose
To date, most smoking cessation attempts still result in failure 1. Damage to the insular cortex, but not to other parts of the brain, disrupts smoking behavior in stroke patients and animals2-4, which supports a critical role for the insula in the smoking cessation. Recently, neuroimaging studies have shown that chronic smokers had altered functional connectivity (FC) of insular subregions compared with non-smokers5-6. However, the recovery neuromechanism underlying tobacco addiction in the FC network of insular subregions remains unclear. Based on our review of previous sudies, we hypothesized that different smoking cessation outcomes (quitting successfully or not) might be associated with different FC changes of insular subregions after smoking cessation treatment. Therefore, by longitudinal comparisons between post- and pre-treatment smokers using resting-state FC, the present study subdivided the insula into two subregions and further investigated the different FC changes of insular subregions between the smokers who quitted successfully and those who relapsed after smoking cessation treatment.Methods
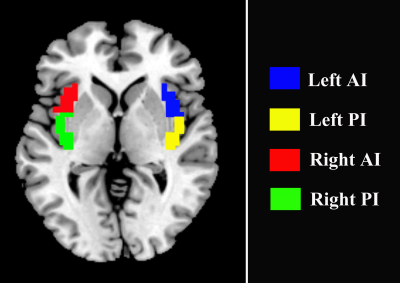
Nicotine-dependent smokers were initially recruited into the study, all of whom underwent magnetic resonance imaging (MRI) scans. After a 12-week treatment with varenicline, a first line drug for smoking cessation, 30 smokers,including 16 smoking quitters and 14 relaspers, who underwent an additional MRI scan were finally included in the study. All the data were acquired using a 3.0 T GE Signa MR scanner. Functional image preprocessing and statistical analyses were conducted using DPABI toolbox (Yan, et al., 2016, http://rfmri.org/DPABI), which is performed on the MATLAB (The Mathworks, Inc., USA). Both left and right insula were segmented into anterior and posterior portion (Fig. 1). Then, the left and right insular subregions were used as seed regions for seed-based FC analysis. FC maps were generated by calculating. Lastly, paired t-tests were used to test the differences of between FC maps of post- and pre- treatment in both quitters and relaspers groups. The resulting statistical maps were corrected for multiple comparisons using Gaussian random field (GRF) correction (voxel Z ˃ 2.3, cluster-level p < 0.05, two-tailed). In addition, to examine whether or not the FC between quitter and relasper groups is different at baseline, the FC maps between the two groups were compared using a independent two-sample t-test.Results
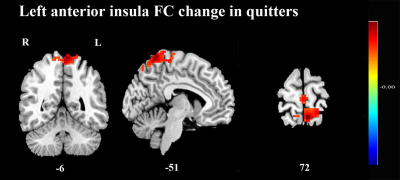
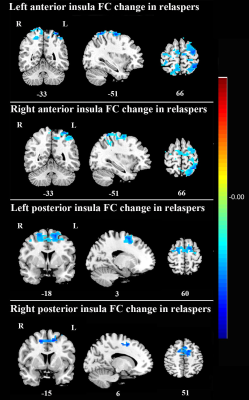
There was no significant difference in age, education, smoking data between the relapsers and quitters. After smoking cessation treatment using varenicline, FC maps between post- and pre- treatment were compared using paired t-tests in both quitter and relasper groups. Compared with the pre-treatment group, quitters of the post-treatment showed significantly increased FC between left anterior insula and left precuneus (Fig 2), but no significant change in the other three insula-subregions-based FC; relapsers of the post-treatment showed significantly decreased FC between bilateral anterior insula and left superior parietal gyrus (Fig 3), and significantly decreased FC between bilateral posterior insula and left dorsolateral superior frontal gyrus (Fig 3). In addition, no significant difference was found in the FC of insular subregions between quitters and relaspers groups at baseline before treatment.Discussion and Conclusion
The present longitudinal study used resting-state fMRI to compare the FC changes of insular subregions after smoking cessation treatment in nicotine-dependent smokers with different cessation outcomes. To our knowledge, this is the first study to examine the FC network of insular subregions to investigate the recovery neuromechanism underlying tobacco addiction and neural correlates associated with the treatment effect of nicotine dependence. Smokers who quitted successfully had increased FC between left anterior insula and a key node of default mode network─the left precuneus, while those who relapse had decreased FC between insular subregions and two major nodes of the executive control network─the left superior parietal gyrus and dorsolateral superior frontal gyrus. Taken together, increased connectivity of insular subregions could help imporve smoking cessation likelihood. These FC network of insular subregions may be important therapeutic targets for nicotine-dependent smokers. These novel findings suggest the recovery neuromechanism underlying the tobacco addiction and the neural correlates associated with the treatment effect of nicotine dependence.Acknowledgements
CW was supported by zhejiang provincial natural science foundation (LQ18H180001) and Medical and Health Scientific Research Fund Project of Zhejiang Province (2017KY080). MZ was supported by the Natural Science Foundation of China Surface Project (81171310).References
1. Cahill K, Stead LF, Lancaster T (2010). Nicotine receptor partial agonists for smoking cessation. The Cochrane database of systematic reviews(12): CD006103.
2. Abdolahi A, Williams GC, Benesch CG, Wang HZ, Spitzer EM, Scott BE, et al (2015). Damage to the insula leads to decreased nicotine withdrawal during abstinence. Addiction 110(12): 1994-2003.
3. Naqvi NH, Rudrauf D, Damasio H, Bechara A (2007). Damage to the insula disrupts addiction to cigarette smoking. Science 315(5811): 531-534.
4. Scott D, Hiroi N (2011). Deconstructing craving: dissociable cortical control of cue reactivity in nicotine addiction. Biological psychiatry 69(11): 1052-1059.
5. Bi Y, Yuan K, Guan Y, Cheng J, Zhang Y, Li Y, et al (2017). Altered resting state functional connectivity of anterior insula in young smokers. Brain Imaging Behav 11(1): 155-165.
6. Stoeckel LE, Chai XJ, Zhang J, Whitfield-Gabrieli S, Evins AE (2016). Lower gray matter density and functional connectivity in the anterior insula in smokers compared with never smokers. Addiction biology 21(4): 972-981.
Figures