3854
Automatic pelvic bone registration of low-field MRI to pre-operative 3T-MRA for vascular interventions1Magnetic Detection & Imaging, University of Twente, Enschede, Netherlands, 2Vascular surgery, Medisch Spectrum Twente, Enschede, Netherlands, 3Robotics and Mechatronics, University of Twente, Enschede, Netherlands
Synopsis
Intra-operative low-field magnetic resonance imaging (LF-MRI) is characterized by poor spatial resolution limiting its use in endovascular catheter tracking with fluoroscopy. Fusion of LF-MRI with 3T-MRI may overcome this issue. To fuse both volumes, we automatically segmented a pelvis from an LF-MRI scan, based on cortical bone detection with Laplacian edge detection. A similarity of 66% was reached between automatic and manual segmentation. Subsequently, the segmented volume was automatically registered to a 3T-MRA. Compared to a Procrustes based manual registration, the automatic registration resulted in a root-mean-square error of 3 mm.
Introduction
Partial or complete obstruction of one or more peripheral arteries is assigned as peripheral arterial occlusive disease (PAOD) and is commonly caused by advanced atherosclerotic plaques deposit. A well accepted strategy for PAOD treatment is percutaneous transluminal angioplasty (PTA), in which stenotic lesions are localized and treated with a balloon catheter and/or with stent placement. Fluoroscopy is widely used to trace the endovascular catheter during PTA, but uses ionizing radiation, requires contrast agents that are unsuited for patients with renal deficiency1 and might lead to ergonomic injuries of personnel caused by wearing lead aprons2.
These problems can be addressed by using intra-operative low-field MRI (LF-MRI). LF-MRI is characterized by an open magnet design but has poor spatial resolution when temporal resolution is maximized to 3-5 frames per second3. Fusion with pre-operative magnetic resonance angiography (MRA) or computed tomography angiography (CTA) may tackle this problem of poor spatial resolution. The overall goal of this work was to demonstrate an automatic segmentation method of the pelvic bone from an LF-MRI scan, followed by rigid registration with 3T-MRA data. This fusion would enable the possibility to provide a high spatiotemporal resolution arterial roadmap to the PTA operator during the intervention.
Methods
An LF-MRI (Esaote G-scan Brio 0.25T) and 3T-MRI (Siemens MAGNETOM Skyra 3T) dataset of the same volunteer were used. For LF-MRI, a dual echo steady-state free precession sequence (SHARC) was used to depict the pelvic bones and for 3T-MRI a non-contrast enhanced (NCE) MRA sequence (NATIVE SPACE) was used for angiography. Due to the limited FOV of the LF-MRI system, only half of the pelvic bone could be segmented.
Segmentation of the LF-MRI pelvic bone was achieved by combining Laplacian-based edge detection, thresholding, morphological operations, Gaussian blurring and filling. The automatic segmentation was compared with a manual segmentation. The 3T-MRI pelvic bones were manually segmented out of the unsubtracted NATIVE SPACE images.
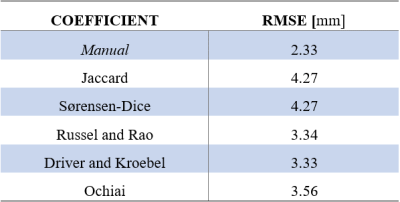
To fuse the two segmented volumes automatically, translational and rotational differences between both volumes were successively corrected by an in-house developed converging algorithm. Translational deviation was estimated in a spherical pattern with a shrinking radius, thus with increasing accuracy. Rotation parameters were found by defining finite discrete rotation possibilities. The rotation parameters converge towards an optimum by decreasing the angular step size. Automatic registration after maximal 30 iterations was assessed with the Root Mean Square Error (RMSE) of five pre-defined bony landmarks in both volumes. Manual registration was performed using the Procrustes algorithm4.
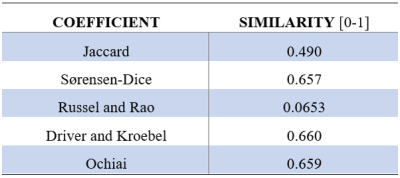
Similarity between both manually and automatically segmented volumes was examined with the following similarity measures: Jaccard, Sørensen-Dice, Russel and Rao, Driver and Kroebel and the Ochiai coefficient. The developed volume-based registration algorithm used all of these measures separately for registration resulting in different RMSEs.
Results
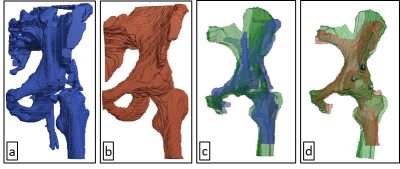
The automatic 3D segmentation of the LF-MRI pelvic bone is visible in Figure 1a and the manually segmented equivalent in Figure 1b. Table 1 shows the quantitative results of segmentation defined by the similarity indices. The final automatic registration result is visible in Figure 1c, and the manual registration result is depicted in Figure 1d. The RMSE in the position of the five anatomic landmarks after manual and automatic registration are denoted in Table 2.Discussion
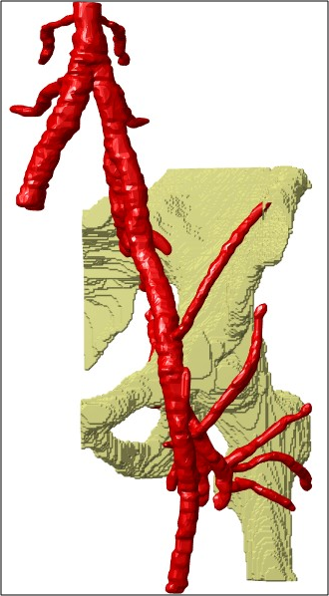
This study proposes a new method for automatic image fusion of 3T-MRI with LF-MRI to provide a PTA operator with an arterial roadmap with high spatio-temporal resolution. Currently, the registration results are hampered by unintended inclusion of the femur which position may vary between different scan moments (see the difference between Figure 1c and 1d). Final registration accuracy can be improved by removing the femur from the segmented volume. In addition, the registration error may decrease by addressing the transformation as a 6D problem, instead of two separate 3D problems. In this research, a first step towards superseding fluoroscopy by LF-MRI during PTA interventions is made. To emphasize the potential value of the achievements reached in this study, Figure 2 shows a NCE 3T-MRA that was manually segmented but automatically registered to LF-MRI with the obtained transformation parameters.Conclusion
In this study, a method for automatic segmentation and registration of the LF-MRI reconstructed pelvic bone was shown with a Driver and Kroebel similarity index of 0.66. Finally, automatic registration of the segmented pelvic bone on LF-MRI to a 3T-MRI derived pelvic bone was managed with an RMSE of 3.3 mm. This fusion method could be applied in other vascular domains.Acknowledgements
No acknowledgement found.References
1. McCullough, P. A. Contrast-Induced Acute Kidney Injury. J. Am. Coll. Cardiol. 51, 1419–1428, DOI: 10.1016/j.jacc.2007.12.035 (2008).
2. Alexandre, D., Prieto, M., Beaumont, F., Taiar, R. & Polidori, G. Wearing lead aprons in surgical operating rooms: ergonomic injuries evidenced by infrared thermography. J. Surg. Res. 209, 227–233, DOI: 10.1016/j.jss.2016.10.019 (2017).
3. Quick, H. H. et al. Interventional MRA using actively visualized catheters, TrueFISP, and real-time image fusion. Magn. Reson. Medicine 49, 129–137, DOI: 10.1002/mrm.10334 (2003).
4. Schönemann, P. H. A generalized solution of the orthogonal procrustes problem. Psychometrika 31, 1–10, DOI:10.1007/BF02289451 (1966).
Figures