3066
Identifying Rapamycin as a potential preventative therapeutic for Alzheimer’s disease through Multimodal MRl1Department of Pharmacology and Nutritional Sciences, University of Kentucky, Lexington, KY, United States, 2Sanders Brown Center on Aging, University of Kentucky, Lexington, KY, United States
Synopsis
The ε4 allele of apolipoprotein E gene (APOE4) is the strongest genetic risk factor for Alzheimer’s disease (AD). Studies have indicated that APOE4 carriers develop vascular and metabolic dysfunctions several decades prior to the clinical symptom of dementia occurs. In this study, we used multi-modal MRI markers to investigate the effect of Rapamycin, a FDA approved drug, on genetically modified pre-symptomatic E4FAD mice, as a preventative therapeutic for AD. Cerebral blood flow and crucial brain metabolites detected by MR spectroscopy were restored in Rapamycin fed mice, consistent with lower BOLD responses, lower cerebrovascular-reactivity (CVR) and decreased Amyloid-beta deposition.
Introduction
The ε4 allele of apolipoprotein E gene (APOE4) is the strongest genetic risk factor for Alzheimer’s disease (AD). Studies have indicated that APOE4 carriers develop vascular deficiency and metabolic dysfunctions several decades prior to the clinical symptom of dementia occurs1,2, including amyloid beta (Aβ) deposition. Although AD research is bustling, a concrete and effective treatment has yet to be discovered. This study aimed to identify the preventative effects of Rapamycin, a FDA approved drug, on genetically modified pre-symptomatic E4FAD mice, as a potential intervention for restoring brain vascular, metabolic functions and ultimately becoming an effective prevention. Specifically, we employed arterial spin labeling (ASL) for cerebral blood flow measurements, BOLD fMRI, brain water content measurement and proton MR spectroscopy (1H-MRS) for brain metabolites determination. Since Rapamycin has been showed restoring cerebrovascular functions in symptomatic hAPP (J20) mice3, we hypothesized that brain structure and functions can be restored by Rapa in this E4FAD mouse model.Methods
Animals E4FAD mice4 co-express human Aβ via 5xFAD mutations (APP K670N/M671L+I716V+V717I and PS1 M1461L+L286V) on C57BL/6 homozygous APOE4 knock-in genotypes5,6. All animal experimental protocols have been approved by the Institutional Animal Care and Use Committee at the University of Kentucky and in compliance with the ARRIVE guidelines. Mice were fed with Control or Rapa supplemented diet for 16 weeks, starting from 3 months of age. Vascular and metabolic neuroimaging MRI and MRS experiments were performed on a 7T MR scanner. T2-weighted structural images were acquired with field of view (FOV) = 18x18 mm2, matrix = 256x256; slice thickness = 1 mm, 10 slices, repetition time (TR) = 1500 ms and echo time (TE) = 35 ms. Quantitative CBF was measured using MRI-based pCASL techniques7. Paired control and label images were acquired in an interleaved fashion with a train of Hanning window-shaped radiofrequency pulses of duration/spacing = 200/200 us, flip angle = 25° and slice-selective gradient = 9 mT/m, and a labeling duration = 2100 ms8. pCASL image analysis was employed with in-house written codes in MATLAB9. 1H- MRS studied the whole brain metabolites. Immunohistochemistry Paraformaldehyde-fixed whole brains were coronally cut. Sections were incubated with an anti-Aβ antibody, MOAΒ-2. Statistics Statistical analyses were completed using GraphPad Prism (GraphPad, San Diego, CA, USA). Two-tailed Welch’s t-test was performed for determination of differences between groups. In addition, binary logistic regression was used to classify DS participants based on their MRS data. Levels of statistical significance were reached when p<0.05.Results
Rapamycin restored cerebral blood flow (CBF) of pre-symptomatic E4FAD mice. In both APP- and APP+ mice, Rapa had significantly increased CBF compared to control group, p=0.023 & p=0.002 respectively. Rapamycin increased water content (blood brain partition coefficient, BBPC), lowered BOLD responses and cerebrovascular-reactivity (CVR) in E4FAD mice. Several brain metabolites were increased in Rapa mice: Alanine, Glutamate, choline-containing pholipids (Phosphocholine, GPC+PCh), NAA and Glx (Glu+Gln). These suggest excitatory neurotransmission, anti-oxidative stress state and neuronal membrane integrity were significantly enhanced. As the hallmark of AD, decreased Amyloid-beta deposition was observed after 16 weeks treatment, which suggests Rapa facilitates A-beta clearance.Discussion
These methods provided a comprehensive analysis of the subjects’ brain vascular health by providing insight on CBF, brain water content, functional MRI, brain metabolites and Amyloid-beta deposition.
E4FAD mice had metabolic and vascular deficits at early age. Our data showed Rapamycin can restore CBF and vascular density. Increased water content suggested Rapamycin leads to the hydration of brain, more specific, hippocampal water content is effectively increased by Rapa, which is shown to prevent brain atrophy structurally and cognition impairment functionally. Metabolic rate reductions as major factor leading to brain structural integrity and cognitive impairment later in life, our data showed positive alteration within Rapa mice. Allied with all, less Amyloid-beta deposition also supports this early intervention is effective. Thus, the findings may pave a way for clinical trial to potentially prevent AD in cognitively normal APOE4 carriers. Future study will further look at neurovascular coupling using functional MRI and mitochondrial activity, a greater understanding of the mechanism need to be elucidated.
Conclusion
These results support that Rapamycin protests CBF and metabolic functions in asymptomatic mice expressing the human APOE4 gene. This shows promise that by effective early intervention, brain health might be optimized to prevent neurological disorders, even in individuals with the highest genetic risk factor APOE4. Vascular neuroimaging can be used as early detection techniques and Rapa holds high potential in preventing AD development.Acknowledgements
No acknowledgement found.References
[1] Thambisetty M., B.-H. L. (2010). APOE epsilon4 genotype and longitudinal changes in cerebral blood flow in normal aging. Arch Neurol, 67, 93-98.
[2] Fleisher. A. S. et. al. (2013). Apolipoprotein E epsilon4 and age effects on florbetapir positron emission tomography in healthy aging and Alzheimer disease. Neurobiol Aging, 34, 1-12.
[3] Ai-Ling Lin, et. al. (2013). Chronic rapamycin restores brain vascular integrity and function through NO synthase activation and improves memory in symptomatic mice modeling Alzheimer’s disease. Journal of Cerebral Blood Flow & Metabolism, 33, 1412–1421.
[4] Youmans KL, et. al. (2012). APOE4-specific changes in Aβ accumulation in a new transgenic mouse model of Alzheimer disease. J Biol Chem, 287:41774–41786.
[5] Oakley H., et. al. (2006). Intraneuronal beta-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer's disease mutations: potential factors in amyloid plaque formation. J Neurosci, 26(40): p. 10129-40.
[6] Sullivan P.M., et. al. (1997). Targeted replacement of the mouse apolipoprotein E gene with the common human APOE3 allele enhances diet-induced hypercholesterolemia and atherosclerosis. J Biol Chem, 272(29): p. 17972-80.
[7] Parikh I., et. al. (2016). Caloric restriction preserves memory and reduces anxiety of aging mice with early enhancement of neurovascular functions. Aging (Albany NY), 8, 2814–2826.
[8] Hong X., et. al. (2015). Evaluation of EPI distortion correction methods for quantitative MRI of the brain at high magnetic field. Magn. Reson. Imaging, 33, 1098–1105.
[9] Alsop D. C., D. J. (2015). Recommended implementation of arterial spin-labeled perfusion MRI for clinical applications: a consensus of the ISMRM perfusion study group and the European consortium for ASL in dementia. Magn. Reson. Med., 73, 102–116.
Figures
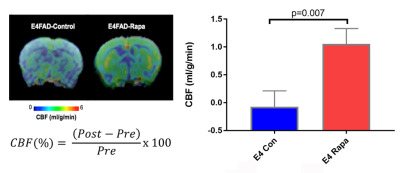
Figure 1. Rapamycin increased Cerebral Blood Flow in E4FAD mice.
Low CBF in E4FAD was very significantly increased by Rapa treatment. (p=0.007, Mean ± SEM = -0.7154 ± 0.4355, Mean ± SEM = 1.51 ± 0.412) Change in CBF measurement from post-treatment mice is compared to pre-treatment mice as a function of change. n=6 per experimental group, P=0.0223, significance of differences between group means was determined using two-tailed Welch’s t-test.
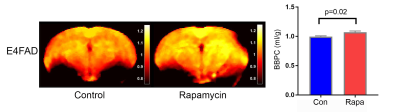
Figure 2. Rapamycin increased water content in E4FAD mice.
Using in vivo neuroimaging, we found that Rapamycin increased water content (blood brain partition coefficient, BBPC) in E4FAD mice, compared to control group. Water content was measured by MRI, calculated as Blood Brain Partition Coefficient (BBPC) = (Waterblood/Waterbrain)/Brain density. Left – BBPC map; Right – quantitative analyses of hippocampus BBPC. n=6 per experimental group, P=0.0223, significance of differences between group means was determined using two-tailed Welch’s t-test.
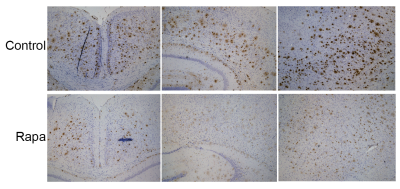
Figure 3. Rapamycin facilitates Amyloid-beta clearance.
Rapa fed mice showed less Amyloid-beta deposition compared to the control group. Images were captured on a Zeiss Axio Imager M1 under identical capture settings at ×4 or ×10 magnification.
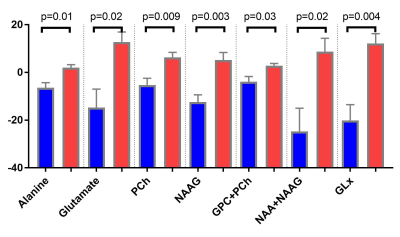
Figure 4. Brain metabolites changes detected by MR spectroscopy.
After 16 weeks treatment, Rapa mice had increased Alanine, Glutamate, choline-containing pholipids, NAAG, NAA+NAAG and Glx compared to control group.
NAA, at 2.0 ppm, is an amino acid derivative synthesized in neurons and transported down axons. Glutamate, the main excitatory neurotransmitter in brain. Glx, which lies between 2.1 and 2.4 ppm, is a mixture of glutamate and glutamine, which is involved in excitatory/inhibitory neurotransmission. Alanine, which resonates at 3.5 ppm, is the precursor of carnosine, represents anti-oxidative stress. Choline-containing compounds are GPC and Phosphate Choline, the biomarkers for membrane phospholipid metabolisms.
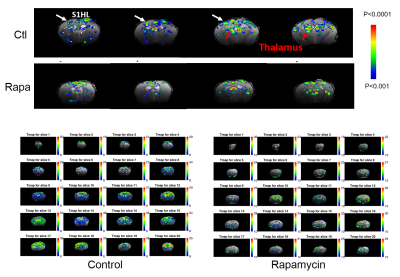
Figure 5. Rapamycin-treated E4FAD mice had lower BOLD responses and lower cerebrovascular-reactivity (CVR).
Blood-oxygen-level dependence (BOLD) responses were tested with right hindpaw stimulation, 1 mA, 9 Hz, 0.3 ms square pulse; 30s duration. CVR was obtained by hypercapnic challenge with 5% CO2 for 3 minutes stimuli period.