2121
Longitudinal study of ventricular remodeling in tetralogy of Fallot patients using CMR coupled with biomechanical modeling1Inria Saclay Ile-de-France, Palaiseau, France, 2King's College London, London, United Kingdom, 3LMS, Ecole Polytechnique, Palaiseau, France, 4Division of Pediatric Cardiology, UT Southwestern Medical Center, Dallas, TX, United States, 5Evelina London Children’s Hospital, Guy's & St Thomas' NHS Foundation Trust, London, United Kingdom
Synopsis
We employ a biophysical modeling framework to augment the information obtained from cardiovascular MRI data of patients with chronic pulmonary valve regurgitation in order to assist in optimal timing of pulmonary valve replacement therapy (PVR). The longitudinal follow-up of patients post-PVR shows no significant change in ventricular ejection fraction. However, the model is able to detect a decreasing request on generation of active myocardial stress while not decreasing the cardiac output – presumably favorable with the long-term prognosis of the heart. Coupling biophysical modeling with MRI data has the potential to further augment the diagnostic value of MRI.
Introduction
CMR is an established gold-standard imaging method for the assessment of heart's function. Ventricular ejection fraction (EF) is considered as a measure of systolic function. The valvular insufficiency is typically quantified by the level of regurgitation fraction (RF). EF is however known to be dependent on the actual ventricular filling (preload) and the resistance of the circulation (afterload). Furthermore, atrio-ventricular regurgitation (AVR) increases EF without a sufficient output into the circulation. Finally, chronic ventricular volume overloading due to an incompetent outflow valve leads to ventricular dilation and an increase of end-diastolic volume (EDV) – an example of pathological ventricular remodeling. The overall interpretation of ventricular function is therefore intricate in complex cases, such as in repaired tetralogy of Fallot patients (rToF), who suffer from right ventricular (RV) volume and/or pressure overload due to chronic pulmonary regurgitation (PR), possible residual outflow tract stenosis, and additionally often tricuspid valve regurgitation (TR). The pulmonary valve replacement (PVR) is timed according to cut-offs in CMR-derived indices of EF, RF and EDV, however, with a limited sensitivity and specificity [1]. Therefore, new criteria need to be found.
The mechanical character of the cardiovascular system suggests employing biomechanical modeling. Cardiovascular modeling interacting with clinical data has already reached some proof-of-concept studies [2]. The computational intensity of complex 3D models is practically out of reach to be directly linked to an MRI console. Model-reduction proposed in [3] – which simplifies the geometry while keeping all physical and physiological assumptions – allows fast computation, and is therefore suitable for linking with a CMR post-processing unit. As presented in [4,5], such a model calibrated to individual patients (including complex cases with a combined PR, TR and reduced EF) allows to access e.g. the mechanical quantitates of ventricular contractility (the active stress generated by the myocardium, i.e. the objective measure of ventricular systolic function not directly visible in image data). The presented work builds on [5], and adds the longitudinal CMR either preceding PVR or follow-up scans 6 months post-PVR. The RV remodeling and reverse-remodeling (observed in the scans) can be characterized – additionally to the CMR derived metrics – by biomechanical quantities, which might play a role in patient management and optimal PVR timing.
Methods
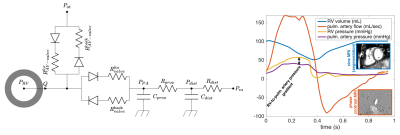
Ten rToF patients were included in the study. All of them underwent percutaneous PVR, during which RV and pulmonary artery (PA) pressures were recorded prior to and after valve deployment. Each patient had a CMR exam before the PVR (see Fig. 1). Three patients underwent in addition a CMR scan 6 months after PVR. Patient-specific biomechanical models [3,4,5] of RV were calibrated for each subject and time point. The step-by-step calibration of the pulmonary circulation (Windkessel model), properties of pulmonary and tricuspid valve, geometrical properties of heart (size, wall thickness) and mechanical properties of myocardium (passive stiffness and active contractility) was performed to match the acquired clinical data. The time-evolution of biomechanical quantities of RV contractility; pulmonary vascular resistance (Rdist); pulmonary vascular capacitance (Cdist) were captured.Results
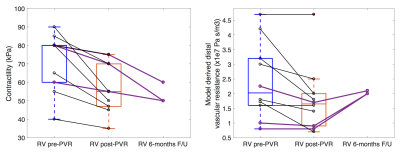
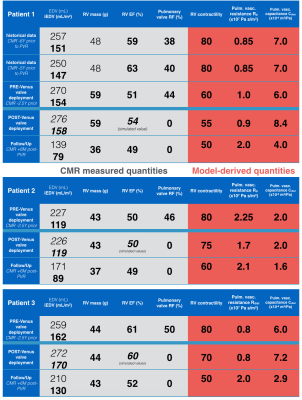
Fig 2. shows a systematic trend of a decrease of myocardial contractility observed immediately after deploying the valve, and a decrease of the resistance in the pulmonary circulation. Tables in Fig. 3 then demonstrate the evolution of the quantities directly obtained from CMR and of biomechanical properties accessed thanks to modelling in three longitudinally followed patients.Discussions
The model-suggested decrease of myocardial contractility after PVR would mean the heart functioning on a more favorable level: providing an adequate cardiac output into the circulation (in fact, even 20-50% higher than during the chronic pulmonary regurgitation [6]) with lower energy needs. The model suggests that the RV contractility is decreasing further 6 months after PVR, where we observe a reverse-remodeling of RV (normalisation of volumes). The immediate decrease of pulmonary vascular resistance might be associated with effectively higher flow through the pulmonary circulation, while the pulmonary pressures did not raise. On the contrary, the CMR derived EF would suggest a worsening of the cardiac function.
Conclusions
The work shows a feasibility of employing a simplified biomechanical modeling framework to characterize the actual pathophysiological state of rToF patients longitudinally during the pathological remodeling of RV (due to chronic overload) and reverse-remodeling after a therapeutic intervention. The longitudinal follow-up data available thanks to regular CMR exams allow to characterize the key components of the process. Such models of long-term evolution have the potential in timing optimization in a number of cardiac pathologies as the presented rToF, and could possibly be adapted also for management of other pathologies, e.g. aortic valve stenosis, or aortic coarctation.Acknowledgements
Associated Inria Team ToFMOD (jointly funded by Inria and UTSW Medical Center Dallas).References
[1] Quail MA, Frigiola A, Giardini A, et al. Impact of pulmonary valve replacement in tetralogy of Fallot with pulmonary regurgitation: A comparison of intervention and nonintervention. Ann Thorac Surg. 2012;94(5):1619-1626. doi:10.1016/j.athoracsur.2012.06.062.
[2] Chabiniok R, Wang VY, Hadjicharalambous M, et al. Multiphysics and multiscale modelling, data–model fusion and integration of organ physiology in the clinic: Ventricular cardiac mechanics. Interface Focus. 2016;6(2):20150083. doi:10.1098/rsfs.2015.0083.
[3] Caruel M, Chabiniok R, Moireau P, Lecarpentier Y, Chapelle D. Dimensional reductions of a cardiac model for effective validation and calibration. Biomech Model Mechanobiol. 2014;13(4):897-914. doi:10.1007/s10237-013-0544-6.
[4] Chabiniok R, Moireau P, Kiesewetter C, Hussain T, Razavi R, Chapelle D: Assessment of atrioventricular valve regurgitation using biomechanical cardiac modeling, Proc. of FIMH 2017, Volume 10263 of LNCS, pp. 401–411, Springer 2017.
[5] Chabiniok R, Tompkins RM, Gusseva M, Tandon A, Greil G, Moireau P, Chapelle D, Hussain T: Augmenting the interpretation of cardiac MRI by biomechanical modeling: Applcation to Tetralogy of Fallot, conference abstract #4788 ISMRM2018.
[6] Lurz P, Nordmeyer J, Muthurangu V, Khambadkone S, Derrick G, Yates R, Sury M, Bonhoeffer P, Taylor AM: Comparison of Bare Metal Stenting and Percutaneous Pulmonary Valve Implantation for Treatment of Right Ventricular Outflow Tract Obstruction, Circulation 2009.
Figures