0887
Increased Functional Connectivity Flexibility During Early Infancy1School of Data and Computer Science, Sun-Yat Sen University, Guangzhou,Guangdong, China, 2Department of Radiology and BRIC, University of North Carolina at Chapel Hill, chapel hill, NC, United States
Synopsis
Understanding the nature of dynamic neural interactions during development is a critical issue of cognitive neuroscience. However, our knowledge
Introduction
Increasing studies focused on delineating how the human brain develops in the first two postnatal years, as it is one of the most critical periods of life with the most rapid development in both brain function and structure1. Our current understanding of brain functional organization in infants was built upon the assumption of static functional connectivity (FC)1-3. However, mounting evidence has been indicating that the brain functional network is highly dynamic (with its major topology changing at every a few seconds) to support a remarkable range of complex cognitive abilities and better interact with the changing environment4. Such an ability could be emerging over the course of development5. However, our knowledge on its early development in neonates and infants is very limited. In this work, we are trying to fill this gap by exploring the early development of the temporal dynamics of the brain functional network. By computing temporal variability of regional FC architecture for each brain region and charting its longitudinal trajectory, our results provide a completely new and thorough view of how brain functional adaption and flexibility emerge and develop during such a pivotal period.Methods
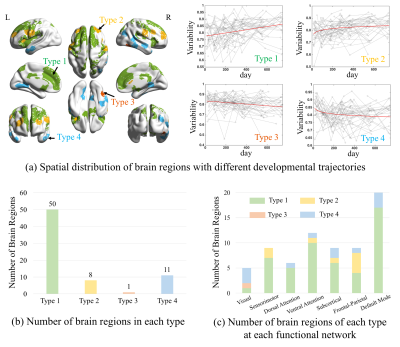
Fifty-one typically developing infants with 200 longitudinal rs-fMRI scans from seven time points (0, 3, 6, 9, 12, 18 and 24 months of age) were used in this study. Data preprocessing was adopted from our recent paper3, with wavelet despiking instead of censoring for alleviating head motion artifacts without destroying temporal continuity that is necessary for dynamic FC calculation. After preprocessing, we parcellated the whole brain into 217 regions (excluding the cerebellum) with Shen et al.’s template6 and extracted mean time series for each brain region. Temporal variability of the regional FC profiles was calculated measuring the dissimilarity of the region’s FC feature vector across all the sliding windows, according to a recent method7. Specifically, we segmented the time courses into N non-overlapping windows with equal lengths. In each window, we computed the Pearson’s correlation coefficients between a brain region k to other regions as a length-217 FC vector of this region. Then, the pairwise similarity between any two FC vectors from the respective windows was calculated and all these similarities were averaged to represent the temporal variability (one minus mean similarity) of the whole-brain FC of the region k. During such a process, different window lengths and different starting points were considered and their results were averaged as the final FC variability measurement7. After that, we used a linear (or a log-linear) mixed-effect regression (LMER) model to delineate possible linear (or exponential) developmental trajectory of the FC variability for each region. Subject-specific intercept and slope were modeled as random effects. Akaike information criterion was used for linear or log-linear model selection. The significance was set to p < 0.05 after false discovery rate correction.Results and Discussion
70 (32% of all 217) regions show significant changes in FC dynamics along development (Fig. 1a). Among them, the regions with the significant increase (58 regions, types 1 and 2) in FC variability, mainly located at high-order function-related regions and association areas (especially in the default mode network (DMN) and attention network (AN)), are much more than those compassing primary visual and temporal areas with decreasing FC variability (12 regions, types 3 and 4). While the whole-brain functional network is gradually reconfigured towards more flexible, dynamic, and adaptive in the first two postnatal years, certain specific regions show unanimously linear increase in FC flexibility. They are especially clumping at the medial prefrontal cortex, indicating that these areas are more and more responding to high-level internal-external environment interaction and coordination for more adaptive behavior. In addition, some regions at the lateral side of the frontal lobe association areas show dramatic increasing FC flexibility in the first few months and saturate at later ages, suggesting a rapid early development in attention system, which might be essential for later development of high-level functional areas.Conclusion
Our study provides the first picture of developmental trajectories of brain functional connectivity dynamics during early infancy. We found that, in the first two postnatal years, human brain is gradually evolved to be more flexible and thus more adaptive to the fast-changing external world for supporting higher-level, more complex cognitive functions. This work fills the gap for early brain functional development studies and further paves the road for future works on normative and abnormal neurodevelopment.Acknowledgements
This study was supported in part by NIH grants (MH100217, MH108914, MH107815, and MH110274). The data was from “Multi-visit Advanced Pediatric brain imaging study for characterizing structural and functional development (MAP Study)”.References
[1] Gao, W., Alcauter, S., Smith, J.K., et al. Development of human brain cortical network architecture during infancy. Brain Structure and Function, 2015, 220, 1173-1186.
[2] Gao, W., Alcauter, S., Elton, A., et al. Functional network development during the first year: relative sequence and socioeconomic correlations. Cerebral Cortex, 2015, 25: 2919-2298.
[3] Wen, X.Y., Zhang, H., Shen, D.G., et al. First-Year Development of Modules and Hubs in Infant Brain Functional Networks, Neuroimage, 2018, In press.
[4] Allen EA, Damaraju E, Plis SM, et al. Tracking whole-brain connectivity dynamics in the resting state. Cerebral Cortex, 2014, 24: 663-676.
[5] Marusak, H.A., Calhoun, V.D., Brown, S., et al. Dynamic functional connectivity of neurocognitive networks in children, Human Brain Mapping, 2017, 38: 97-108.
[6] Shen, X., Tokoglu, F., Papademetris, X. and Constable, R.T. Groupwise whole-brain parcellation from resting-state fMRI data for network node identification. Neuroimage, 2013, 82: 403-415.
[7] Zhang, J., Cheng, W., Liu, Z., et al. Neural, electrophysiological and anatomical basis of brain-network variability and its characteristic changes in mental disorders. Brain, 2016, 139: 2307-2321.
[8] Yeo, B.T., Krienen, F.M., Sepulcre, J., The organization of the human cerebral cortex estimated by intrinsic functional connectivity. Journal of Neurophysiology, 2011, 106: 1125-1165.
Figures