0051
Optogenetic fMRI reveals ventral hippocampal modulatory effects on large-scale visual processing1Laboratory of Biomedical Imaging and Signal Processing, The University of Hong Kong, Hong Kong, China, 2Department of Electrical and Electronic Engineering, The University of Hong Kong, Hong Kong, China
Synopsis
Hippocampus has been traditionally associated to learning, memory, navigation and emotional behaviors. However, little is known regarding whether and how it influences the processing of large-scale visual sensory information. In this study, we combined optogenetic stimulation and visual fMRI to investigate the influence of the ventral hippocampus on visual processing across the central visual pathway, including superior colliculus, lateral geniculate nucleus and visual cortex. Our optogenetic fMRI results reveal for the first time the differential influences of high and low frequency ventral hippocampal activities on visual processing along the central visual pathway.
Purpose
The hippocampus (HP) is an essential brain structure that plays a prominent role in central nervous system functions such as learning1-3, memory4,5, sensory processing6 and spatial navigation7,8. In particular, HP has been shown to be critical for visual perception and memory such as spatial navigation and object recognition9,10. It is widely believed through anatomical and lesion studies11 that the functions of hippocampus are segregated along its dorsoventral axis with the dorsal HP (dHP) associated predominantly with cognitive functions and information processing, while the ventral HP (vHP) is associated with the regulation of stress and emotion12,13. However, it remains unclear whether such segregation of functions is absolute. Specifically, the question is whether vHP can influence sensory processing, such as visual processing. Here, we combined hippocampal optogenetic stimulation and large-scale fMRI detection to examine whether and how vHP modulates visual processing across the entire visual pathway, including the superior colliculus (SC), lateral geniculate nucleus (LGN) and visual cortex (VC).Method
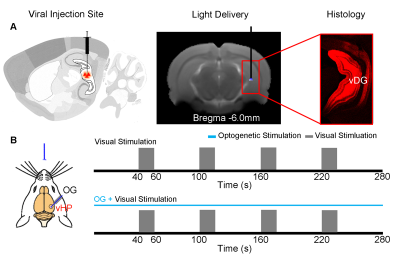
Animal Preparation: 3μl AAV5-CaMKIIα::ChR2(H134R)-mCherry was injected to the ventral dentate gyrus (vDG) in vHP of adult SD rats (200–250 g, male, N=5). After 4 weeks, an opaque optical fiber cannula (d=450μm) was implanted at the injection site as a means to deliver optical stimulation. All experiments were performed under 1.0–1.5% isoflurane.
Visual and Optogenetic Stimulation: To map out the responses upon vHP optogenetic stimulation, blue light was presented at 5Hz and 40Hz (30% duty cycle; 40mW/mm2) in a block design paradigm (20s light-on 60s light-off). To examine the effects of vHP optogenetic stimulation on visual processing, a combined visual and optogenetic stimulation approach was employed. Binocular visual stimulus was delivered via an optical fiber placed 3cm in front of both eyes, while vHP excitatory neurons were excited continuously at 5Hz/40Hz. 1Hz flashing light (0.5mW) was presented in a 20s ON 40s OFF block design. Visual fMRI trials with and without continuous optogenetic stimulation (5Hz or 40Hz) were interleaved. fMRI Acquisition and Analysis: fMRI data was acquired at 7T using GE-EPI (FOV = 32×32 mm2, matrix = 64×64, α=56°, TE/TR = 20/1000ms, and 10 slices with 1mm thickness and 0.2mm slice gap). Standard fMRI preprocessing was performed before GLM analysis was applied to identify significant BOLD responses (p<0.001). BOLD signal profiles were extracted from anatomically defined ROI.
Results
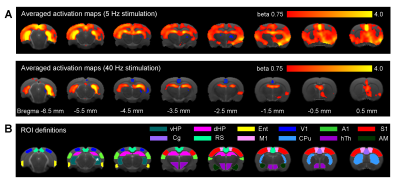
Figure 2 shows that 5Hz optogenetic excitation of vHP excitatory neurons evoked brain-wide positive BOLD activations in hippocampal formation (vHP; dHP; entorhinal cortex), cortical (visual; sensorimotor; cingulate; retrosplenial) and subcortical regions (caudate putamen; hypothalamus). Meanwhile, 40Hz optogenetic excitation vHP evoked only local positive BOLD activations within the hippocampus (vHP; dHP).
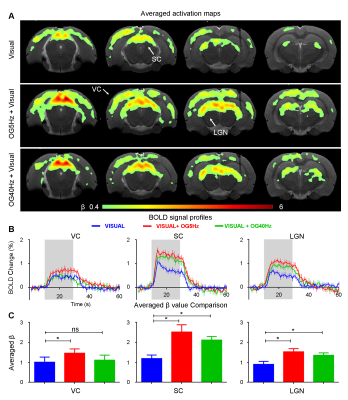
Figure 3 presents BOLD responses to binocular visual stimulation. As expected, visual stimulation evoked robust positive BOLD responses in VC, SC and LGN14. The BOLD responses to the visual stimulus at SC and LGN were significantly enhanced during continuous 5Hz and 40Hz optogenetic stimulation (P<0.05). Interestingly, an enhanced BOLD response in VC was only observed during continuous 5Hz optogenetic stimulation, but not during 40Hz stimulation.
Discussion & Conclusion
The present study demonstrated the role of hippocampal modulations on visual processing, specifically the vHP which was previously known to have weak reciprocal projections with the visual system. We showed that 5Hz optogenetic stimulation of ventral hippocampus (vHP) evoked brain-wide positive BOLD activations in hippocampal, cortical and subcortical regions. These cortical and subcortical activations were absent upon 40Hz optogenetic stimulation, while retaining strong activations in the hippocampus (i.e., vHP and dHP). It was found that the visual responses in SC and LGN were significantly increased during both continuous 5Hz and 40Hz vHP stimulation, whereas the enhanced VC response was only observed upon 5Hz, not 40Hz stimulation. These results indicate that different spatiotemporal characteristics of ventral hippocampal activities can modulate visual processing differently.
It has been previously reported that HP has a frequency-dependent spatiotemporal gradient across its dorsoventral axis, such that HP facilitates downstream brain-wide propagation at low-frequencies15. One of the key low frequency oscillations in HP is theta oscillations15, which has been shown to be pivotal for coordinating population of neurons to integrate sensory information and facilitate sensory processing16. In addition to low frequencies, studies also showed that theta-gamma coupling in HP can promote visual memory processing in the hippocampal-cortical network17,18. This suggests that high frequency hippocampal activities can also facilitate sensory processing, which was shown in our current findings. In summary, our study presents valuable insights into the large-scale roles of vHP on visual processing. We demonstrated that the spatiotemporally-specific downstream targeting of optogenetically-evoked vHP activities can differentially modulate visual processing along the central visual pathway.
Acknowledgements
This work was supported by the Hong Kong Research Grant Council (C7048-16G and HKU17103015 to E.X.W.).References
1. Moser, E., Moser, M. B., & Andersen, P. (1993). Spatial learning impairment parallels the magnitude of dorsal hippocampal lesions, but is hardly present following ventral lesions. Journal of Neuroscience, 13(9), 3916-3925.
2. Moser, M. B., Moser, E. I., Forrest, E., Andersen, P., & Morris, R. G. (1995). Spatial learning with a minislab in the dorsal hippocampus. Proceedings of the National Academy of Sciences, 92(21), 9697-9701.
3. Fanselow, M. S., & LeDoux, J. E. (1999). Why we think plasticity underlying Pavlovian fear conditioning occurs in the basolateral amygdala. Neuron, 23(2), 229-232.
4 Scoville, W. B., & Milner, B. (1957). Loss of recent memory after bilateral hippocampal lesions. Journal of neurology, neurosurgery, and psychiatry, 20(1), 11.
5. Squire, L. R. (1992). Memory and the hippocampus: a synthesis from findings with rats, monkeys, and humans. Psychological review, 99(2), 195.
6. Mohedano-Moriano, A., Martinez-Marcos, A., Pro-Sistiaga, P., Blaizot, X., Arroyo-Jimenez, M. M., Marcos, P., ... & Insausti, R. (2008). Convergence of unimodal and polymodal sensory input to the entorhinal cortex in the fascicularis monkey. Neuroscience, 151(1), 255-271.
7. Buzsáki, G., & Moser, E. I. (2013). Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nature neuroscience, 16(2), 130.
8. Maguire, E. A., Burgess, N., Donnett, J. G., Frackowiak, R. S., Frith, C. D., & O'keefe, J. (1998). Knowing where and getting there: a human navigation network. Science, 280(5365), 921-924.
9. Aly, M., Ranganath, C., & Yonelinas, A. P. (2013). Detecting changes in scenes: The hippocampus is critical for strength-based perception. Neuron, 78(6), 1127-1137.
10. Ji, D., & Wilson, M. A. (2007). Coordinated memory replay in the visual cortex and hippocampus during sleep. Nature neuroscience, 10(1), 100.
11. Strange, B. A., Witter, M. P., Lein, E. S., & Moser, E. I. (2014). Functional organization of the hippocampal longitudinal axis. Nature Reviews Neuroscience, 15(10), 655.
12. Fanselow, M. S., & Dong, H. W. (2010). Are the dorsal and ventral hippocampus functionally distinct structures?. Neuron, 65(1), 7-19.
13. Dong, H. W., Swanson, L. W., Chen, L., Fanselow, M. S., & Toga, A. W. (2009). Genomic–anatomic evidence for distinct functional domains in hippocampal field CA1. Proceedings of the National Academy of Sciences, 106(28), 11794-11799.
14. Chan, R.W., Leong, A.T., Ho, L.C., Gao, P.P., Wong, E.C., Dong, C.M., Wang, X., He, J., Chan, Y.S., Lim, L.W. & Wu, E.X. (2017). Low-frequency hippocampal–cortical activity drives brain-wide resting-state functional MRI connectivity. Proceedings of the National Academy of Sciences, 114(33), E6972-E6981.
15. Colgin, L. L. (2016). Rhythms of the hippocampal network. Nature Reviews Neuroscience, 17(4), 239.
16. Jutras, M. J., Fries, P., & Buffalo, E. A. (2013). Oscillatory activity in the monkey hippocampus during visual exploration and memory formation. Proceedings of the National Academy of Sciences, 110(32), 13144-13149.
17. Axmacher, N., Henseler, M. M., Jensen, O., Weinreich, I., Elger, C. E., & Fell, J. (2010). Cross-frequency coupling supports multi-item working memory in the human hippocampus. Proceedings of the National Academy of Sciences, 200911531.
18. Tamura, M., Spellman, T. J., Rosen, A. M., Gogos, J. A., & Gordon, J. A. (2017). Hippocampal-prefrontal theta-gamma coupling during performance of a spatial working memory task. Nature communications, 8(1), 2182.
Figures