5603
Magnetic Resonance Elastography Measures Predict White Matter Microstructure and AgingBoris Gutman1, Aaron T Anderson2,3, Nicolas Gallo4, and John G Georgiadis4
1Stevens Neuroimaging and Informatics institute, University of Southern California, Marina Del Rey, CA, United States, 2Mechanical Science & Engineering, University of Illinois at Urbana-Champaign, Urbana, IL, United States, 3Beckman Institute for Advanced Science and Technology, University of Illinois at Urbana-Champaign, Urbana, IL, United States, 4Biomedical Engineering, Illinois Institute of Technology, Chicago, IL, United States
Synopsis
We assessed the relationship between microstructural measures derived from diffusion tensor imaging (DTI) and shear moduli estimated from magnetic resonance elastography (MRE). We also compared the predictive power of fractional anisotropy, storage and loss moduli, estimation using nonlinear inversion (NLI) to model age in humans. Our results suggest some statistical relationship between MRE measures, particularly the loss modulus, and microstructure (FA, RD). The decrease in FA with age is accompanied by an increase in viscosity, especially in association fibers (Superior Longitudinal Fasciculus). The regional MRE measure provides more predictive power than the best regional FA.
Introduction
Viscoelastic properties of the brain can be measured indirectly in-vivo, with promising aging biomarker potential. It has been shown that brain tissue mechanics are affected by both healthy aging and dementia. However, to date little consensus exists regarding the direction and trajectory of such changes, due in part to underpowered studies and the variety of reconstruction (inversion) methods. In this study, we assess the relationship between DTI and MRE measures, as well as regional viscoelastic changes due to age.Methods
The experiment from a previous study5 was repeated on seven young (24-32 years old) and seven older (55-76 years old) males. The MR experiment included: high-resolution anatomical MPRAGE, diffusion tensor imaging (DTI), anterior-posterior (AP) excitation MRE, and left-right (LR) excitation MRE. The MRE displacement employed a 3D multislab, multishot spiral MRE sequence for generating 3D, full vector field complex displacement data at 50 Hz with 2 x 2 x 2 mm^3 isotropic spatial resolution7. For the anterior-posterior (AP) excitation, the soft head pad was placed at the posterior of the head, standard used in many previous studies8-10. For the left-right (LR) excitation, the liver pad was placed on the right side of the subject's head. Padding was placed between the subject's head and the side of the coil and between the liver pad and the coil for mechanical coupling and to reduce head movement. Nonlinear inversion (NLI) estimated heterogeneous, isotropic viscoelastic properties11. Additionally, matched field-of-view and resolution (relative to MRE) diffusion tensor imaging (DTI) and T1-weighted MPRAGE structural image at 0.9 x 0.9 x 0.9 mm3 isotropic resolution (TR/TI/TE = 2000/900/2.2 ms) were acquired. Using the ENIGMA-DTI protocol, we computed the study FA skeleton (TBSS) projecting onto it additional DTI and MRE measures: Radial Diffusivity, storage (elastic) and loss (viscous) moduli (SM, LM). Finally, all skeletonized measures were regionally averaged using the MNI JHU white matter (WM) atlas12 (Figure 1). We performed linear regression (a) within each region to model RD and FA as a function of LM and SM, and (b) using each regional measure to predict age group.Results & Discussion
FA and RD were correlated with SM at nominal significance (p < 0.05) in the internal capsule, cingulum. FA was additionally correlated with SM in the superior fronto-occipital fasciculus (SFO). FA and RD were correlated with LM at nominal significance in the external capsule and superior longitudinal fasciculus (SLF). FA was additionally correlated with LM in the cingulum and fornix. Only the FA-LM association in SLF passed false discovery rate correction. Figure 2 and Table 2 show all results. Age group differences reached nominal significance for FA in the corona radiata (anterior), cingulum, and SFO. Among the MRE measures, only LM reached p<0.05, with age-related increases in the corona radiate (posterior) and SLF. It is notable that although LM and FA are strongly associated in SLF, and SLF LM predicts age more accurately than any FA measure, FA in SLF is no significantly associated with age.Conclusions
We have presented a study on WM microstructure, mechanical tissue properties and aging. Our findings suggest that elasticity and viscosity may be strongly associated with white matter microstructure, particularly in long association fibers. Though some overlap exists between vicsous and elastic regional associations with FA and RD, it is clear that each component is independently correlated with microstructure. Further, we have shown that viscosity may be a better biomarker of aging than FA alone. This may be explained by the association between viscosity and the slowing of neuronal degeneration in aging.Acknowledgements
Support for ATA and JGG was provided by NSF Grant CMMI-1437113. Partial support was provided by the Biomedical Imaging Center of the Beckman Institute for Advanced Science and Technology at the University of Illinois at Urbana-Champaign (UIUC), NIH Grant R01-EB018230, and NIH/NIBIB Grant R01-EB001981. This research is part of the Blue Waters sustained-petascale computing project, which is supported by the National Science Foundation (awards OCI-0725070 and ACI-1238993) and the state of Illinois. Blue Waters is a joint effort of the University of Illinois at Urbana-Champaign and its National Center for Supercomputing Applications.References
[1] Arani, A., M. C. Murphy, et al. "Measuring the effects of aging and sex on regional brain stiffness with MR elastography in healthy older adults." Neuroimage. 2015;111: 59-64. [2] ElSheikh, M., A. Arani, A. et al. "MR Elastography Demonstrates Unique Regional Brain Stiffness Patterns in Dementias." American Journal of Roentgenology. 2017;209(2): 403-408. [3] Lamoureux PL, MR O'Toole, et al. Slowing of axonal regeneration is correlated with increased axonal viscosity during aging. Bmc Neuroscience 2010;11. [5] Anderson, A.T., Van Houten, E.E.W., et al. Observation of direction-dependent mechanical properties in the human brain with multi-excitation MR elastography. JMBBM. 2016;59, 538–546. [7] Johnson, C.L., Holtrop, J.L., et al. 3D multislab, multishot acquisition for fast, whole-brain MR elastography with high signal-to-noise efficiency. Magn. Reson. Med. 2014; 71, 477–485. [8] Murphy, M.C., Huston 3rd, J., et al. Measuring the Characteristic Topography of Brain Stiffness with Magnetic Resonance Elastography. PLoS ONE. 2013;8, e81668. [9] Schwarb, H., Johnson, C.L., et al. Medial temporal lobe viscoelasticity and relational memory performance. NeuroImage. 2016;132, 534–541. [10] Johnson, C.L., Schwarb, H., et al. Viscoelasticity of subcortical gray matter structures. Hum Brain Mapp. 2016;37, 4221–4233. [11] Van Houten, E.E.W., Paulsen, et al. An overlapping subzone technique for MR-based elastic property reconstruction. Magn. Reson. Med. 1999; 42, 779–786. [12] Hua, K., Zhang, J., et al., Tract probability maps in stereotaxic spaces: analysis of white matter anatomy and tract-specific quantification. NeuroImage. 2008;39(1):336-347.Figures
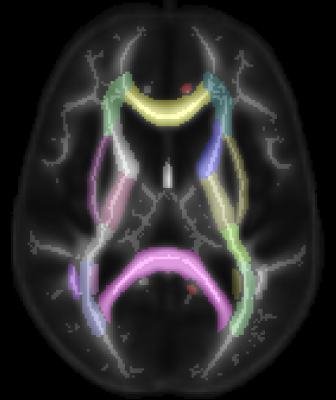
Figure 1: ENIGMA-DTI Atlas
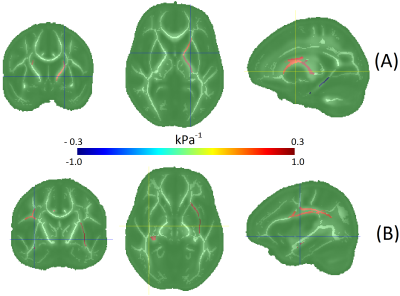
Figure 2: Regional fractional anisotropy associations with (A) storage
modulus - G', and (B) loss modulus – G''
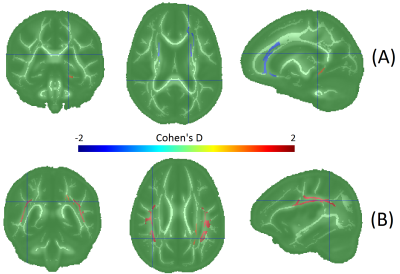
Figure 3: Age group differences in
regional DTI and MRE measures. Warm colors indicate higher values in the older
group, and cool colors indicate decrease with age.
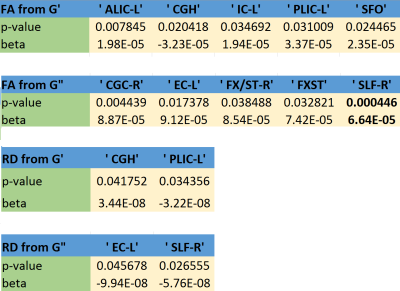
Table 1: FA and RD associations with G’ and G”. Legend:
ALIC-L: Anterior limb of internal capsule, left
PLIC-L: Posterior limb of internal capsule, left
CGH: Cingulum (hippocampus)
IC-L: internal capsule (whole), left
SFO: Superior fronto-occipital fasciculus
CGC-R: Cingulum (cingulate gyrus), right
EC-L: External capsule, left
FX/ST-R: Fornix cres/ Stria terminalis, right
SLF-R: Superior longitudinal fasciculus, right
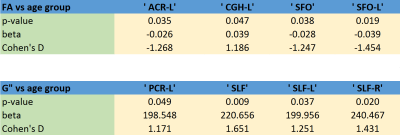
Table 2: FA and G” regional associations with age. Legend:
ACR-L: Anterior corona radiata, left
PCR-L: Posterior corona radiata, left
CGH: Cingulum (hippocampus)
SFO: Superior fronto-occipital fasciculus
SLF-(R/L): Superior longitudinal fasciculus, (right/left)