5323
Diffusion-weighted MRI of the prostate without susceptibility artifacts: Multi-shot STEAM with radial undersampling1Biomedizinische NMR Forschungs GmbH, Max-Planck-Institute for Biophysical Chemistry, Göttingen, Germany
Synopsis
Undersampled radial STEAM MRI allows for diffusion-weighed imaging without susceptibility artifacts. Here, this technique was developed for applications to the prostate by moving to a multi-shot acquisition and reconstruction method to optimally process data with limited SNR. Numerical simulations defined the conditions for accurate ADC measurements. In vivo studies of healthy subjects resulted in ADC values in the central gland of the prostate which are consistent and in agreement with previous literature values. A comparison of DW STEAM and DW EPI of the prostate confirmed the major benefit of STEAM sequences which are free of susceptibility-induced distortions.
Introduction
Susceptibility artifacts in diffusion-weighted (DW)
single-shot EPI of the prostate [1] may be avoided by DW single-shot
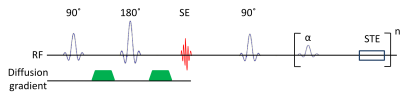
STEAM MRI sequences [2] as depicted in Fig. 1. Moreover, in
comparison to DW spin-echo sequences, the method does not suffer from a high radiofrequency
(rf) energy deposition or violations of the CPMG condition. Recently, a
combination of undersampled radial trajectories, iterative image reconstruction
with L2-regularization and denoising using modified non-local means (NLM) [3]
markedly improved the SNR in DW single-shot STEAM MRI of the brain [4]. An
extension to DW MRI of the prostate requires multiple averages for adequate SNR
when using external rf coils. This work presents a solution of the respective multi-shot
reconstruction problem for low-SNR acquisitions with inconsistent
motion-associated phases.
Methods
Multi-shot STEAM MRI with radial undersampling relies on the same sequence as described [4], but rotates the sampling trajectory in successive shots for optimal coverage of k-space. Each DW image is reconstructed from the data of all shots using a three-step method. First, coil sensitivities $$$C_{n}$$$ for the virtual channel $$$n=1,...,N$$$ and images $$$I_{m}$$$ for all shots $$$m=1,...,M$$$ are reconstructed by solving a nonlinear inverse problem [4]. Second, denoised phase maps $$$e^{iφ_{m}}=\frac{NLM(I_{m})}{|NLM(I_{m})|}$$$ are calculated from these images using a modified non-local means algorithm [3]. Third, the image content r is jointly estimated from all shots [5] by minimizing the functional $$$ \sum_{m,n} \|Y_{m,n}-P_{m} \mathcal{F} (C_{n} e^{iφ_{m}} r) \|^{2} + α \|r\|^{2}$$$ using nonlinear inversion, with the raw data $$$Y_{m,n}$$$, the projection onto the k-space trajectory $$$P_{m}$$$, and a regularization parameter $$$α$$$. Post-processing includes denoising with NLM.
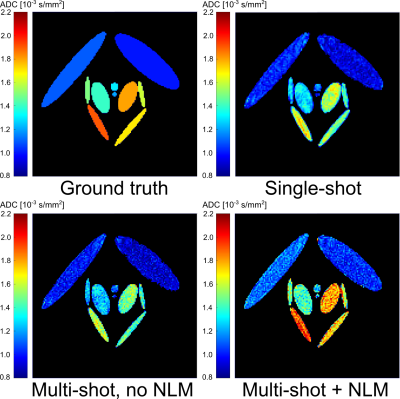
For validation, a numerical phantom of ellipses with pre-defined ADC values and a random phase for each ellipse and each diffusion weighting was used. After simulated radial sampling, white Gaussian noise was added to the raw data to achieve a pre-defined SNR per receive channel. Image reconstruction was performed using the proposed method ("multi-shot + NLM") and for comparison also with a multi-shot reconstruction without phase map denoising ("multi-shot, no NLM") and a single-shot reconstruction with magnitude averaging as for the brain [4].
Prostate studies employed the
following acquisition parameters: 1.4 x 1.4 mm2 resolution, 21
slices of 3.5 mm thickness, 200 mm FOV, 19 radial spokes per shot, 200 Hz pixel-1
bandwidth, three diffusion directions, 7 shots with b = 50 s mm-2,
17 shots with b = 600 s mm-2, 5 s repetition time , 6 min 25 s total
acquisition time. The clinical DW-EPI prostate protocol of the vendor served
for comparison using adjusted diffusion weightings, number of averages, and
repetition times to fit the STEAM protocol. In vivo MRI data were acquired at 3T
(Siemens Magnetom Prisma) with 80 mT m-1 gradients and a combination
of the 18-channel thorax coil and suitable elements of the spine coil.
Results and discussion
As shown in Fig. 2 for simulated data and the proposed
reconstruction method, a SNR per receive channel of 1.5 was sufficient to yield ADC maps in good agreement with the ground truth. The two
other tested versions resulted in systematic deviations (i.e., lower ADC values).
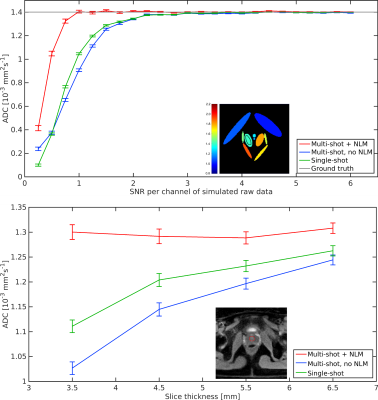
Quantitative comparisons in Fig. 3 revealed that the SNR per channel must be at
least 1 for accurate ADC measurements using the proposed method, whereas
multi-shot reconstructions without NLM and single-shot reconstructions require
an SNR per channel of 3. In vivo measurements in the central gland of the
prostate show an increase of the ADC with increasing slice thickness for the
two reference methods, whereas the proposed method is independent of the slice
thickness, while ADC values of $$$(1.30\pm0.02)\times10^{-3} mm^{2}s^{-1}$$$ are in agreement with reference values [6] of $$$(1.47\pm0.24)\times10^{-3} mm^{2}s^{-1}$$$. In vivo results for DW STEAM MRI and DW EPI are
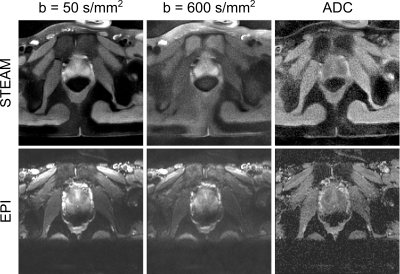
summarized in Fig. 4. STEAM MRI offers undistorted images of the human prostate,
where the boundary to the colon is well delineated in both DW images and ADC
maps. For EPI, a superior contrast-to-noise ratio is achieved, however, at the
expense of geometric distortions affecting the shape of the prostate and
particularly its lower boundary.
Conclusion and outlook
The present results demonstrate the feasibility of DW
multi-shot STEAM MRI of the
prostate. The numerical experiments set the ground for reliable and accurate measurements
of the ADC under conditions of limited SNR when using external rf coils. The
resulting ADC values of
healthy subjects are consistent and in agreement with previously published values.
Compared to DW-EPI, the STEAM MRI technique presents with lower
contrast-to-noise ratios, but greatly benefits from the absence of
susceptibility-induced distortions. Future developments include a model-based
reconstruction technique as well as clinical trials on patients.
Acknowledgements
No acknowledgement found.References
1. Li et al., Feasibility study of 3-T DWI of the prostate: readout-segmented versus single-shot echo-planar imaging. Am J Roentgenol 2015; 205:70-6.
2. Nolte et al., Rapid isotropic diffusion mapping without susceptibility artifacts: whole brain studies using diffusion-weighted single-shot STEAM MR imaging. Magn Res Med 2000; 44:731-6.
3. Klosowski and Frahm, Image denoising for real-time MRI. Magn Reson Med 2017; 77: 1340-5.
4. Merrem et al., Rapid diffusion-weighted imaging of the brain without susceptibility artifacts: single-shot STEAM with radial undersampling and iterative reconstruction. Invest Radiol 2017; 52:428-33.
5. Uecker et al., Inverse reconstruction method for segmented multishot diffusion-weighted MRI with multiple coils. Magn Reson Med 2009; 62: 342-8.
6. Emad-Eldin et al., Diffusion-weighted MR imaging and ADC measurement in normal prostate, benign prostatic hyperplasia and prostate carcinoma. Egypt J Radiol Nuc Med 2013; 44: 339-47.
Figures