4529
Comparison of fMRI and fNIRS mapping and systemic influences using a specifically designed multimodal probe for ultrahigh-resolution mapping1McLean Hospital, Harvard University, Belmont, MA, United States, 2Purdue, West Lafayette, IN, United States
Synopsis
fMRI is the gold standard of human functional brain imaging, although functional near-infrared spectroscopy (fNIRS) is easier to administer and lower in cost. Therefore studies have tried to compare the two techniques. However results have been variable, partly due to only rough spatial correspondence between fMRI and fNIRS, and systemic physiological noise influences. In the current study we use a previously described multimodal fNIRS/fMRI probe with optodes directly integrated into the coil structure. We characterize the spatial and temporal overlap between task-evoked fMRI and fNIRS signal, as well as their systemic physiological influences with high spatial and temporal correspondence.
Background
Functional near-infrared spectroscopy (fNIRS) has become increasingly popular for the study of brain function and has several advantages relative to fMRI, including ease of use and administration, and low cost. In addition, fNIRS measures both oxy- and deoxyhemoglobin independently. However the spatial resolution of fNIRS is inferior, and putative neuronal signals are contaminated by the blood signals from extra-cerebral layers1,2, leaving fMRI as the gold standard of in vivo human brain imaging. It is therefore unsurprising that both modalities are often used together to help guide one another. However results of these studies have been variable as the fMRI and fNIRS data are usually acquired sequentially rather than simultaneously. Other sources of variation may include the difficulty establishing the spatial correspondence between fMRI and fNIRS, as well as systemic physiological noise3.
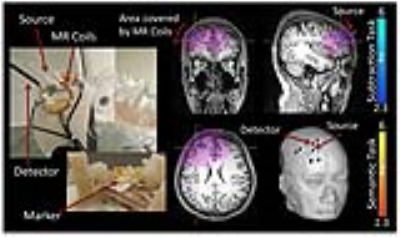
We have previously reported on the development of a special multimodal fNIRS/fMRI probe4. fNIRS source and detector fibers were integrated into the construction of a receive-only phased array RF coil on a curved surface, enabling close contact between the probe and the head (see Figure 1, left side), and precise alignment between the modalities. This coil can acquire high spatial resolution fMRI images (1.2x1.2x1.8mm) at high speed (2.5Hz), while simultaneously recording fNIRS signals from the same area. We have now acquired multimodal data from 10 subjects with this probe.
Purpose
Evaluate spatial and amplitude correspondence of fNIRS and fMRI with high precision equipment and determine the influence of systemic signals in both modalities.Target Audience
Researchers interested in multimodal imaging as well as clinicians interested in the validation of bed-side techniques. In addition this research will be of interest for those quantifying influence of systemic signals on functional hemodynamic measurements.Methods
10 subjects were measured with our specifically designed multimodal fMRI/fNIRS probe4. Measurements with the multimodal probe on the Siemens Trio 3T (fMRI) and the ISS Imagent (fNIRS) were acquired during resting state as well as during 15 minute long mental subtraction and rapid semantic tasks. The 3D printed probe features 3 overlapping circular receive only RF-coils, closely formed to the right frontal area of approximately 8x4.5cm with depth sensitivity of ~5cm. Integrated fNIRS (6.25Hz) probes for deep (3-4cm source-detector distance) and shallow measurements (1cm source-detector distance) were integrated into the design. For comparison, peripheral measurements were also taken with fNIRS. Two types of fMRI acquisition were performed; a high spatial resolution acquisition on a broad field of view with 1.5s TR, and 5 consecutive stacked data sets (1.8mm x 5 for each stack) with a TR of 0.4s acquired using our previously described technique5. Outer volume suppression was used to limit the field of view. Data analysis was conducted in MATLAB (for fNIRS) and FSL (for fMRI). FSL (FEAT) and in house Python software (RapidTiDe)6 were used for localization of the focal activation detected by fNIRS as well as fMRI.Discussion
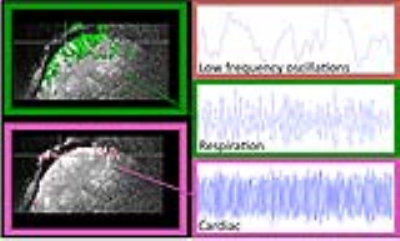
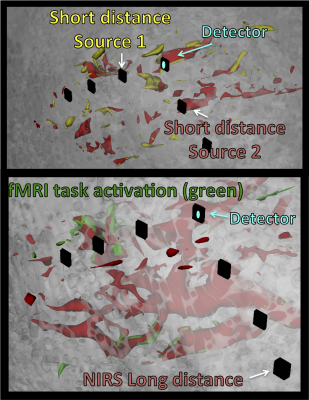
This dataset offers dense information on fNIRS and fMRI. The probe covered the expected activation areas for both tasks. Clear activation patterns below the probe for fNIRS and fMRI are depicted (see Figure 1, right side). The contribution of known low frequency systemic signals in fMRI could be isolated and their influence could be evaluated (Figure 2) and compared with the signal from fNIRS. For fNIRS the high resolution multimodal acquisition allowed us to differentiate signals from the skin, skull, pial vessels and brain. Short and long distance measurements of fNIRS showed high involvement of the vasculature especially directly below the probes (Figure 3).Acknowledgements
No acknowledgement found.References
1 Gagnon, L. et al. Improved recovery of the hemodynamic response in diffuse optical imaging using short optode separations and state-space modeling. Neuroimage 56, 1362-1371, doi:10.1016/j.neuroimage.2011.03.001 (2011).
2 Kirilina, E. et al. Identifying and quantifying main components of physiological noise in functional near infrared spectroscopy on the prefrontal cortex. Front Hum Neurosci 7, 864, doi:10.3389/fnhum.2013.00864 (2013).
3 Funane, T. et al. Concurrent fNIRS-fMRI measurement to validate a method for separating deep and shallow fNIRS signals by using multidistance optodes. Neurophotonics 2, 015003, doi:10.1117/1.NPh.2.1.015003 (2015).
4 Hocke, L. M., Cayetano, K., Tong, Y. & Frederick, B. Optimized multimodal functional magnetic resonance imaging/near-infrared spectroscopy probe for ultrahigh-resolution mapping. Neurophotonics 2, 045004, doi:10.1117/1.NPh.2.4.045004 (2015).
5 Tong, Y. & Frederick, B. Concurrent fNIRS and fMRI processing allows independent visualization of the propagation of pressure waves and bulk blood flow in the cerebral vasculature. Neuroimage 61, 1419-1427, doi:10.1016/j.neuroimage.2012.03.009 (2012).
6 Frederick, B., Nickerson, L. D. & Tong, Y. Physiological denoising of BOLD fMRI data using Regressor Interpolation at Progressive Time Delays (RIPTiDe) processing of concurrent fMRI and near-infrared spectroscopy (NIRS). Neuroimage 60, 1913-1923, doi:10.1016/j.neuroimage.2012.01.140 (2012).
7 Tong, Y., Lindsey, K. & Frederick, B. B. Partitioning of physiological noise signals in the brain using concurrent near-infrared spectroscopy (NIRS) and fMRI (Quebec, Canada, 2011).
Figures