4194
Mapping Gadolinium Entry and Clearance from the Brain using Dynamic MRI1Centre for Advanced Biomedical Imaging, Division of Medicine, University College London, London, United Kingdom
Synopsis
Recently, evidence for gadolinium retention in the brain was provided, raising concerns regarding the safety of these agents. This accumulation raised important questions, most crucially, by which pathway does gadolinium enter the brain. We provide insight into this by dynamic monitoring of the infiltration and clearance of gadolinium into the brain parenchyma following an IV injection. Following gadolinium injection, inflow of contrast into the cerebrospinal fluid spaces was prominent. 25mins post injection, contrast enhancement is detected in the periventricular regions and hypothalamus. This finding provides insight into the entry routes by which gadolinium infiltrates the brain following a systemic administration.
Introduction
Recent evidence for the retention of gadolinium (Gd) based contrast agents in the brain following repeated IV delivery, has led to safety concerns.1 As yet, it is unclear as to the route of ingress and clearance from the brain. We aim to address this question by visualizing the infiltration of Gd into the brain parenchyma following systemic (IV) injection. This method may also enable the assessment of rates of interstitial solute clearance, and thus represents a novel and minimally invasive measure of glymphatic function, which has been proposed as a key pathway for waste removal, and may be important in the aetiology of many prominent neuro-degenerative diseases.2Methods
Data reported here were acquired from 6 healthy C57/B6 mice (2-3 months of age) using a 9.4T Agilent MRI scanner (anesthesia: isoflurane in air/O2). A 3D T1-weighted gradient echo sequence was employed with parameters: TR = 15ms, TE = 3.4ms, flip angle = 15°, NA = 3, FOV = 1.5 × 1.5 × 2.0cm, acquisition time = 12.5 min, matrix size: 128 × 128 × 128, image resolution: 0.12 × 0.12 × 0.15mm. The baseline image was followed by an intravenous injection of a high dose bolus (10mmol/Kg) of Gadodiamide (Omniscan, GE Healthcare).Results
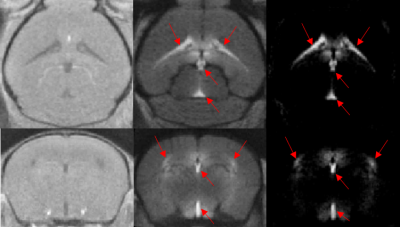
Following IV delivery of Gd, inflow of contrast agent into the cerebrospinal fluid (CSF) spaces, notably the ventricular system, was prominent (fig1-red arrows). This demonstrates the ability of this agent to cross the blood-CSF barrier and suggests that entry to the brain parenchyma may be via the CSF.
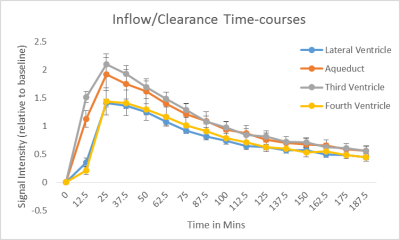
In order to quantify the inflow and clearance of the gadolinium from the CSF, time-intensity curves from manually drawn regions of interest (ROIs) were obtained (fig2).
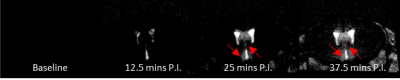
Infiltration of Gd was observed into parenchymal regions immediately surrounding the ventricles, with contrast enhancement in the periventricular regions and hypothalamus 25 minutes post injection (fig3). This finding provides insight into the entry routes by which gadolinium infiltrates the brain following a systemic administration.
Discussion
A previous report noted hyper intensity on T1-weighted images (unenhanced) in the dentate nucleus and globus pallidus regions in patients who received repeated doses of linear gadolinium based contrast agents which raised safety concerns. The mechanism of contrast accumulation in these brain regions could not be established, mainly due to the fact that the experiments were performed at a single time-point where the contrast agent had already been deposited in the brain. In this study, the infiltration and clearance of gadolinium into the mouse brain was dynamically monitored following a systemic injection. We provide evidence that the primary route of infiltration of contrast agent into the brain is via the blood-CSF barrier. We further demonstrate, for the first time, that gadolinium appears to infiltrate the brain parenchyma immediately proximal to the ventricles. By monitoring the washout of Gd from the tissue, this methodology may also represent a useful tool to image glymphatic clearance and its role in neuro-degenerative conditions such as Alzheimer's disease, non-invasively.Conclusion
We have captured the dynamic inflow of contrast agent from the blood to brain for the first time, using a minimally invasive approach. Initial analysis suggests that Omniscan appears to accumulate in the CSF spaces before moving into periventricular regions. Work is ongoing to fully characterize this pathway across all brain regions.Acknowledgements
This work is supported by the EPSRC-funded UCL Centre for Doctoral Training in Medical Imaging (EP/L016478/1) and the Department of Health’s NIHR-funded Biomedical Research Centre at University College London Hospitals.References
1- Kanda T, et al. High Signal Intensity in the Dentate Nucleus and Globus Pallidus on Unenhanced T1-weighted MR Images: Relationship with Increasing Cumulative Dose of a Gadolinium-based Contrast Material. Radiology 2014 270:3, 834-841
2- Iliff J. J, et al. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid beta. Science translational medicine 4, 147ra111, doi:10.1126/scitranslmed.3003748 (2012).
Figures