4183
The impact of pad thickness on RF burn prevention: is a 1mm pad sufficient?1Toshiba Medical Research Institute USA, Mayfield Village, OH, United States
Synopsis
RF burns are typically categorized according to appearance and suspected mechanism. Skin/skin contact burns are postulated to be caused by significant current flows through a restricted contact area. While not definitively proven, patients are instructed to not clasp hands and pads should be routinely used to minimize skin/skin contact. The effectiveness of a thin pad is investigated with EM modeling. A reported calf burn (peak 10g SAR:whole body SAR ratio ~86) may be avoidable with a 1 mm air gap (SAR hotspot ratio ~2). Understanding that thin pads are sufficient may permit greater flexibility in possible pad solutions.
Introduction
The continued investigation of patient RF burn mechanisms and the development of prophylactic solutions is necessary because the mechanisms are not fully understood thereby hindering the development of countermeasures. It is theorized that skin/skin contact burns are the result of significant current flows across a restricted contact area resulting in ohmic heating. Pads are universally recommended, but perhaps not deployed frequently enough, to disrupt points of possible skin/skin contact. This abstract investigates the minimum gap spacing required to reduce the local SAR to appropriate levels. By clarifying what constitutes a sufficiently thick pad, greater flexibility of solutions may result in more frequent use of pads.Methods
Three plausible clinical situations were investigated by numerical EM modeling with software package Sim4Life (ZMT, Zurich, Switzerland):
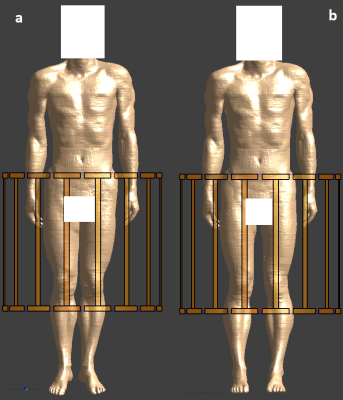
- A very high local SAR hotspot clinical configuration (e.g. touching calves. Figure 1a)
- A passive solution (e.g. legs spread. Figure 1b)
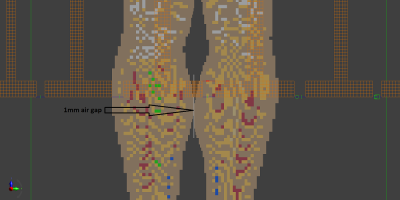
- Thin separation pad (e.g. 1 mm air separation. Figure 2).
Posable adult male model Duke1 (ZMT, Zurich, Switzerland) was positioned in a generic whole body Tx coil based on ISO/TS109742 (16-rung high-pass birdcage, diameter 750mm, length 650mm. RF shield diameter 790mm, length 850mm.3,4). The coil was tuned to 64MHz and driven with two voltage sources in quadrature mode. The hotspot condition modeled is based on a reported 1.5T burn case.5 The model was configured to faithfully reproduce the reported clinical condition, but the specifics of the transmit coil are unknown. No attempt was made to maximum the SAR hotspot ratio by repositioning Duke. Touching calves are clinically possible, but not necessarily a comfortable position for the patient. The passive “legs spread” solution may not always be available, depending on the clinical imaging case, patient size, receive coil used etc., nor necessarily comfortable for the patient. Furthermore, “legs spread” may only move the point of skin/skin contact and not completely break the contact. Whether or not comfortable, a pad will interrupt the current flow between the calves, but may not necessarily significantly reduce the E-fields when the calves are in close proximity.
Results
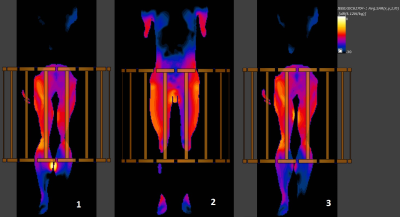
The clinically faithful peak 10g : whole body (WB) SAR hotspot ratio with calves touching (case #1) is 86.4 (peak 10g: 8.12 W/kg, whole body: 0.094 W/kg, nominal B1+ field averaged in the central axial slice: 1.1 µT) and is significantly higher than typical clinical hotspot ratios, which can be over 20. Global peak 10g : WB SAR and local 10g (at calves) : WB SAR hotspot ratios for all three leg configurations are shown in Table 1. Figure 3 shows 10g SAR distribution in coronal slices. Global peak 10g SAR values are located in the slices for case #1 and #2. The slice shown for case #3 is not the one with global peak SAR. Instead, it is the same slice as case #1 to show the absence of hotspot at calves once 1mm air gap is inserted.Discussion
It is not necessary to use thick pads, which may be uncomfortable for the patient or too bulky, to reduce the SAR hotspot ratio to a safe level otherwise routinely seen in clinical imaging. However, if a thin pad was used it is also possible that it could rapidly become soaked in sweat, in which case the protection could be transitory.Conclusion
Pads as thin as 1 mm are likely sufficient to stop skin/skin contact burns. This recommendation is likely not applicable to other RF burn mechanisms such as skin/bore liner contact.Acknowledgements
No acknowledgement found.References
- Gosselin MC, Neufeld E, Moser H, et al. Development of a new generation of high-resolution anatomical models for medical device evaluation: the Virtual Population 3.0. Physics in Medicine and Biology. 2014; 59(18): 5287-5303.
- ISO/TS 10974:2012 Assessment of the safety of magnetic resonance imaging for patients with an active implantable medical device. Geneva, Switzerland: International Organization for Standardization, 2012.
- Murbach M, Neufeld E, Kainz W, et al. Whole-body and local RF absorption in human models as a function of anatomy and position within 1.5T MR body coil. Magn. Reson. Med. 2014; 71(2):839-845.
- Murbach M, Cabot E, Neufeld E, et al. Local SAR enhancements in anatomically correct children and adult models as a function of position within 1.5T MR body coil. Prog Biophys Mol Biol. 2011; 107(3):428-433.
- Knopp MV, Essig M, Debus J, et al. Unusual burns of the lower extremities caused by a closed conducting loop in a patient at MR imaging. Radiology. 1996 ;200(2):572-575.
Figures