3589
High Temporal Resolution Dynamic BOLD MRI in Tracking Kidney Response to Hypoxia and Hyperoxia in a Rat AKI model at 7T1School of Biomedical Engineering, Guangdong Provincial Key Laborary of Medical Image Processing, Southern Medical University, Guang Zhou, China, 2Philips Healthcare, Guang Zhou, China
Synopsis
Blood oxygenation level-dependent magnetic resonance imaging has shown potential to monitoring the tissue oxygenation. However, the absolute T2* value has shown limited usefulness to surrogate the renal microcirculation. This work we monitor the AKI rat renal response to hypoxia and hyperoxia to tracking the hemodynamic changes in the progress of acute kidney injury.
Purpose
To evaluate AKI rat renal real-time microcirculation through the renal response to hypoxia and hyperoxia monitored by dynamic BOLD MRI.Introduction
Renal hypoxia and hypoperfusion play an important role in the development of acute kidney injury (AKI)1. Early detection of renal hemodynamic change may contribute to early diagnosis and prevent further deterioration of AKI. Blood oxygenation level-dependent magnetic resonance imaging (BOLD-MRI) provides an opportunity to noninvasively monitor the tissue oxygenation changes by the endogenous contrast deoxy-hemoglobin2,3. However, the absolute T2* value has shown limitation to surrogate the tissue oxygenation due to the kidney water content, vascular volume etc4. In this work, we monitored the renal hemodynamic change response to hypoxia and hyperoxia using high temporal resolution dynamic BOLD-MRI to reflect the renal microcirculation in a rat acute kidney injury (AKI)modelMethods
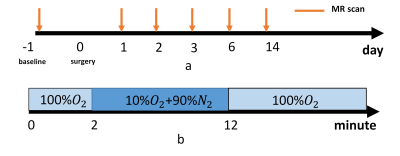
The study was approved by the local Ethics Review Board. AKI was induced by renal pedicle clamping of unilateral kidney for 45min. All the MR experiments were performed on a 7T small animal scanner (PharmScan, Bruker, Germany). 3 female adult SD rat (weight: 230~250g) underwent MR scan at day0, day1, day2, day3, day6 and day14 after renal pedicle clamping (Fig1.a).
During the experiments, the animals are anesthetized on 5% isoflurane in room air (1L/min) in an induction chamber, and then intubated with a 14G-angiocatheter on experiment table. The animals anesthetization was maintained with 3% isoflurane(1L/min) for by endotracheal tube and their respiration was controlled by ventilator and kept constant 65 breaths per minute during the entire examination. A water blanket was used to maintain the body temperature at 37 and the oximeter sensor was clipped on the hand paw to monitor the oxygen saturation pressure.
The gas challenge started with 2min hyperoxia(100% oxygen) followed by 10min hypoxia (10% oxygen) and end with hyperoxia(100% oxygen) (Fig 1.b)
Before gas challenge, a low resolution T2WI was performed for drawing ROI on hole inner stripe of outer medullar and the parameters were: TR=4000ms, TE=58.9ms, resolution = 0.5*0.5*1mm3. High temporal resolution BOLD-MRI was implemented by using multi-gradient echo sequence with following parameters: TR=40ms, TE/=2/4ms, 8 echo, flip angle = 15, single slice, resolution = 0.5*0.5*1mm3, dummy scan = 400, dynamic time= 4s400ms, actual acquisition time 9.6s(triggered)..
Results
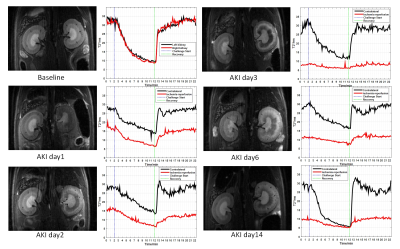
Figure 2 shows the BOLD curve of different time points.T2* value first significantly decreased on day1,day2 and day3,then slightly increased on day6, day14. BOLD curve of injured kidney took longer time to recover to stable level than contralateral kidney after hypoxia on day1 and day2. But on day3-14, the BOLD curve showed little response to gas challenge.Discussion&Conclusion
This study used a high temporal resolution BOLD MRI to monitor renal response to hypoxia/hyperoxia in rat AKI model. In our preliminary work, different characteristics of dynamic BOLD curve injured kidney VS contralateral kidney, and the difference gas response at different time point after surgery were observed .Acknowledgements
noReferences
1. Ali T, Khan I Fau - Simpson W, Simpson W Fau - Prescott G, Prescott G Fau - Townend J, Townend J Fau - Smith W, Smith W Fau - Macleod A, Macleod A. Incidence and outcomes in acute kidney injury: a comprehensive population-based study. (1046-6673 (Print)).
2. Ogawa S, Lee Tm Fau - Kay AR, Kay Ar Fau - Tank DW, Tank DW. Brain magnetic resonance imaging with contrast dependent on blood oxygenation. (0027-8424 (Print)).
3. Ogawa S. Finding the BOLD effect in brain images. (1095-9572 (Electronic)).
4. Niendorf T, Pohlmann A Fau - Arakelyan K, Arakelyan K Fau - Flemming B, Flemming B Fau - Cantow K, Cantow K Fau - Hentschel J, Hentschel J Fau - Grosenick D, Grosenick D Fau - Ladwig M, Ladwig M Fau - Reimann H, Reimann H Fau - Klix S, Klix S Fau - Waiczies S, Waiczies S Fau - Seeliger E, Seeliger E. How bold is blood oxygenation level-dependent (BOLD) magnetic resonance imaging of the kidney? Opportunities, challenges and future directions. (1748-1716 (Electronic)).