2654
Motion correction of PET images using Spherical Navigator echoes (SNAVs) on a hybrid PET-MR scanner1Robarts Research Institute, London, ON, Canada, 2Medical Biophysics, Western University, London, ON, Canada, 3Lawson Health Research Institute, London, ON, Canada, 4Siemens Canada, Oakville, ON, Canada
Synopsis
Head motion during brain imaging with hybrid PET-MR degrades the quality of both the PET and MR images. Simultaneous acquisition with the two modalities provides the opportunity for MR motion measurement techniques to be used for correction of PET data. In this study, spherical navigator echoes (SNAV) were used for retrospective motion correction of PET images. A phantom was repositioned several times during a list mode acquisition. The list mode data was binned into motion states based on the SNAV measured motion, and a motion-corrected PET reconstruction was performed. SNAV motion correction successfully removed blurring in the PET images.
Introduction
Head motion during brain imaging with hybrid PET-MR may cause artefacts and blurring in both the PET and MR images. The hybrid imaging system provides the opportunity for MR motion measurement techniques to be used for motion correction of the PET data. MR motion correction techniques have been successfully applied in PET brain imaging,1,2 but these techniques use the MR scanner exclusively for motion measurement and do not acquire diagnostic MR images simultaneously.
Spherical navigator echoes (SNAV) – a 3D k-space navigator-based motion measurement technique – can be interleaved within brain MRI sequences to enable simultaneous motion corrected PET and MRI. The SNAV technique can measure brain rotations and translations with sub-millimeter and sub-degree accuracies and has been applied successfully for retrospective correction of MR brain images.3 In this work we demonstrate successful retrospective motion correction of PET images using SNAVs.
Methods
SNAV pulse sequence: The SNAV pulse sequence is a modified FLASH sequence where the Cartesian gradient waveforms are replaced by the gradient waveforms required for the SNAV’s spherical trajectory. The SNAV is a spherical shell in k-space with a radius of 0.4cm-1. It is acquired in two shots – one for each hemisphere – with a TR of 15 ms.
Phantom experiment: The experiment was performed on a Siemens 3T Biograph mMR with a 4-channel head coil. Eight 9 – 12 mm diameter vials were inserted into a pineapple (Fig. 1), known to be a suitable phantom for MR navigators, and filled with FDG. Each vial contained between 1.5 and 2 ml of FDG (approximately 20kBq/mL). A 7 min reference PET scan was acquired, while the phantom was stationary, followed by 7 min scans during which the phantom was manually repositioned three times. For each acquisition, SNAV data were acquired simultaneous with the PET list mode data.
Motion measurement & correction: SNAVs were processed in Matlab; the rotations and translations were determined by comparing SNAVs acquired during the PET scan to those acquired in a baseline scan. A detailed description of this method can be found in Johnson et al. 2016.3 The measured motion profile was used to sort the list-mode data into four motion states. RTA reconstruction4 was then performed in which each bin was reconstructed using the Ordinary Poisson (OP) algorithm. The four reconstructed images were finally transformed based on the measured motion and summed to form a final motion corrected image.
Results
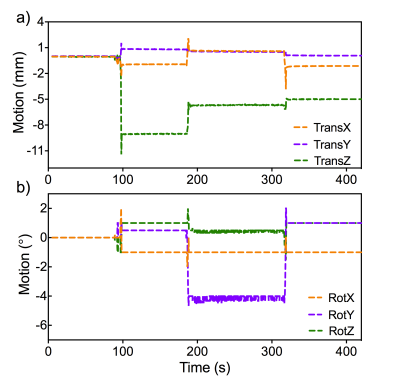
An example SNAV-derived motion profile is shown in Fig. 1. Translations up to 11 mm and rotations up to 4° were measured. Coronal slices of the reference, uncorrected and corrected images are shown in Fig. 3. The motion-related distortion is clearly seen in the uncorrected image (Fig. 3b). The SNAV-corrected image (Fig. 3c) closely resembles the reference (static) image and the expected circular cross-sections are restored. Quantitatively, motion correction using the SNAVS resulted in a reduction of the full width half max (Fig. 3d) from 11.5 mm in the uncorrected image to 9 mm in the corrected image, compared to 9 mm in the reference image.Discussion
Six-degree-of freedom motion correction using SNAVs successfully removed blurring due to motion from the PET images. SNAVs can be integrated within an MR imaging sequence in order to perform motion correction of the MR data in addition to the PET data. We have shown previously that motion correction of in-vivo MR brain images is possible with an SNAV navigated SPGR sequence.3 We have recently incorporated SNAVs into the turbo-flash sequence, this navigated image sequence is expected to provide significant improvement to the diagnostic quality of PET images, while acquiring diagnostic (motion corrected) MR images simultaneously. Prospective MR motion correction – the focus of ongoing work – is expected to improve the motion correction of MR images by eliminating the need for interpolation of image data.Acknowledgements
No acknowledgement found.References
(1) Catana C, et al. MRI-assisted PET motion correction for neurologic studies in an integrated MR-PET scanner. J Nucl Med 2011
(2) Huang C, et al. Motion compensation for brain PET imaging using wireless MR active markers in simultaneous PET-MR: phantom and non-human primate studies. Neuroimage 2014
(1) Johnson PM, et al. Retrospective 3D motion correction using spherical navigator echoes. Magn Reson Imaging 2016
(4) Picard Y, Thompson CJ. Motion correction of PET images using multiple acquisition frames. IEEE Trans Med Imag 1997
Figures

Pineapple phantom with vials filled with FDG (20kBq/mL)