2426
Kurtosis as a potential tool to differentiate breast hematological malignancies from breast cancer1Department of Diagnostic Imaging and Nuclear Medicine, Graduate School of Medicine, Kyoto University, Kyoto, Japan, 2Department of Hematology and Oncology, Graduate School of Medicine, Kyoto University, Kyoto, Japan, 3Department of Diagnostic Pathology, Graduate School of Medicine, Kyoto University, Kyoto, Japan, 4Department of Breast Surgery, Graduate School of Medicine, Kyoto University, Kyoto, Japan
Synopsis
Since breast hematological malignancies show various image findings, it is not easy to differentiate them from breast cancer using conventional MRI. Non-Gaussian diffusion MRI is a relatively new method using multi b values from low to high, reflecting the interaction of water molecules with tissue features. We compared non-Gaussian parameters of breast hematological malignancies and breast cancer to investigate the advantage of non-Gaussian diffusion imaging. Our preliminary results suggest potential advantage of kurtosis as a marker of cellular structure and usefulness in differential diagnosis between breast hematological malignancies and breast cancer.
Introduction
Although breast hematological malignancies (lymphoma and leukemia) and invasive breast cancer sometimes show similar MR image findings1, hematological malignancies have different biological and histological characteristics from breast cancer and the management strategy is considerably different. Thus, the accurate imaging diagnosis of hematological malignancies prior to biopsies warranted for appropriate management and early systemic evaluation for staging. Dynamic contrast-enhanced MRI (DCE-MRI) findings were various and the considerable overlap of apparent diffusion coefficient (ADC) values between breast hematological malignancies and breast cancer has been reported in previous studies1-3. Hence combination of DCE-MRI and diffusion weighted image (DWI) is not sufficient enough to provide accurate differential diagnosis. Non-Gaussian diffusion MRI can provide detailed information, such as tissue microstructure beyond ADC. One can quantify the degree of non-Gaussian diffusion of water molecules with Kurtosis model, and it is supposed to reflect the interaction of water molecules with tissue features, especially cell membranes4. Hematological malignancies possess high cellular density, with characteristic small round cell in lymphoma. Since such information on tissue microstructure would be helpful for diagnosis, we hypothesized that non-Gaussian diffusion parameters, particularly kurtosis, could be useful for differential diagnosis. Thus, the purpose of this study was to investigate the advantage of non-Gaussian diffusion imaging for differential diagnosis between breast hematological malignancies and breast cancer.Materials and methods
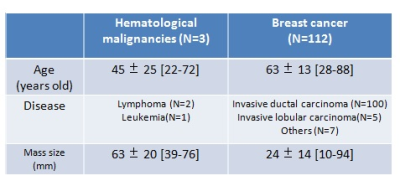
Patients 115 female patients were enrolled between May 2013 and March 2015. All of them had breast hematological malignancies or breast cancer, which were diagnosed by histopathologic analysis on initial biopsy. All patients had breast mass larger than 10mm on MR images, where we defined region of interest (ROI). The characteristics of patients and mass lesions are summarized in Table 1. DWI image acquisition protocol and estimated non-Gaussian parameters Each patient underwent a bilateral breast MR imaging with a 3T MR machine (Tim Trio; Siemens Healthcare, Erlangen, Germany) using a 16-channel breast array coil. DW images were acquired with 16 b values ranging from 5 to 2500 sec/mm2. As for non-Gaussian diffusion parameters, theoretical ADC at b value of 0 sec/mm2 (ADC0) and kurtosis (K) were estimated from DWI images with multi b values, using the kurtosis model5. The following formula was used to estimate ADC0 and K. S/S0 = {exp [-2b ADC0 + K (b ADC0)2/3] + NCF }1/2 Here S0 is the theoretical signal acquired at b=0 and NCF (noise correction factor) is a parameter which characterizes the intrinsic non-Gaussian noise contribution within images. A synthetic ADC (sADC) was also calculated by using b values of 200 and 1500 sec/mm2 (sADC200-1500), as well as standard ADC by using b values of 0 and 800 sec/mm2 (ADC0-800). Image analysis Firstly, ROIs were placed on the largest mass for each patient. Then, non-Gaussian parameters (ADC0 and K), sADC200-1500 and ADC0-800 were calculated as mean value in the ROIs. Then, those values of hematological malignancies were compared with those of breast cancer. The parametric maps were also generated for each ROI. All the analysis were performed using MATLAB (MathWorks, Natick, MA, USA).Results
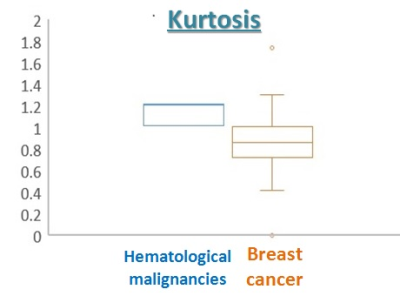
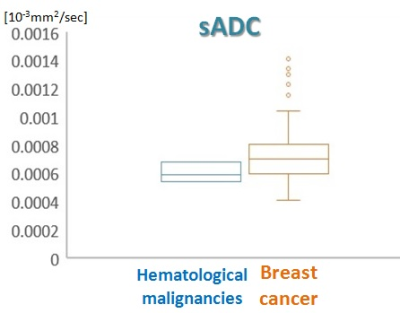
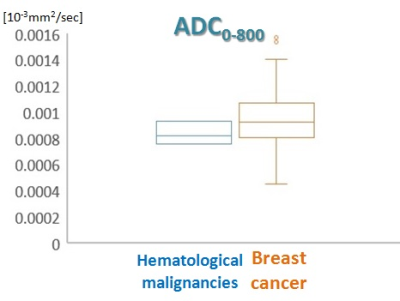
The parametric maps of breast mass (K, ADC0) of lymphoma are shown in Figure 1, showing moderately homogeneous pattern. The peripheral area of the tumor shows high K and low ADC0 values reflecting high cellularity, while the central area shows relatively low K and high ADC0 values reflecting comparative low cellularity. Taken together, it suggests heterogeneity of the lesion. The mean value of K was slightly higher in hematological malignancies than that in breast cancer (Figure 2). The mean values of sADC200-1500 and ADC0-800 were slightly lower in hematological malignancies than in breast cancer, although the overlap was observed between both groups (Figure 3, 4).Discussion
These results agree with our hypothesis that non-Gaussian diffusion parameters, particularly kurtosis could be helpful for differential diagnosis between breast hematological malignancies and breast cancer. Because non-Gaussian diffusion MRI can reflect microstructure of the lesions, K is considered to reflect high cellular density characteristic to tumor cells in breast hematological malignancies. K is also considered to reflect structural complexity, such as proliferation6. Considering overlap between both groups, combining it with other parameters which reflect other characteristic such as homogeneity over the entire lesion may lead to increasing diagnostic accuracy of MRI. Although the data size is limited due to the rarity of breast hematological malignancies, our preliminary results suggest potential advantage of kurtosis as a marker of cellular structure and usefulness in differential diagnosis between breast hematological malignancies and breast cancer.Conclusion
Non-Gaussian diffusion parameter, kurtosis (K) can be a helpful index for differential diagnosis between hematological malignancies in breast and breast cancer.Acknowledgements
No acknowledgement found.References
1. Wang L, Wang D, Chai W, Fei X, Luo R, Li X. MRI features of breast lymphoma: preliminary experience in seven cases. Diagn Interv Radiol 2015;21(6):441-447.
2. Irshad A, Ackerman SJ, Pope TL, Moses CK, Rumboldt T, Panzegrau B. Rare breast lesions: correlation of imaging and histologic features with WHO classification. Radiographics 2008;28(5):1399-1414.
3. Alexey Surov. Imaging Findings of Hematologic Diseases Affecting the Breast. Semin Ultrasound CT MRI 2013; 34: 550-557
4. Iima M, Le Bihan D. Clinical Intravoxel Incoherent Motion and Diffusion MR Imaging: Past, Present, and Future. Radiology 2016;278(1):13-32.
5. Iima M, Kataoka M, Kanao S, et al. Intravoxel Incoherent Motion and Quantitative Non-Gaussian Diffusion MR Imaging: Evaluation of the Diagnostic and Prognostic Value of Several Markers of Malignant and Benign Breast Lesions. Radiology 2017:162853.
6. Sun K, Chen X, Chai W, et al. Breast Cancer: Diffusion Kurtosis MR Imaging-Diagnostic Accuracy and Correlation with Clinical-Pathologic Factors. Radiology. 2015; 277(1): 46-55.
Figures