2361
Title: Self-Regulation of vmPFC Activation Using Real-Time fMRI NeurofeedbackAhmad Mayeli1,2, Vadim Zotev1, Raquel Phillips1, Hazem Refai2, Martin Paulus1, and Jerzy Bodurka1,3
1Laureate Institute for Brain Research, Tulsa, OK, United States, 2Electrical and Computer Engineering, University of Oklahoma, Tulsa, OK, United States, 3College of Engineering, Stephenson School of Biomedical Engineering, University of Oklahoma, Tulsa, OK, United States
Synopsis
In this study, we have examined the feasibility of training healthy human subjects to self-regulate the hemodynamic activity of the vmPFC using real-time fMRI neurofeedback (rtfMRI-nf). Eight healthy subjects took part in experimental group with real rtfMRI neurofeedback from vmPFC and four in control group with a sham feedback from HIPS region. The results show significant vmPFC BOLD activity differences between the groups, demonstrating the feasibility of targeted modulation of the vmPFC using the rtfMRI-nf.
Purpose
Real-time functional magnetic resonance imaging neurofeedback (rtfMRI-nf) allows individuals to regulate hemodynamic activity of a target brain region in real time1,2. Studies employing rtfMRI-nf have demonstrated participants’ ability to learn volitional control of neurophysiological activity in various brain regions, including the amygdala, anterior cingulate cortex and parahippocampal cortex2,3,4. The ventromedial prefrontal cortex (vmPFC) plays important roles in regulation of anxious emotion and persistence responses in the face of uncontrollable setbacks5, 6. Furthermore, the vmPFC is one of the critical loci of a dynamic and flexible neural circuit that may underlie emotional and behavioral control and active resilient coping7. In this study, we examined the feasibility of training healthy human subjects to self-regulate the hemodynamic activity of the vmPFC. Participants in the experimental group were provided with an ongoing information about the blood oxygenation level dependent (BOLD) activity in the vmPFC in the form of rtfMRI-nf signal (variable-height bar). They were instructed to raise the rtfMRI-nf signal by self-relevant value-based thinking, e.g. “think of situation that make you feel accomplish, competent, or good at something”. Participants in the control group performed the same task. However they were provided with a sham neurofeedback based on BOLD activity of the left horizontal segment of the intraparietal sulcus (HIPS) region instead. The results show significant vmPFC BOLD activity differences between the groups, demonstrating the feasibility of targeted modulation of the vmPFC using the rtfMRI-nf.Methods
The study included 12 healthy subjects (6 females). The experiments were performed using GE MR750 3T MRI scanner with the 8-channel receive-only head coil. For the whole brain fMRI, a single-shot gradient echo EPI with sensitivity encoding (SENSE8) with FOV/slice=240/2.9mm, TR/TE=2000/30ms, SENSE acceleration R=2, 96×96, flip=90°, 34 axial slices was employed. T1-weighted MPRAGE sequence was used for anatomical reference and to define ROIs. We implemented the neurofeedback stimulus via our custom real-time fMRI system9 utilizing the real-time features of AFNI10 and a custom developed graphic user interface (GUI) software. For each subject, three spherical ROIs (7 mm radius in Talairach space) were transformed to the EPI image space using each subject’s high resolution MPRAGE structural data. These ROIs are defined at the vmPFC, left Amygdala and the left horizontal segment of intraparietal sulcus (HIPS) region. AFNI real-time plug-in was used to perform volume registration of EPI images in our neurofeedback implementation and to export mean values of fMRI signals for the three ROIs in real time. An average fMRI signal from the target ROI updated every 2s and was presented as a red bar (Fig.1). Eight subjects took part in experimental group with real rtfMRI neurofeedback (target ROI: vmPFC). For the other 4 subjects, a sham feedback from HIPS region was provided during neurofeedback runs. The experiment design is shown on Fig. 1. Each run (except Rest) starts with a 40 s rest block and a 40 s count block respectively, proceeding with 40 s long block with Think, and Count conditions. For the Think condition, the subject was asked to think about thoughts that are important for them which are specific, vivid, and highly arousing in order to activate vmPFC so as to raise the level of the red bar displayed on the screen. The target level (blue bar) was raised from run to run. No neurofeedback was provided (no bars displayed) during the Rest and Count conditions or during the entire Recall and Transfer runs. The fMRI data analysis was performed in AFNI based on GLM analysis described elsewhere2.Results
Figure 2 exhibits the average fMRI signal changes for the vmPFC during the Think condition compared to the Count condition for each neurofeedback run for the experimental and sham groups. The results show that (1) recall of self-relevant value-based thoughts robustly activates vmPFC, (2) subjects in experimental group are able to increase vmPFC activation during all neurofeedback runs, (3) subjects in the experimental condition show significant vmPFC activation during the transfer run.Discussion and conclusion
Our preliminary results demonstrate that by using rtfMRI-nf from the vmPFC during recall of self-relevant value-based thoughts, healthy individuals can learn to self-regulate their vmPFC BOLD responses.Acknowledgements
This work was supported by Laureate Institute for Brain Research.References
[1] deCharms RC, et al. Applications of real-time fMRI. Nat. Rev. Neurosci. 2008; 9:721. [2] Zotev V, et al. Self-regulation of amygdala activation using real-time fMRI neurofeedback. PLoS ONE 2011; 6:e24522. [3] deCharms RC, et al. Control over brain activation and pain learned by using real-time functional MRI. Proc. Natl. Acad. Sci. 2005; 102:18626. [4] Weiskopf N, et al. Self-regulation of local brain activity using real-time functional magnetic resonance imaging (fMRI). J. Physiol. Paris 2004; 98: 357. [5] Somerville LH, et al. Interactions between transient and sustained neural signals support the generation and regulation of anxious emotion. Cereb Cortex 2013; 23: 49. [6] Bhanji JP, et al. Perceived control influences neural responses to setbacks and promotes persistence. Neuron 2014; 83:1369. [7] Sinha R, et al. (2016) Dynamic neural activity during stress signals resilient coping. Proc. Natl. Acad. Sci. 2016; 113: 8837. [8] Pruessmann KP, et al. (1999) SENSE: sensitivity encoding for fast MRI. Magn. Reson. Med. 1999; 42:952. [9] Bodurka J. Real time software for monitoring MRI scanner operation. Paper presented at the Proceedings of Human Brain Mapping Conference, Melbourne. Neuroimage 2008; 41(Supp. 1): S85. [10] Cox RW. Jesmanowicz A. Real-time 3D image registration for functional MRI. Magn. Reson. Med. 1999: 42: 1014.Figures
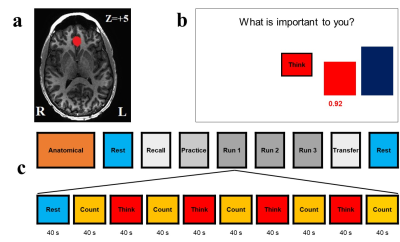
Figure 1. a) vmPFC ROI (marked in red) for rtfMRI neurofeedback. b)
GUI screen with neurofeedback bars (red) and target bars (blue); c)
experimental protocol.
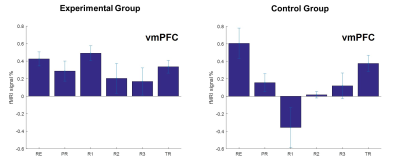
Figure 2. The mean percent signal change (averaged across Think
conditions during a given neurofeedback run) in the BOLD signal for vmPFC
region for experimental (left plot) and control groups (right plot). The error
bars represent the standard deviation of the mean.