2058
Practical parameter setting for simultaneous measurement of CBF and ATT with Hadamard-encoded ASL: Special reference for clinical practice1Radiological Center, University of Fukui Hospital, Yoshida-gun, Japan, 2Division of Health Sciences, Graduate School of Medical Sciences, Kanazawa Unversity, Kanazawa, Japan, 3Department of Radiology, Faculty of Medical Science, University of Fukui, Yoshida-gun, Japan, 4Global MR applications and Workflow, GE Healthcare Japan, Hino, Japan, 5Department of Medical Imaging, Faculty of Life Sciences, Kumamoto University, Kumamoto, Japan, 6Division of Ultrahigh Field MRI, Institute for Biomedical Sciences, Iwate Medical University, Shiwa-gun, Japan, 7GE Healthcare, Calgary, AB, Canada
Synopsis
Hadamard-encoded ASL (H-ASL) is a time-efficient method for measuring arterial transit time (ATT). The larger encoding matrix extends the scan time, but the accuracy of the ATT with a different encoding matrix was not clarified. This study aimed to propose a practical parameter selection in H-ASL for clinical use. The ATT was not significantly different between 3 and 7 delay encodings. Cerebral blood flow (CBF) obtained with 3 delay encodings with a linear division block design was equivalent to that obtained without encoding. Three delay encodings with a linear division block design provides accurate ATT and CBF within 4 minutes.
Introduction
Arterial spin labeling (ASL) signals are intrinsically affected by arterial transit time (ATT), as well as by perfusion signals. Non-compensated ATT gives rise to quantification errors and reduced tissue perfusion signals, indicating that compensation for ATT is essential for accurate estimation of cerebral blood flow (CBF).1 Correction of ATT extends the examination time owing to multiple post-labeling delay (PLD) acquisitions. Recently, Hadamard-encoded ASL (H-ASL), which simultaneously obtains high-resolution ATT maps and volumetric perfusion-weighted images, was demonstrated as a time-efficient method.2 However, the larger size of the encoding matrix led to longer acquisition times. In the clinical routine framework, the scan time of one sequence is desired to be within 5 minutes. Thus, the 7 delay encoding scheme with H-ASL was not feasible in the clinical framework without image quality degradation, i.e., thick slices and/or low resolution. Moreover, robust parameter selection for accurate ATT and CBF measurement is clinically needed, since hemodynamics are dependent on gender, age, and disease.3,4 Therefore, the purpose of the present study was to propose a practical parameter setting of H-ASL for clinical use. We compared the CBF, ATT-compensated CBF (ATC-CBF), and ATT varying with the number of delays in young and elderly healthy subjects.Materials and Methods
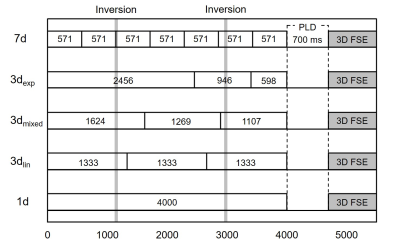
Seventeen healthy volunteers (young males, n = 7, 24.6±4.7 years old; young females, n = 5, 23.8±1.1 years old; elderly males, n=5, 54.6±2.4 years old) were enrolled to cover a wide age range and both genders. H-ASL was performed on a 3.0 T MRI unit (Discovery 750, GE Healthcare) with 32-channel head array coil. Pseudo-continuous labeling with 3D fast spin-echo (FSE) spiral readout was used with a labeling duration (LD) of 4000 ms, PLD of 700 ms, 1 delay (1d; not encoded), 3 delay (3d), 7 delay (7d), TR of 6225 ms, TE of 10.5 ms, FOV of 240 mm, 512 points with 6 interleaves, and the number of signal average of 1. Scan times were 1 min 30 s for 1d, 3 min 23 s for 3d, and 5 min 54 s for 7d. With 3d, we used 3 different block designs (3dlin, 3dmixed, and 3dexp; Fig. 1). A vascular suppression gradient was not applied. A reference image was acquired using saturation recovery 3D-FSE with a saturation time of 2000 ms. ATT was calculated using signal-weighted delay, which was previously reported as a robust method.1 NEURO FLEXER (Nihon Medi-Physics) was used to automatically delineate the regions of interest in the anterior cerebral artery, middle cerebral artery, and posterior cerebral artery territories at the basal ganglia level. ANOVA was used for statistical analyses.Results
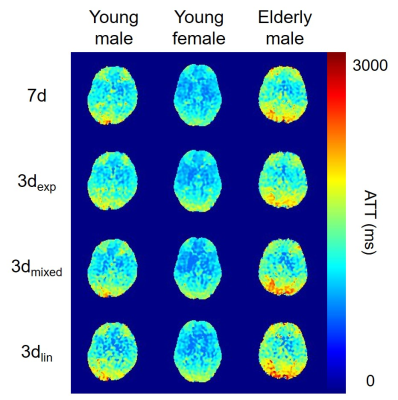
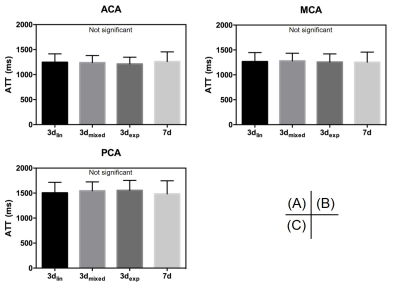
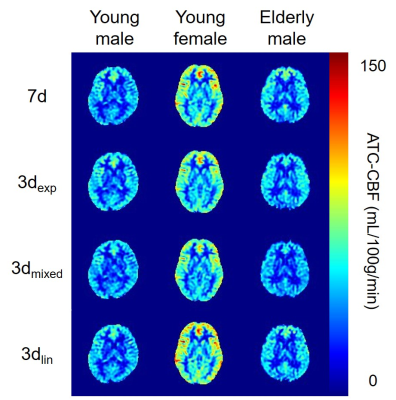
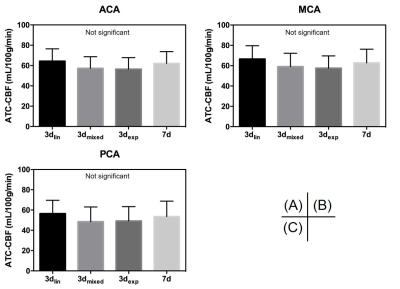
There was no significant difference in ATT in all vascular territories among the block designs (Fig. 2, 3). Similar results were observed within each group. CBF was not significantly altered among the encoding designs. However, CBF obtained with 3dmixed and 3dexp was smaller than that obtained with 1d, but this result was not statistically significant (data not shown). ATC-CBF was not significantly different (Fig. 3, 4). ATC-CBF obtained with 3dmixed and 3dexp was smaller than that obtained with 3dlin and 7d. Clear dependence of ATT and CBF on gender and age was observed.Discussion
The present study demonstrated that H-ASL with 3d encodings is an accurate and robust method for ATT measurement with short scan times. Concerning the ATT, the accuracy was not different between the 3d and 7d. Although CBF obtained with 3dlin and 7d was almost equivalent to that obtained with 1d, which was considered as a reference value, CBF obtained with 3dmixed and 3dexp was smaller than the reference CBF. This could be attributed to the degraded labeling efficiency. H-ASL needs label-control RF switching to encode the labeling bolus. In addition, two inversion pulses were applied during the labeling bolus. The timing of these combinations might have affected the labeling efficiency. In terms of the accuracy of CBF and ATT, 3d with a linear division block design was the best parameter selection for clinical use. 3dlin covers the ATT range from 700 ms to 3366 ms. We believe that this range could cover the entire ATT that may be clinically encountered. However, with the extremely prolonged ATT, e.g., Moyamoya disease3 and cerebral artery stenosis/occlusive disease,5 additional single-delay scan with long LD and long PLD should be considered. A combination of 3dlin and this additional scan will provide accurate ATTs.Conclusion
3d with a linear division block design is the best parameter setting for the clinical use of H-ASL. Additional single-delay scan should be considered in patients with extremely prolonged ATTs.Acknowledgements
No acknowledgement found.References
1. Dai W, Robson PM., Shankaranarayanan A, et al. Reduced resolution transit delay prescan for quantitative continuous arterial spin labeling perfusion imaging. Magn Reson Med. 2012; 67 (5), 1252–1265.
2. Dai, W, Shankaranarayanan A, Alsop DC. Volumetric measurement of perfusion and arterial transit delay using hadamard encoded continuous arterial spin labeling. Magn Reson Med. 2013; 69 (4): 1014–1022.
3. Tsujikawa T, Kimura H, Matsuda T, et al. Arterial transit time mapping obtained by pulsed continuous 3D ASL imaging with multiple post-label delay acquisitions: Comparative study with PET-CBF in patients with chronic occlusive cerebrovascular disease. PLoS One. 2016; 11 (6): e0156005.
4. Fujiwara Y, Matsuda T, Kanamoto M, et al. Comparison of long-labeled pseudo-continuous arterial spin labeling (ASL) features between young and elderly adults: special reference to parameter selection. Acta Radiol. 2017; 58 (1): 84–90.
5. Takeuchi K, Isozaki M, Kanamoto M, et al. Arterial transit time measured by multi-delay ASL perfusion for evaluating major cerebral artery stenosis/occlusive disease: correlation with 15O-H2O and 15O2 gas PET-CBF and OEF. Proceeding of 25th ISMRM. 4727.
Figures