1609
Comparison of three diffusion models: monoexponential vs. intravoxel incoherent vs. stretched model1Seoul National University Hospital, Seoul, Republic of Korea, 2Philips Healthcare Korea, Seoul, Republic of Korea
Synopsis
A diffusion heterogeneity index (α) derived from a stretched exponential model may serve as a more sensitive parameter for hepatic fibrosis compared with paramters from mono-or bi-exponential diffusion weighted imaging (DWI).
Introduction
In liver imaging, biexponential model using intravoxel incoherent motion (IVIM) has been applied to grade hepatic fibrosis and showed better diagnostic performance than monoexponential diffusion (1, 2). Bi-exponential diffusion weighted imaging (DWI) has been applied to liver imaging and showed better diagnostic performance to grade hepatic fibrosis. However, there is an unmet need for other models due to long data acquisition, reliability of the parameters and low diagnostic performance for fibrosis detection compared with stiffness imaging. A stretched-exponential model is one of non-mono-exponential diffusional models which provides explanation for non-exponential component in water diffusion (3). The purpose of this study is to compare the parameters of diffusion-weighted imaging using different models in patients with chronic liver disease.Methods
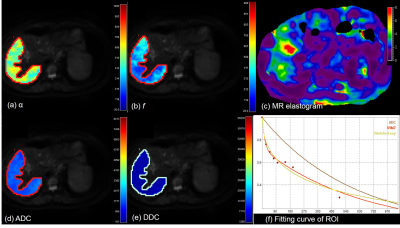
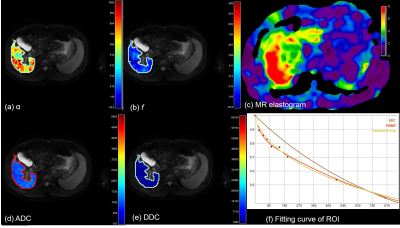
In this prospective study, 40 patients with chronic liver disease (CLD) who underwent liver MRI including diffusion weighted image were enrolled. Thirty out of 40 patients underwent MR elastography successfully at the same scanner. Diffusion weighted images (DWI) were performed at 3T scanner (Ingenia or Ingenia CX, Philips Healthcare, Best, the Netherlands), using nine b-values (0-900sec/mm2). Scan parameters were as follows: TR/TE, 1322/66 (msec); FOV, 350-400; flip angle, 90; NSA, 2; respiratory triggered. DWI were analyzed using three models of monoexponential (apparent diffusion coefficient, ADC), intravoxel incoherent (true diffusion (Dt), pseudodiffusion (D*), and perfusion fraction (f)), and stretched model (distributed diffusion coefficient (DDC), and alpha (α)) using following equations.
S(b) = S(0)[(1-f)e-bDt + fe-bD* ] (Eq 1)
S(b) = S(0)e-[bDDC]α (Eq 2)
The parameters were evaluated between groups with CLD-Child class A and Child class B. In addition, the correlation coefficient was obtained between diffusion parameters and liver stiffness (kPa) in 30 patients with MR elastography.
Results and Discussion
There were 36 patients with CLD-Child A and 4 patients with Child B. Patients with Child B showed lower f (17.7±7.0% vs. 24.7±5.8%, P=0.009) and D* (48.2±34.4 vs. 75.7±40.2 (x10-3)mm2/sec, P=0.04) than patients with CLD or Child A. In stretched model, α showed a significantly higher value in patients with Child B (0.62±0.08 vs. 0.47±0.09, P<0.0001) (Figs 1-2). However, ADC and DDC did not show significant difference between the two groups (P= 0.58, 0.98, respectively). Α also showed a significant correlation with liver stiffness value (r=0.5, 95% CI: 0.18, 0.73) in 30 patients with successful MR elastography acquisition (3.58±1.3 kPa in CLD-Child A vs. 7.3±0.6 kPa in Child B, P=0.001). f and D* did not show a significant correlation with liver stiffness in this group (P=0.07, 0.4, respectively). According to our observation, α from stretched model was able to provide information of the liver parenchyma. Given that Child class B often has advanced fibrosis stage, the higher heterogeneity index (α) may indicate advanced hepatic fibrosis, as liver stiffness was higher in those patients. Considering that the parameters related with true diffusion (ADC, Dt, and DDC) did not show a significant difference or significant correlation with liver stiffness value, the potential ability of α as a surrogate marker of hepatic fibrosis in DWI should be investigated further.Conclusion
Diffusion heterogeneity index (α) of stretched model showed a significant difference between patients with different Child-Pugh classification, and also significant correlation with liver stiffness from MR elastography, whereas other diffusion parameters of monoexponential, biexponential models did not.Acknowledgements
No acknowledgement found.References
1. Yoon JH, Lee JM et al, Evaluation of hepatic fibrosis using intravoxel incoherent motion in diffusion-weighted liver MRI. JCAT 2014;38(1): 110-116
2. Chung SR, Lee SS et al, Intravoxel incoherent motion MRI for liver fibrosis assessment: a pilot study. Acta Radiol 2015;56(12):1428-1436
3. Bennett KM, Hyde JS, Schmainda KM, Water diffusion heterogeneity index in the human brain is insensitive to the orientation of applied magnetic field gradients. Magn Reson Med 2006;56:235-239