1049
Deep Convolutional Neural Network Enhanced 3D High Resolution Turbo Spin Echo Intracranial Vessel Wall Imaging1Philips Research North America, Cambridge, MA, United States, 2Center for Biomedical Imaging Research, Department of Biomedical Engineering, Tsinghua University, Beijing, China, 3The Institute of Artificial Intelligence and Robotics, Xi'an Jiaotong University, Xi'an, China, 4Philips Research Hamburg, Hamburg, Germany, 5Vascular Imaging Lab, Department of Radiology, University of Washington, Seattle, WA, United States
Synopsis
Turbo spin echo (TSE) imaging with variable flip angle (VFA) is commonly used for three-dimensional (3D) high resolution intracranial vessel wall imaging. However, different tissues may experience various blurring effects particularly for longer TSE factor. In this study, a deep convolutional neural network is trained to provide a solution for this special deblurring problem. Combined with a signal-to-noise ratio (SNR)-priority VFA design scheme, the developed technique can provide a better tradeoff across scan efficiency, point spread function and SNR for 3D TSE acquisitions. Preliminary results have demonstrated its improvement for sharper delineation of intracranial vessel wall and plaque boundaries at isotropic 0.5mm resolution.
Introduction
The three-dimensional (3D) turbo spin echo (TSE) sequence with variable flip angle (VFA) refocusing pulses is widely used for high resolution intracranial vessel wall imaging (VWI)1-5. The VFA design strategy based on the extended phase graph6-8 can provide an ideal point spread function (PSF) for a specific tissue type, although with certain signal-to-noise ratio (SNR) penalty, while other tissues may experience different filtering effects leading to various PSF profiles. However, considering scan efficiency and voxel size, this SNR penalty might not be well compensated at 3.0T particularly for T1 weighted TSE scans, which however play an important role for intracranial VWI3-5. To preserve sufficient SNR, high resolution 3D TSE intracranial VWI may result in different image blurring problems for different tissues. In this study, a deep convolutional neural network (CNN) is developed as a post-processing method to restore the penalized high frequency structures during an optimized SNR-priority TSE image acquisition. Its performance is investigated in phantoms and in vivo using isotropic 0.5mm 3D T1 weighted TSE intracranial VWI at 3.0T.Methods
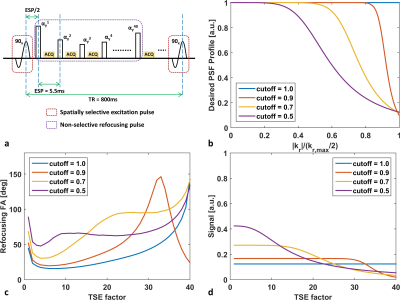
Optimization for T1 weighted TSE sequence: To further improve the SNR, the echoes generated through the entire Carr-Purcell-Meiboom-Gill echo train are acquired8 so that the echo time (TE) can be reduced to 5.4ms for isotropic 0.5mm resolution. The signal evolutions in the entire echo train are modulated by tuning the VFA design to achieve a desired PSF profile (e.g. Butterworth low-pass or all-pass filtering profile) for the vessel wall (T1/T2 = 844ms/39ms)9. The attenuation ratio of PSF gains between highest and lowest frequency component was 10, while the TSE factor of 40 was considered as a reasonable choice for scan efficiency (figure 1).
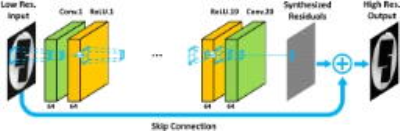
Deep CNN architecture for image super-resolution: A residual learning based deep CNN10, consisting of 20 convolutional layers with 64 channel of 3x3 filters in each layer (figure 2), is trained to synthesize the residuals or high frequency differences between different simulated low-pass filtered images and its original ones. The stochastic gradient descent with Nesterov momentum and the gradient clipping approach are used for training the parameters of this deep CNN. The training and testing of CNN were implemented with MatConvNet on a single GPU (NVIDIA, TITAN X).
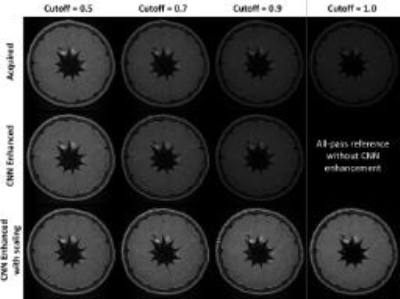
MR experiments: All of the data acquisitions were performed on a Philips Ingenia 3.0T MR scanner with 32-channel head coil. An orange was scanned for 4 times with different VFA schemes by configuring various cutoff frequencies from 0.5 to 1.0 (1.0 means all-pass filtering). The trained deep CNN was applied to enhance the low-pass filtered images and the restored results were compared with the acquired all-pass filtered images. Two healthy volunteers were also recruited to evaluate the performance of the developed technique in comparison to one of the existing methods11 for intracranial VWI. In addition, one image dataset from a previously recruited patient with intracranial atherosclerosis was retrospectively enhanced with deep CNN to assess its performance for plaque delineation.
Results
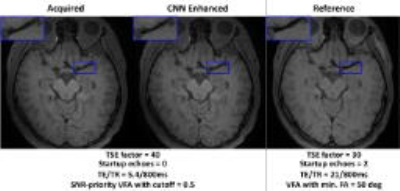
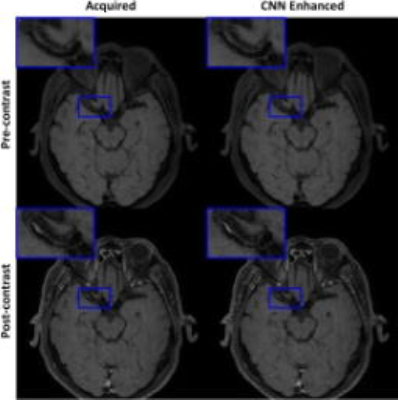
Given the same TSE factor, the low-pass filtering PSF design of VFA scheme can improve the SNR and the deep CNN can estimate the penalized high frequency structures during acquisition (figure 3). For healthy volunteer scans, the image quality of the developed method is comparable to the existing method, and the CNN enhanced image can provide sharper intracranial vessel wall delineation. In addition, the developed approach further improves the T1 image contrast due to reduced TE and the scan efficiency by ~33% due to longer echo train length (figure 4). Figure 5 demonstrates the improved visualization of intracranial plaque on the CNN enhanced images, indicating that deep CNN can also work separately to retrospectively enhance the acquired 3D TSE images.Discussion and Conclusion
Deep CNN has a multilayer architecture with multiple local filters in each convolutional layer, which can provide a powerful solution for TSE induced different blurring effects from different tissues. In addition, PSF profile driven VFA design scheme enables a SNR-priority TSE image acquisition without complete suppression of the high frequency components. The combination of optimized VFA design and deep CNN enhancement approach can provide a promising tool to make a superior tradeoff across scan efficiency, SNR and PSF for 3D TSE imaging. The preliminary results have demonstrated that the developed deep CNN enhanced high resolution TSE VWI technique can significantly improve the delineation of intracranial vessel wall without adverse impact on the image contrast. With this technique, 3D TSE based high resolution intracranial VWI can be better used at 3.0T to detect intracranial atherosclerosis and cryptogenic stroke.Acknowledgements
This study is supported by the grant from National Institutes of Health (5R01NS092207).
References
1. Qiao Y, Steinman DA, Qin Q, Etesami M, Schär M, Astor BC, Wasserman BA. Intracranial arterial wall imaging using three-dimensional high isotropic resolution black blood MRI at 3.0 Tesla. J Magn Reson Imaging. 2011;34(1):22-30.
2. Qiao Y, Steinman DA, Qin Q, Etesami M, Schär M, Astor BC, Wasserman BA. Intracranial plaque enhancement in patients with cerebrovascular events on high-spatial-resolution MR images. Radiology. 2014;271(2):534-42.
3. Yang H, Zhang X, Qin Q, Liu L, Wasserman BA, Qiao Y. Improved cerebrospinal fluid suppression for intracranial vessel wall MRI. J Magn Reson Imaging. 2016;44(3):665-72.
4. Fan Z, Yang Q, Deng Z, Li Y, Bi X, Song S, Li D. Whole-brain intracranial vessel wall imaging at 3 Tesla using cerebrospinal fluid-attenuated T1-weighted 3D turbo spin echo. Magn Reson Med. 2017;77(3):1142-1150.
5. Yang Q, Deng Z, Bi X, Song SS, Schlick KH, Gonzalez NR, Li D, Fan Z. Whole-brain vessel wall MRI: A parameter tune-up solution to improve the scan efficiency of three-dimensional variable flip-angle turbo spin-echo. J Magn Reson Imaging. 2017;46(3):751-757.
6. Hennig J, Weigel M, Scheffler K. Calculation of flip angles for echo trains with predefined amplitudes with the extended phase graph (EPG)-algorithm: principles and applications to hyperecho and TRAPS sequences. Magn Reson Med. 2004;51(1):68-80.
7. Busse RF, Hariharan H, Vu A, Brittain JH. Fast spin echo sequences with very long echo trains: design of variable refocusing flip angle schedules and generation of clinical T2 contrast. Magn Reson Med. 2006;55(5):1030-7.
8. Mugler JP 3rd. Optimized three-dimensional fast-spin-echo MRI. J Magn Reson Imaging. 2014;39(4):745-67.
9. Coolen BF, Poot DH, Liem MI, Smits LP, Gao S, Kotek G, Klein S, Nederveen AJ. Three-dimensional quantitative T1 and T2 mapping of the carotid artery: Sequence design and in vivo feasibility. Magn Reson Med. 2016;75(3):1008-17.
10. Kim J, Lee JK, Lee KM. Accurate image super-resolution using very deep convolutional networks. CVPR 2016.
11. Balu N, Zhou Z, Hatsukami T, Mossa-Basha M, Yuan C. Accelerated Multi-Contrast High Isotropic Resolution 3D Intracranial Vessel Wall MRI Using a Tailored K-Space Undersampling and Partially Parallel Reconstruction Strategy. ISMRM 2017, p2790.
Figures