0891
In-vivo diffusion-fMRI using Incomplete Initial Nutation Diffusion Imaging (INDI)1Champalimaud Research, Champalimaud Centre for the Unknown, Lisbon, Portugal, 2Department of Computer Science, University College London, London, United Kingdom
Synopsis
Diffusion functional-MRI (dfMRI) aims to capture microstructural changes occurring with neural activity. One of the confounding factors for dfMRI is that zero and nonzero b-values need to be acquired to deliver accurate changes in diffusivity, and those images are typically separated by at least one repetition time. Recently, Incomplete initial Nutation Diffusion Imaging (IDNI) was proposed for mapping the two images with a separation of <50ms. Here, we performed the first INDI-fMRI experiments using forepaw-stimulated rats. Functional signals are captured well both in ROI and voxel-wise analyses, enabling a more direct comparison of each signal’s time course.
Introduction
Diffusion functional-MRI (dfMRI) has been proposed as a non-BOLD method capable of mapping brain activity using a mechanism purportedly unrelated to hemodynamics1-3. However, to fully assess the mechanisms underlying dfMRI, a method that decouples T2 effects from diffusion effects is required. To date, dfMRI studies aiming to map changes in the diffusion coefficient with activity were mainly performed by interleaving zero and nonzero b-value acquisitions. Since T2 effects occur on scales of TR, the BOLD/non-BOLD effects may be intermixed. We here propose the use of Incomplete Initial Nutation Diffusion Imaging (INDI)4 for mapping the two b-values with <50 ms delay. The first results from rat forepaw stimulations, are discussed.Methods
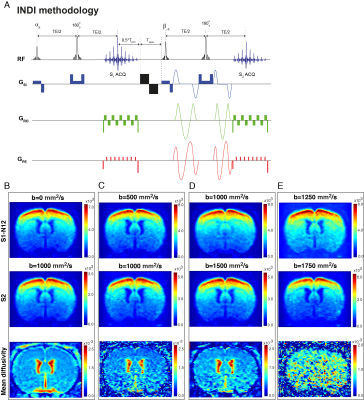
Animal preparation and setup: All experiments were preapproved by the local animal ethics committee operating under local and EU laws. Long Evan rats, 8-10 weeks old (n=5), were induced into deep anesthesia with 5% isoflurane and maintained under 2.5% isoflurane while two needles were inserted into the left forepaw’s digits 1-2 and 4-5. Animals were then switched to medetomidine sedation (bolus: 0.05mg/ml/kg, constant infusion: 0.1mg/ml/kg). Stimulation paradigm: Following 45 seconds of rest, electrical pulses were delivered to the forepaw with a square waveform comprising 1.5mA, 10Hz and 3ms stimulus duration, for 15 seconds followed by 45 seconds of rest for a total of 6 stimulation periods. Functional Imaging: All experiments were performed using a 9.4T Bruker BioSpec scanner equipped with a gradient system producing up to 660 mT/m isotropically. An 86 mm quadrature resonator was used for transmittance, while a 4-element cryoprobe was used for reception. Four coronal slices of interest were scanned between +1.68 and -4.36 mm from bregma. The INDI sequence4 (Figure 1A) was applied in fMRI mode using the following parameters: TR/TE=1500/40 ms, FOV=15.6 x 12.0 mm, matrix size of 78x60, partial Fourier 1.33, interpolation in read direction 1.05, slice thickness 1.5 mm, in-plane resolution 200µmx200µm). Pairs of images were acquired with b value pairs of: [0,1000], [500,1000], [1000,1500], [1250,1750] and Δ/δ = 19.5/14.2 ms, b tensor [zeta,theta,phi] = [54.73,0,0] deg, respectively. Data analysis: Images were denoised using Veraart’s MP-PCA approach5 and then spatially realigned using SPM in Matlab (The Mathworks, Nattick, USA). Then, regions of interest (ROIs) were drawn in the forelimb primary somatosensory cortex (layer IV), Mean±standard error of the mean signals were extracted and drift-corrected. The average temporal evolution was calculated by averaging all stimulation epochs from all animals. The spectral analyses involved Fourier and harmonics analyses. All anatomical information was inferred from the Paxinos&Watson rat brain atlas6.Results and Discussion
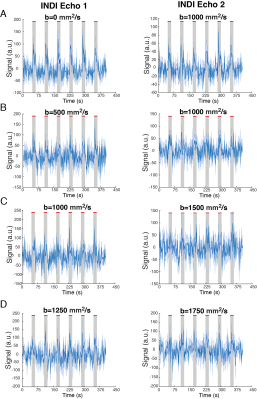
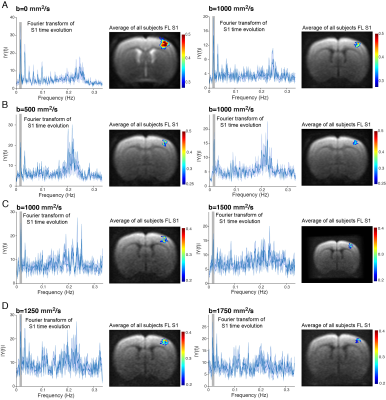
Raw data from a representative rat in the INDI-fMRI experiments are shown in Figure 1B-D. Despite that zero/nonzero b-values were acquired within ~50 ms, the signals remain within acceptable signal to noise ratio (SNR). The ROI analysis averaging the INDI-fMRI time series from all animals (Figure 2) showed significant responses in all b-values upon forepaw stimulation, whether originating from INDI’s S1 or S2 (i.e., the first or the second INDI echoes). Similar patterns were clearly observable even in single animals. To map the areas of activation from each b-value in a data-driven fashion and without assuming any a-priori response function, Fourier analysis was performed (Figure 3). Zero b-value images clearly show T2-BOLD contrast while the higher b-value images reveal more focal patterns. Importantly, the same focal patterns were also found for non-INDI dfMRI under our hands (data not shown). To avoid filtering large vessels at different b-values, INDI-fMRI experiments were also performed using nonzero b-values in the first echo as well; indeed, the ensuing INDI-S1 and INDI-S2 patterns are much more similar, suggesting that b=0 images are dominated by vascular components, while higher b-value images are dominated by tissue. Unfortunately, in this study, the quantitative mean diffusivity maps extracted from INDI suffered from heteroscedastic noise, and thus were difficult to interpret. Several solutions may resolve this drawback, such as nonlinear fitting using more time/decay points along the INDI signals or lowering the resolution to gain even higher SNR, and enable mapping the time evolution of diffusivities in the brain upon neural activity.Conclusion
To our knowledge, this is the first study to employ INDI4-fMRI for mapping diffusion-driven fMRI signals. The <50 ms separation of two b-values is favorable for mapping changes in diffusivity without bias due to TR interleaving. Heteroscedasticity needs to be dealt with to enable the interpretation of the mean diffusivity time evolution.Acknowledgements
This study was supported by funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (Starting Grant, agreement No. 679058) and one of the authors was supported by EPSRC grant number M507970.References
1. Tsurugizawa T, Ciobanu L, Le Bihan D. Water diffusion in brain cortex closely tracks underlying neuronal activity. Proc Natl Acad Sci U S A. 2013;110(28):11636-11641.
2. Abe Y, Tsurugizawa T, Le Bihan D. Water diffusion closely reveals neural activity status in rat brain loci affected by anesthesia. PLoS Biol. 2017;15(4):e2001494.
3. Jasanoff A. Bloodless FMRI. Trends Neurosci. 2007;30(11):603-610.
4. Ianus A, Shemesh N. Incomplete initial nutation diffusion imaging: An ultrafast, single-scan approach for diffusion mapping. Magn Reson Med. 2017.
5. Veraart J, Fieremans E, Novikov DS. Diffusion MRI noise mapping using random matrix theory. Magn Reson Med. 2016;76(5):1582-1593.
6. Paxinos G, Watson C . The Rat Brain in stereotaxic coordinates. 7th edition. Academic Press. 2014.
Figures