5251
Attentional modulation on the fMRI signal at human visual cortex revealed by fine timing characteristics but not amplitudes1Institute of Biomedical Engineering, National Taiwan University, Taipei, Taiwan, 2Aim for the Top University Project Office, National Taiwan Normal University, Taipei, Taiwan, 3Institute of Applied Physics, National Chengchi University, Taipei, Taiwan, 4Research Center for Mind Brain and Learning, National Chengchi University, Taipei, Taiwan, 5Institute of Neuroscience, National Yang-Ming University, Taipei, Taiwan, 6Department of Neuroscience and Biomedical Engineering, Aalto University, Espoo, Finland
Synopsis
By asking subjects engaging a decision task of two levels of difficulty based on foveal stimuli, we measured the BOLD dynamics elicited by checkerboard flashing shown
Target audience
Scientists interested in the advantage of fine temporal features of the BOLD signal.Purpose
Attentional modulation is an important mechanism in shaping information processing. For visual processing in human, top-down attentional modulation has been reported at extrastriate1 and striate cortices2,3 using fMRI measurements. The BOLD signal at subcortical areas along the visual processing pathway, including lateral geniculate nucleus and pulvinar, was also found to be enhanced or suppressed by attention4-6. However, attention has always been reported to change the magnitude of the fMRI signal. Here we hypothesize that different cognitive states, including attentional modulation, may be better manifested by temporal characteristics of fMRI responses. Manipulating the attention toward peripheral vision via requesting subjects engaging either an easy or difficult attention task at the central visual fixation, we measured the BOLD signal elicited by peripheral checkerboard stimulation using a high precision simultaneous-multi-slice inverse imaging (SMS-InI)7. We found that, while two tasks requiring different attentional loads elicited similar BOLD signal amplitudes, the hemodynamics significantly differed from each other in temporal characteristics, including onset, time-to-half, and the width of the BOLD signal.Methods
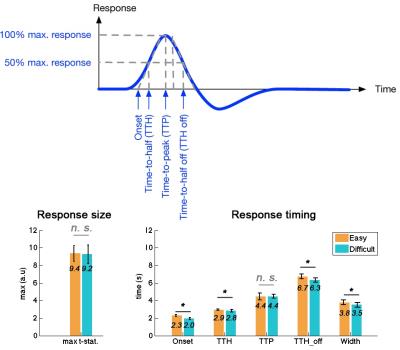
Twenty-four subjects were recruited to this study with written informed consents approved by the Institute Review Board. The fMRI experimental paradigm was adopted from a previous study4,5. Specifically, subjects were instructed to fixate at the center of the visual field to engage an easy or a difficult task based on foveal stimuli and to ignore the peripheral visual stimuli. In parallel with foveal visual stimuli, checkerboard reversals (8o visual angle; 8 Hz reversal rate; full contrast; 500 ms duration) were randomly presented to the subjects. Four runs (2 easy and 2 difficult attention tasks) were collected from each subject. Each run lasted 4 minutes, including 30 trials of easy/difficult task stimuli and 60 trials of peripheral checkerboard reversals. MRI data were collected on a 3T scanner (Skyra, Siemens, Erlangen, Germany) using a 32-channel head coil array. High resolution anatomical data were collected using the MRRAGE sequence. Functional MRI data were collected by SMS-InI, which provides 0.1 s sampling rate with 5 mm isotropic resolution and whole-brain coverage7. Dynamic t-statistics on the hemodynamics were estimated by the General Linear Model using finite impulse response function bases. Five timing indices were measured from the average hemodynamic response within the ROI: onset, time-to-half (TTH), time-to-peak (TTP,), time-to-half-during-off (TTH-off), and Width. The variability of timing indices for the group average responses were estimated over 100 bootstrap iterations to sample across subjects.Results
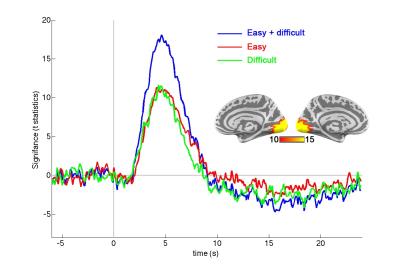
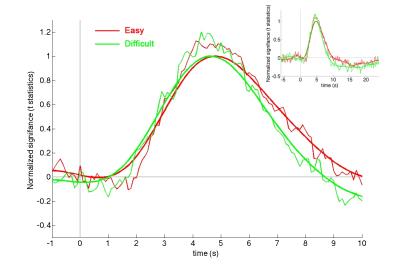
Across subjects, the reaction time for the easy task (431 ± 49 ms) was significantly shorter than that for the difficult task (512 ± 70 ms; p < 0.00001), suggesting two tasks had different effects over behavior. Strong hemodynamic responses were elicited at the visual cortex (Figure 1). To improve SNR and to examine fMRI waveforms without being confounded by the response amplitude, we moving-averaged the hemodynamic responses using a zero-phase moving-average (5 data points) filter and normalized the maximal amplitude to one. The normalized BOLD waveforms for easy and difficult tasks show different Onsets, TTHs, TTPs, and width (Figure 2). Quantitatively, there was no significant difference between the maximal amplitudes of the visual cortex responses when subjects engaged easy (9.4 +/- 0.9) and difficult (9.2 +/- 1.1) tasks (p = 0.41). However, the Onset, TTH, TTH_off, and Width differed significantly between easy (2.3 s +/- 0.1 s, 2.9 s +/- 0.1 s, 6.7 s +/- 0.3 s, 3.8 s +/- 0.3 s, respectively) and difficult (2.0 s +/- 0.1 s, 2.8 s +/- 0.1 s, 6.3 s +/- 0.2 s, 3.5 s +/- 0.3 s, respectively) tasks (all p’s < 0.00001). However, TTP did not differ between two tasks (easy: 4.4 s +/- 0.4 s; difficult: 4.5 +/- 0.2 s).Discussion
Considering the limited attention capacity8, the degree to process ignored stimuli is determined by the amount of spare attentional capacity. Accordingly, the BOLD signals elicited by checkerboards in easy and difficult tasks represented the BOLD signals in a relatively high and low attentional state, respectively. Our results demonstrated that the fine attentional modulation on the BOLD signal that has no amplitude difference can be disclosed by their differential timing characteristics, suggesting the advantage of sampling fMRI in high speed to further elucidate the neural correlates to behaviors and cognitive states. Since the hemodynamic responses were examined at the same region, the differences in timing characteristics were most likely attributed to neuronal, rather than vascular, origin.Acknowledgements
This work was partially supported by Ministry of Science and Technology, Taiwan (103-2628-B-002-002-MY3, 105-2221-E-002-104), and the Academy of Finland (No. 298131).References
1. Moran J. & Desimone R.Science.1985; 229:782-784.
2. Gandhi S. P., Heeger D. J. & Boynton G. M.Proc Natl Acad Sci U S A.1999; 96:3314-3319.
3. Somers D. C., Dale A. M., Seiffert A. E. et al.Proc Natl Acad Sci U S A.1999; 96:1663-1668.
4. Kastner S., O'Connor D. H., Fukui M. M. et al.J Neurophysiol.2004; 91:438-448.
5. O'Connor D. H., Fukui M. M., Pinsk M. A. et al.Nat Neurosci.2002; 5:1203-1209.
6. Schneider K. A. & Kastner S.J Neurosci.2009; 29:1784-1795.
7. Chu Y. H., Hsu Y. C. & Lin F. H.Proc Intl Soc Magn Reson Med.2016; 946.
8. Lavie N. & Tsal Y.Percept Psychophys.1994; 56:183-197.
Figures