5098
Feasibility of GRASP DCE-MRI in children with Juvenile Idiopathic Arthritis (JIA)1Radiology, AMC, Amsterdam, Netherlands, 2Biomedical Engineering & Physics, AMC, Amsterdam, Netherlands, 3Division of Imaging Sciences and Biomedical Engineering, King’s College London, London, United Kingdom
Synopsis
Juvenile Idiopathic Arthritis (JIA) is the most common rheumatic disease in childhood and represents one of the leading causes of pediatric acquired disability. The aim of our study was to evaluate feasibility of high spatiotemporal resolution GRASP DCE-MRI in children with JIA and secondly to compare image quality between GRASP and conventional DCE-MRI in evaluating synovitis. In this pilot study, GRASP DCE-MRI proved to be feasible in a child with JIA. Moreover, for the evaluation of synovitis significantly higher spatial resolution and much cleaner signal-enhancement plots could be obtained using GRASP compared to conventional DCE-MRI.
Purpose
Juvenile Idiopathic Arthritis (JIA) is the most common rheumatic disease in childhood and represents one of the leading causes of pediatric acquired disability.[1] It is characterized by prolonged synovitis that can lead to destruction of joints.[2] Since more effective anti-rheumatic drugs (biologicals) are increasingly used in JIA, the key treatment goal is to obtain complete suppression of joint inflammation in order to prevent these destructive changes.[3] Currently, quantitative outcome measures that objectively and quantitatively evaluate disease activity and therapy response in children with JIA are lacking.
In JIA, Dynamic Contrast-Enhanced (DCE) MRI has been suggested as an accurate and objective outcome measure.[4-7] A high temporal resolution is essential for DCE time-intensity-curve shape analysis. On the other hand, synovitis is considered present when the inflamed synovial membrane measures ≥2 mm. A high spatial resolution is therefore desired.
Recently, a study reported the use of the golden-angle radial k-space sampling, compressed sensing, and parallel-imaging reconstruction (GRASP) in DCE-MRI [8], delivering high spatiotemporal resolutions. We hypothesized that the use of GRASP DCE-MRI in the quantitative evaluation of synovitis could be feasible in JIA patients as well. Thus, the aim of our study was to evaluate feasibility of high spatiotemporal resolution GRASP DCE-MRI in children with JIA and secondly to compare image quality between GRASP and conventional DCE-MRI in evaluating synovitis.
Methods
Patients
For this feasibility study we collected data of two children with oligo-articular JIA and knee involvement, fulfilling the international league of associations for rheumatology (ILAR) criteria for JIA.[9]
MRI-protocol
In both patients DCE-MRI and conventional 3D T1W fat saturated post-contrast images were obtained using a 3.0-T magnet. No sedation was used. Both children were placed in the supine position with the knee joint centrally in the magnetic field in a dedicated knee coil. Forty-five seconds after the initiation of the DCE-MRI sequence, a bolus of a contrast agent followed by a 15 ml saline chase was delivered at an injection rate of 3 ml/sec by using an automatic injection device (Medrad, Warrendale, PA, USA). In one JIA patient a conventional DCE-MRI was used, while in the other a GRASP DCE-MRI was used. Conventional DCE-MRI
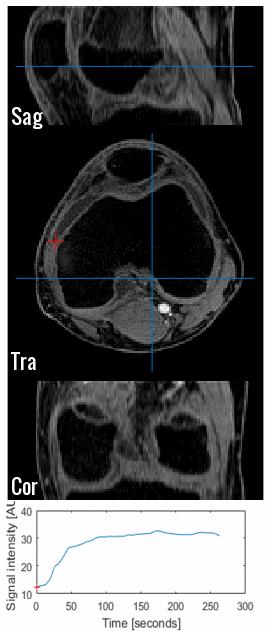
The standard DCE acquisition was performed using a conventional 3D ultrafast GE (THRIVE) technique with the following parameters: TR/TE 3.76/1.91ms, FA=12°, slice thickness 5 mm, 24 slices, FOV=240x240mm. These parameters resulted in a voxel size of 1.4x1.4x5mm and temporal resolution of 6.3 seconds.
GRASP DCE-MRI
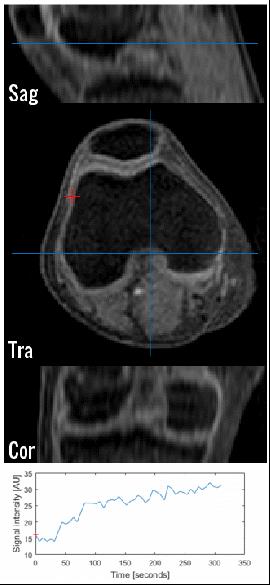
A 4D turbo field echo (TFE) acquisition (3 spatial dimensions+time) was performed using a stack-of-stars radial readout with tiny golden angle increments[10]. Sequence parameters were: FOV=200×200×72mm3 with 1.28 oversampling and 0.75 partial fourier in the Z-direction TR/TE=4.3/1.71ms, and FA=12°. The acquisition was performed with 1×1×2.7mm voxels. For each radial angle, spokes from all stacks were acquired before advancing to the next radial angle with a tiny golden angle increment of ~38.9°. After every 18th k-line, a SPIR fat-suppression pre-pulse was added. Post processing and reconstruction was done in MATLAB. Data were corrected for eddy currents and B0-phase delays.[11] Data were sorting using 37 spokes per dynamic phase (corresponding to 6.6 s, nearly equal to THRIVE DCE) with a reconstruction matrix size of 200×200×36, corresponding to a 1×1×2 mm3 resolution. This corresponds to an undersampling factor of 9.5 times. We used the BART toolbox[12] to perform a parallel-imaging CS reconstruction with a temporal total-variation l1-regularization constraint r=0.25, and 250 iterations.
Comparisons
Transverse (and coronal and sagittal reconstructions) conventional DCE and GRASP-DCE images were compared between both clinically active JIA patients. In both subjects, conventional T1W post-contrast images were compared to the final dynamic of the DCE series.
Results
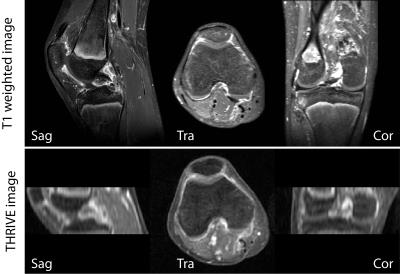
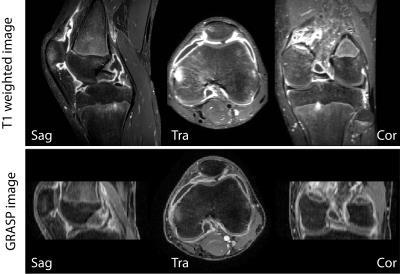
GRASP-DCE was successfully performed in a pediatric JIA patient. Figs. 1 and 2 show a movie of the DCE-series using THRIVE (Fig. 1) and GRASP (Fig. 2). Note the much higher spatial resolution and much cleaner signal-enhancement plots using GRASP DCE compared to the conventional DCE images. Figs. 3 and 4 show comparison of the final dynamic phase of the conventional DCE-MRI (Fig. 3) and GRASP DCE (Fig. 4) to post-contrast T1W images. Both patients had synovitis visible at MRI. In contrast to the conventional DCE-MRI, GRASP images are - as a result of much higher spatial resolution - almost equal to the diagnostic post-contrast 3D T1W FS MR images.Conclusion
In this pilot study, GRASP DCE-MRI proved to be feasible in a child with JIA. Moreover, for the evaluation of synovitis significantly higher spatial resolution and much cleaner signal-enhancement plots could be obtained using GRASP compared to conventional DCE-MRI.Acknowledgements
No acknowledgement found.References
1 Ravelli A, Martini A (2007) Juvenile idiopathic arthritis. Lancet 369:767-778
2 Haverman L, Grootenhuis MA, van den Berg JM et al (2012) Predictors of health-related quality of life in children and adolescents with juvenile idiopathic arthritis: Results from a web-based survey. Arthritis Care Res(Hoboken) 64:694-703
3 Oen K, Reed M, Malleson PN et al (2003) Radiologic outcome and its relationship to functional disability in juvenile rheumatoid arthritis. JRheumatol 30:832-840
4 Workie DW, Dardzinski BJ, Graham TB, Laor T, Bommer WA, O'Brien KJ (2004) Quantification of dynamic contrast-enhanced MR imaging of the knee in children with juvenile rheumatoid arthritis based on pharmacokinetic modeling. Magn ResonImaging 22:1201-1210
5 Workie DW, Graham TB, Laor T et al (2007) Quantitative MR characterization of disease activity in the knee in children with juvenile idiopathic arthritis: a longitudinal pilot study. PediatrRadiol 37:535-543 6 Malattia C, Damasio MB, Basso C et al (2010) Dynamic contrast-enhanced magnetic resonance imaging in the assessment of disease activity in patients with juvenile idiopathic arthritis. Rheumatology(Oxford) 49:178-185
7 Hemke R, Lavini C, Nusman CM et al (2014) Pixel-by-pixel analysis of DCE-MRI curve shape patterns in knees of active and inactive juvenile idiopathic arthritis patients. Eur Radiol 24:1686-1693
8 Rosenkrantz AB, Geppert C, Grimm R et al (2015) Dynamic contrast-enhanced MRI of the prostate with high spatiotemporal resolution using compressed sensing, parallel imaging, and continuous golden-angle radial sampling: preliminary experience. J Magn Reson Imaging 41:1365-1373
9 Petty RE, Southwood TR, Manners P et al (2004) International League of Associations for Rheumatology classification of juvenile idiopathic arthritis: second revision, Edmonton, 2001. JRheumatol 31:390-392
10 Wundrak S, Paul J, Ulrici J, Hell E, Rasche V (2015) A Small Surrogate for the Golden Angle in Time-Resolved Radial MRI Based on Generalized Fibonacci Sequences. IEEE Trans Med Imaging 34:1262-1269
11 Moussavi A, Untenberger M, Uecker M, Frahm J (2014) Correction of gradient-induced phase errors in radial MRI. Magn Reson Med 71:308-3123
12 BART: version 0.2.09 (2015) DOI: 10.5281/zenodo.31907
Figures