5011
T2-weighted Dixon TSE for accelerated simultaneous grading of whole body skeletal muscle fat infiltration and edema in patients with neuromuscular diseases1Department of Diagnostic and Interventional Neuroradiology, Technical University of Munich, Munich, Germany, 2Department of Diagnostic and Interventional Radiology, Technical University of Munich, Munich, Germany, 3Department of Neurology, Technical University of Munich, Munich, Germany, 4Friedrich-Baur-Institut, Ludwig-Maximilians-University, Munich, Germany, 5Philips Healthcare, Hamburg, Germany
Synopsis
The assessment of fatty infiltration and edema in the whole body musculature of patients with neuromuscular diseases typically requires the separate performance of a T1-weighted sequence and a fat suppressed T2-weighted sequence. T2-weighted Dixon TSE enables the generation of T2-weighted fat-separated and water-separated images, which could be used to simultaneously assess fatty infiltration and edema and to reduce total scan time. The present study examines the diagnostic performance of whole body T2-weigthed Dixon TSE imaging in 10 patients with neuromuscular diseases.
Purpose
MRI has been emerging as a popular technique in the diagnostic work up of patients with neuromuscular diseases in order to characterize whole body disease progression and guide muscle biopsy sites.1 The most important skeletal muscle degenerative alternations include fatty infiltration and edematous changes, which have been traditionally assessed based on the grading of T1-weighted images 2 and STIR images 3, respectively. However, the acquisition of two separate sequences in a whole body scan setting can lengthen the total acquisition time. A T2-weighted Dixon Turbo Spin Echo (TSE) sequence enables the simultaneous generation of T2-weighted fat-separated and water-separated images 4,5, which could be used to simultaneously assess fatty infiltration and edematous changes, respectively. Therefore, the purpose of the present study is to examine the diagnostic performance of T2-weighted Dixon TSE for accelerated simultaneous grading of whole body skeletal muscle fat infiltration and edema in patients with neuromuscular diseases.Methods
Subjects:10 patients (6 females and 4 males; median age 46 years; range 30 – 63 years) with varied hereditary neuromuscular diseases were recruited: muscular dystrophies (n=8), ALS (n=1) and congenital myopathy (n=1). Diagnosis was performed on the basis of muscle biopsies and/or genetic testing.
MRI measurements:
Patients underwent whole-body MRI on a 3T system (Ingenia, Philips Healthcare). Three groups of pulse sequences were performed in 5-6 axial stacks starting from the neck region to the talocalcanean joint: a 2-point Dixon FFE in order to generate T1-weighted images, a STIR TSE in order to generate T2-weighted images and a 2-point Dixon TSE in order to generate T2-weighted fat- and water-separated images (Table 1). Water-fat separation of the TSE and FFE images was performed online using the mDixon algorithm 5.
Muscle grading:
In order to compare the diagnostic performance of the three sequences, two different visual rating scales were used to grade the degree of pathological transformation in the patients’ muscles. For each patient 26 thigh muscles, 13 muscles on each leg, were scored conformingly by two evaluators. Severity of fatty infiltration was categorized via four different grades using the Mercuri grading scale2: score 1 for muscles with normal appearance, score 2 for muscles with fat filling not more than 30% of the volume, score 3 for muscles with fatty infiltration of 30 to 60% and score 4 for fatty infiltration of more than 60%. The Mercuri grading scale was applied independently on Dixon FFE fat and the T2-weighted Dixon TSE fat images. To rate hyperintensity indicating edema a three point scale published by Morrow et al.3 was used: score 0 for the absence of hyperintensity, score 1 for mild hyperintensity and score 2 for definite hyperintensity. The Morrow grading scale was applied independently on STIR TSE sequence and T2-weighted Dixon TSE water images. The agreement between the grading from different sequences was assessed using the Kappa agreement test.
Results
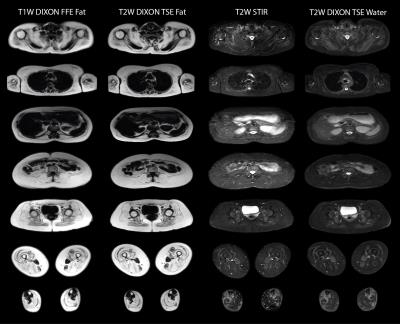
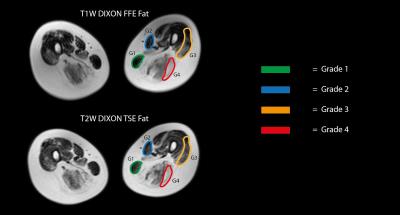
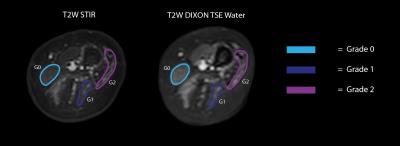
Figure 1 shows representative slices of the whole body scan of a severely affected LGMD2A patient. Figure 2 shows the Mercuri grading in a patient’s thigh. Affected muscles were graded equally on both Dixon FFE fat and T2-weighted Dixon TSE fat images: the gracilis with score 1, the sartorius with score 2, the vastus lateralis with score 3 and the long head of biceps femoris with score 4. Table 2 summarizes the excellent agreement for the Mercuri grading based on the Dixon FFE fat and the T2-weighted Dixon TSE fat images (Kappa coefficient = 0.93). Figure 3 shows the Morrow grading in a patient’s thigh. Affected muscles were graded equally on both the STIR TSE and the T2-weighted Dixon TSE water images: the gracilis with score 0, the long head of biceps femoris and the semitendinosus with score 1 and the vastus lateralis with score 2. Tabel 3 summarizes the excellent agreement for the Morrow grading based on the STIR TSE and the T2-weighted Dixon TSE water images (Kappa coefficient = 0.89).
Discussion and Conclusion
The present analysis shows a
statistically equivalent rating of fatty infiltration in Dixon FFE fat and T2-weighted
Dixon TSE fat images and a statistically equivalent rating of edematous changes
in STIR TSE and T2-weighted Dixon TSE water images. The replacement
of the two sequences by only one obviously reduces the total scan time. In
addition, the employed T2-weighted Dixon TSE protocol does not
require any breath-holding. However, the generally brighter appearance of
muscle tissue in the T2-weighted Dixon TSE water requires careful
comparison of the muscles within one patient. In conclusion, T2-weighted
Dixon TSE can be considered as a good alternative in the diagnostics of
neuromuscular diseases.Acknowledgements
The present work was supported by the German Society for Muscle Diseases, the European Research Council (grant agreement No 637164-iBack and grant agreement No 677661-ProFatMRI) and Philips Healthcare.References
1. Hollingsworth KG, et al. Toward harmonization of protocols for MRI outcome measures in skeletal muscle studies: Consensus recommendations from two TREAT-NMD NMR workshops, 2 May 2010, Stockholm, Sweden, 1-2 October 2009, Paris, France, Neuromuscul Disord 2012; S54-S672. Mercuri E, et al. Muscle MRI in Inherited Neuromuscular Disorders: Past, Present, and Future. J Magn Reson Imaging 2007; 25:433
3. Morrow JM, et al. Muscle MRI reveals distinct abnormalities in genetically proven non-dystrophic myotonias, Neuromuscul Disord 2013; 23:637
4. Reeder SB, et al. Iterative decomposition of water and fat with echo asymmetry and least squares estimation (IDEAL): application with fast spin echo imaging, Magn Reson Med 2005; 54:636
5. Eggers H, et al. Dual-echo Dixon imaging with flexible choice of echo times, Magn Reson Med 2011; 65:96
Figures