4193
Optimizing pediatric leptomeningeal metastasis detection: technical considerations1Diagnostic Imaging, St. Jude Children's Research Hospital, Memphis, TN, United States
Synopsis
MRI has assumed a central role in the detection of leptomeningeal metastasis for risk stratification of pediatric CNS tumors, but not all sequences are created equal for detection of tumor in CSF. In this presentation, we describe the strengths, weaknesses and tradeoffs of sequences commonly used for this purpose, and suggest guidelines for a targeted imaging protocol with increased sensitivity for leptomeningeal tumor, in less scan time.
Purpose:
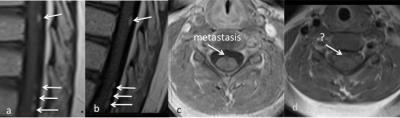
Detection of leptomeningeal metastasis (LM) is critical to staging and prognosis of childhood CNS cancers like medulloblastoma and ependymoma. 1 Though CSF examination is the historical gold standard for diagnosis, technological advances have earned MRI a central role in metastasis detection; recent work finds MRI more predictive of survival than CSF analysis. 2-4 However, not all sequences are created equal for detection of tumor in the CSF, and pediatric MR imaging presents additional unique challenges such as patient motion, acoustic noise and scan time reduction. In this exhibit, we discuss the strengths and weakness of common sequences for LM detection; technical alternatives for reduction of motion, acoustic noise and scan time; and present a suggested targeted imaging protocol based on current best imaging practice.Outline of Content:
With visual examples, we will discuss:
1. Signal characteristics of LM and role of complementary sequences for detection
2. Optimizing scan planes and slice thickness for tumor/metastasis location and patient size
3. Strengths and weaknesses of T1 FLAIR, SE, FSE/TSE, gradient echo and ultrafast spoiled gradient echo (VIBE/FAME/LAVA/THRIVE) sequences in terms of time, resolution, SNR and CSF artifact
4. Utility and optimization of post-contrast FLAIR, DWI, TrueFISP/bSSFP and subtraction images for metastasis detection
5. Reducing imaging time: targeted sequences, k-space undersampling (HASTE, partial Fourier imaging), parallel imaging
6. Reducing acoustic noise: lowering bandwidth, longer echo spacing, modified gradient wave forms, alternate encoding (PETRA, SWIFT, zero-TE)
7. Reducing motion artifact: 2D vs. 3D, non-Cartesian acquisition schemes, motion correction
Summary:
MRI has assumed a central role in the detection of leptomeningeal metastasis for risk stratification of pediatric CNS tumors. Analysis of the strengths, weaknesses and tradeoffs of commonly used sequences guides design of a targeted imaging protocol with increased sensitivity for leptomeningeal tumor, in less scan time.Acknowledgements
We wish to thank St. Jude Neuroradiologists Noah Sabin MD/JD and Kathleen Helton MD for their unwavering support and willingness to review and critique countless images in pursuit of imaging excellence.References
1. Engelhard HH, Corsten LA. Cancer Treat Res 2005;125:71-85
2. Maroldi R, Ambrosi C, Farina D. Eur Radiol 2005;15:617-626
3. Pang J, Banerjee A, Tihan T. Journal of neuro-oncology 2008;87:97-102
4. Terterov S, Krieger MD, Bowen I, et al. J Neurosurg Pediatr 2010;6:131-136
Figures