3329
Hyperpolarized xenon by d-DNP using the clinical GE SpinLab polarizer system1Department of Clinical Medicine, The MR Research Centre, Aarhus University Hospital, Aarhus N, Denmark, 2Department of Electrical Engineering, Center for Hyperpolarization in Magnetic Resonance, Technical University of Denmark, Kgs. Lyngby, Denmark, 3GE Healthcare, Denmark
Synopsis
Hyperpolarized (HP) 129Xe have been demonstrated as a useful probe for magnetic resonance (MR) lung imaging and show promise for in vivo perfusion imaging and brown adipose tissue characterization. Reports of large polarization enhancements for 129Xe using dynamic nuclear polarization (DNP) have raised expectations that DNP can be an alternative to the standard spin exchange optical pumping (SEOP) method. We show that it is possible to produce HP 129Xe gas using the clinical GE SpinLab polarizer, thus extending the practical use of the system beyond the primary purpose of hyperpolarizing liquid biomolecules.
Purpose
Hyperpolarized (HP) 129Xe for magnetic resonance (MR) has wide use in material sciences, structural chemistry and medicine. In the medical field 129Xe has been demonstrated as a useful probe for MR lung imaging and proposed as a blood tracer for in vivo perfusion imaging1. The xenon probe is therefore promising for clinical assessment of brain function2, and may aid in the diagnosis and prognosis of brain diseases. Additionally, HP 129Xe imaging has been demonstrated as a tracer for the presence and thermogenic activity of brown adipose tissue (BAT), in relation to the study of weight regulation3. Standard production of HP 129Xe is via spin exchange optical pumping (SEOP). However, reports of large polarization enhancements for 129Xe via dynamic nuclear polarization (DNP)1,4 have raised expectations that DNP can be an alternative method for producing HP 129Xe for patients. We therefore investigated the possibility for production of HP 129Xe using the clinical GE SpinLab polarizer, extending the practical use of this system.Materials and methods
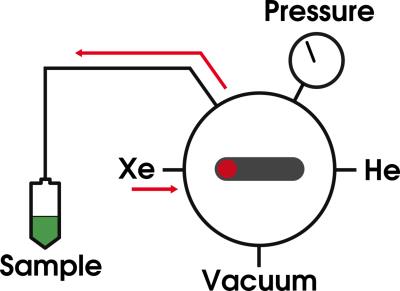
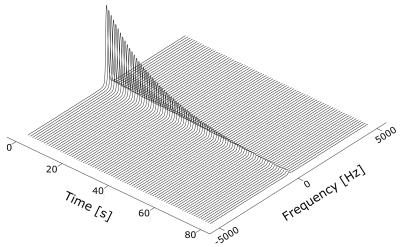
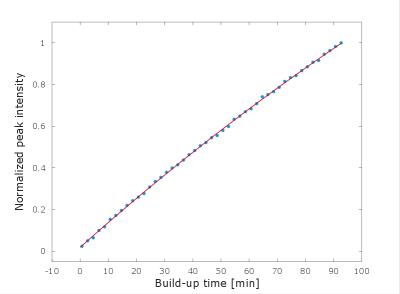
Solid state samples of 0.80 ± 0.01 mL 1-propanol / 12 mM AH111501 radical, infused with natural abundance (n.a.) or enriched (70% 129Xe) xenon gas at 5.0 ± 0.2 bar partial pressure for 60 ± 1 minutes at room temperature were prepared, using a custom built gas handling system, see Figure 1. Subsequently, the samples were inserted into the 5 T SpinLab polarizer at Aarhus University Hospital, depicted in Figure 2. Microwaves at 140.01 GHz were used to irradiate the samples for 150 ± 2 minutes at 0.90 ± 0.04 K followed by dissolution, transport and MR measurement. HP / thermal MR experiments were performed with 9° ± 1° / 90° ± 1° flip angles and TR = 1 s / 300 s on a 9.4 T small bore rodent MR scanner (Agilent, UK) using a volume radio frequency (RF) coil (Doty scientific, US) with 1 / 200 averages. A recorded signal decay curve of the resulting HP 129Xe gas is shown in Figure 3. Transport times for the n.a. / enriched gas experiments (n = 2 / 5) were 32 ± 1 s / 26 ± 1 s at earth field. Finally, an example of the solid state DNP build-up curve is presented in Figure 4. The solid state data was recorded using the 6.7 T SpinLab polarizer available at the Technical University of Denmark, using microwaves at 187.96 GHz. Data analysis was performed in Matlab (MathWorks, Natick, MA, USA).Results
The experiments resulted in enhancements / polarizations of 214 ± 22 / 0.18 ± 0.02 % and 446 ± 28 / 0.38 ± 0.02 % (n = 2 / 4) with T1 = 29 ± 2 s and 27 ± 2 s (n = 2 / 5) for n.a. and enriched gas, respectively. Additionally, solid state build-up times in the five to six-hour range was determined from the solid state build-up experiments (n = 2), using the described approach.Discussion and conclusion
The experiments have shown that it is possible to hyperpolarize 129Xe using the SpinLab system. However, substantial work is required to shorten build-up times and to achieve similar enhancements to SEOP. Strides should also be taken to prolong the T1 lifetime of the resulting HP gas. These issues can be addressed on multiple fronts, including optimization of the radical and xenon concentrations, optimization of the polarization build-up time, using different solvents, improving the phantom and optimizing the transport conditions. Future uses for this application include the in vivo detection of BAT activity in animal models as well as lung and perfusion imaging. This will be accomplished in combination with the implementation of a fast multi-echo balanced steady state free precession (BSSFP) imaging scheme, capable of producing high resolution spectroscopic maps of the gaseous and dissolved xenon phases, using the iterative Dixon type reconstruction technique (IDEAL)5.Acknowledgements
No acknowledgement found.References
1 A. Capozzi, C. Roussel, A. Comment, J.N. Hyacinthe; Optimal Glass-Forming Solvent Brings Sublimation Dynamic Nuclear Polarization to 129Xe Hyperpolarization Biomedical Imaging Standards. Phys. Chem. C 199 (2015) 5020-5025.
2 M. L. Mazzanti, R. P. Walvick, X. Zhou, Y. Sun, N. Shah, J. Mansour, J. Gereige, M. S. Albert; Distribution of Hyperpolarized Xenon in the Brain Following Sensory Stimulation: Preliminary MRI Findings. Plos One. 7 (2011) 1-7.
3 R. T. Branca, T. He, L. Zhang, C. S. Floyd, M. Freeman, C. White, A. Burant; Detection of brown adipose tissue and thermogenic activity in mice by hyperpolarized xenon MRI. Proceedings of the National Academy of Sciences of the United States of America, 111(50) (2014) 18001–18006.
4 A. Comment, S. Jannin, J. Hyacinthe, P. Miville, R. Sarkar, P. Ahuja, P. R. Vasos, X. Montet, F. Lazeyras, J. Valle, P. Hautle, J. A. Konter, B. van den Brandt, J. Ansermet, R. Gruetter, and G. Bodenhausen; Hyperpolarizing Gases via Dynamic Nuclear Polarization and Sublimation. Phys. Rev. Lett. 105 (2010) 018104.
5 J. Leupold, S. Månsson, J. S. Petersson, J. Hennig, O. Wieben; Fast multiecho balanced SSFP metabolite mapping of 1H and hyperpolarized 13C compounds. Magma, 22(4) (2009) 251-6.
Figures