2743
A 2D combined myocardium T1 and T2 mapping1Center for Biomedical Imaging Research, Department of Biomedical Engineering, School of Medicine, Tsinghua University, Beijing, People's Republic of China
Synopsis
In this study, we developed a 2D combined myocardial T1 and T2 mapping sequence that uses a combination of saturation pulse and T2 preparation pulse and allows simultaneously obtaining T1 and T2 map with acceptable breath holding time (12 heartbeats). High quality multiple T1 and T2 weighted images were obtained in the other cardiac cycle between the saturation pulse and T2 preparation pulse. Phantom experiment showed that T1 and T2 measured by proposed method highly correlated with reference methods. In vivo experiment showed that the proposed sequence can yield comparable myocardium T1 and T2 values with the conventional separated T1 and T2 mapping sequences.
Introduction
Cardiac MR T1 and T2 mapping techniques appeal great attention to quantify the myocardium tissue characteristics in clinic recently1. In general, T1 mapping and T2 mapping are separately scanned, thus having disadvantages of relative long scan time and possible misregistration between the T1 and T2 maps. These disadvantages can be overcome by performing T1 and T2 mapping in a single scan2,3,4. Recently, a joint myocardial T1 and T2 mapping sequence based on a combination of saturation recovery and T2 preparation pulses has been proposed4. However, due to the short saturation recovery time, this sequence may suffer from low signal to noise ratio (SNR).
In this study, we sought to develop a 2D combined myocardial T1 and T2 mapping sequence to obtain high quality T1 and T2 maps simultaneously.
Methods
Sequence:
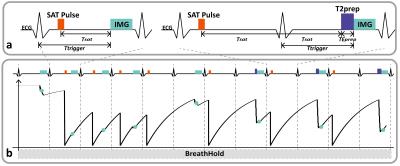
Diagram of the proposed sequence and its signal evolution are shown in figure 1. The preceding part of the proposed sequence firstly acquires an image without any magnetization preparation. Then 3 images are acquired with different T1 weightings by using saturation pulse at different recovery times [100, 300 and 500] ms. At last 4 hybrid T1-T2 weighted images are acquired with a same T1 weighting but different T2 weightings using T2 preparation (T2prep) pulse (TE = 0, 25, 35 and 45ms). In order to gain more available signal before the T2 preparation pulses, we finish every hybrid T1-T2 weighted image in 2 cardiac cycles. ECG trigger was used for readout in diastole. Overall the acquisition of 8 T1 and T2 weighted images can be completed within 12 heartbeats in a breathhold.
T1 and T2 fitting:
Pixel-wise T1 and T2 fitting were conducted with the following model:
S = A –B* exp(-Tsat/T1)) exp(-TEprep/T2)
Where S is the signal intensity, A and B are the coefficients of fitting model, Tsat and TEprep represented saturation recovery delay time and T2 preparation echo time, respectively.
Phantom and healthy volunteer experiments:
All imaging studies were performed on a 3T MR system (Achieva TX, Philips Healthcare, Best, Netherlands). An 8-channel head coil was used for phantom measurements, and a 32-channel cardiac coil was used for in vivo study.
Fifteen T1 [500-2000] ms and T2 [20-80ms] phantom vials were prepared and scanned with the proposed sequence. The reference T1 and T2 values were measured by using inversion recovery and CPMG spin echo sequences, respectively. Approved by the local institutional review board, in-vivo experiments were performed on 4 healthy volunteers (3 males, HR=58-80BPM), with written informed consent obtained. All subjects were scanned with the proposed sequence, SASHA (for T1 mapping)5, and a conventional 2D T2 mapping6 . Typical imaging parameters shared in these three sequences were: single slice, FA=35°, SENSE factor=2, partial echo=0.85, voxel size=1.7x2.1x8mm3, single shot bSSFP with linear profile order.
Results
From the phantom experiment, there were
strong correlations between the proposed method and the spin echo based
reference methods for T1 (R2 = 0.99, N = 15, p< 0.001) and T2 (R2 = 0.87, N
= 15, p<0.001) measurements.
In the in-vivo study, the myocardium T1
values measured by SASHA5 and proposed method were 1567±54.7ms and 1566±57ms
respectively. The myocardium T2 values measured by compared 2D T2 mapping
method6 and the proposed sequence were 43.9±4.2ms and 39.5±4.6ms, respectively.
In both phantom and in-vivo experiments, the propose method produced higher T1
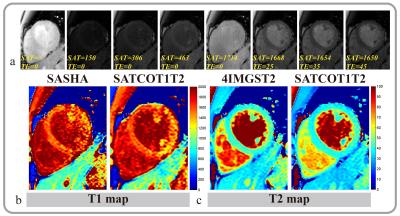
and lower T2 than the compared methods. Representative in-vivo images were
shown in figure 2.Conclusion
In this study, we proposed a 2D combined
myocardial T1 and T2 mapping sequence in a breathhold. The sequence can improve
the original T1 and T2 weighted images by extension of the saturation recovery
delay. Both phantom and in vivo experiments showed that the proposed sequence
can yield comparable results with the conventional separate T1 mapping and T2
mapping sequences. The preliminary results indicated the feasibility of the
proposed sequence for clinical application.Acknowledgements
We are appreciated the helpful discussion and suggestion from Dr. Daniel A. Herzka from Johns Hopkins University.References
1. Cardiovascular Division, et al. JACC. 2013.
2. Blume U, et al. JMRI. 2009.
3. Hamilton JI, et al. MRM. 2016.
4. Akçakaya M,et al. MRM. 2015.
5. Chow K, et al. MRM. 2013.
6. Giri S, et al. JCMR. 2009.
Figures