2183
Enhanced neuroplasticity by physical exercise for hypoxic ischemic injured rat brain monitored by BOLD-fMRI1Center for Neuroscience Imaging Research, Institute for Basic Science (IBS), Suwon, Korea, Republic of, 2Department of Health Sciences and Technology, SAIHST, Sungkyunkwan University, Seoul, Korea, Republic of, 3Department of Radiology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea, Republic of, 4Center for Molecular and Cellular Imaging, Samsung Biomedical Research Institute, Seoul, Korea, Republic of, 5Department of Global Biomedical Engineering, Sungkyunkwan University, Suwon, Korea, Republic of
Synopsis
Physical exercise is known to boost the spontaneous recovery, but the exact mechanism for exercise induced recovery after brain injury is not clearly known. In this study, we show enhanced neuroplasticity with physical exercise on the intact side as well as the damaged side for the HI injured rat brain by calculating the brain activation maps from BOLD-fMRI: Increased inter-hemispheric transfer and intra-hemispheric extension of activated areas were observed with physical exercise covering the widespread sensory-motor related areas in the contralesional hemisphere.
Purpose
Hypoxic ischemic (HI) injury causes a major brain injury in the newborn, leading to encephalopathy associated with cognitive and behavior deficits later in life1. However, the developing brain is known to be prone to remarkable spontaneous recovery from injury compared to the adult to compensate for loss of function through reorganization of neural networks and/or recruitment of new cortical areas2,3. Physical exercise as a mean of rehabilitation is known to boost the spontaneous recovery4, however, the exact mechanism for exercise induced recovery after brain injury is not clearly known. In this work, we demonstrate the enhanced recovery by physical exercise for hypoxic ischemic injured rat brain by BOLD-fMRI. The extents and locations of neural plasticity with and without physical exercise after the injury are to be compared.Materials and Methods
Animal Preparations: A total of 24 Sprague-Dawley rats (postnatal day7, 16-20g) were randomly divided into four groups: Group1, HI injury_no-exercise (n= 6); Group 2, HI injury_physical-exercise (n= 6); Group 3, Sham operation, no-exercise (n=6); Group 4, Sham operation_physical-exercise (n=6). All rats were subjected to right common carotid artery (CCA) occlusion and subsequent hypoxic exposure to 8% O2 for 150 min (36℃) for hypoxic ischemic injury (Rice-vannucci model). Sham operated rats were subjected to only incision into the skin on the right CCA, which was then closed immediately after.
Physical exercise and behavior test: Physical-exercise was performed with a rotarod (Rotarod performance test) 5 days per week for 5 min per each session, which began at 4th week after the injury until 9th week. For behavior test, all subjects were on the accelerating rotarod and measured for the time of falling down once a week.
BOLD-fMRI experiment: All BOLD-fMRI data were acquired at 6wks and 9wks after the injury using Bruker 7T MRI scanner (Bruker Biospoin GmbH, Ettlingen, Germany) equipped with an array head coil for receiving and quadrature birdcage coil for transmitting. Rats were intubated with a 16 G vascular catheter to ventilation (PCO2=35~45, PO2=80~100 and isoflurane= 1.2%). Electrical stimulation was applied to each forepaw for BOLD-fMRI at a frequency of 12 Hz (pulse width= 1.0 ms, current= 1.4 mA). The stimulation paradigm consisted of pre-stimulus for 20s, stimulus for 20s and post-stimulus for 40s (Boxcar design) (Fig. 1).We used single-shot gradient echo EPI sequence using the following acquisition parameters; TE= 60 ms, TR= 1000 ms, flip angle= 45°, number of average=1, field of view= 30 (readout) ×15 (phase encoding) mm2, matrix size= 64×32, in-plane resolution= 469×469 μm2, slice thickness= 1.5 mm, number of slices = 5 coronal slices, number of repetition= 80.
Data Analysis: All preprocessing was performed using the Analysis of Functional NeuroImages (AFNI) and FMRIB Software Library (FSL) packages including slice timing correction, motion correction, temporal normalization, linear registration, spatial smoothing and spatial normalization. And voxel-wise cross correlation analyze was conducted between with the BOLD temporal time series data and the electric stimulation paradigm.
Results and Discussion
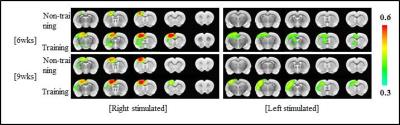
For the HI injured rats, almost all one side of hemisphere including cortical and subcortical areas is subjected to severe neonatal HI brain damage producing a porencephalic cyst covering majority of the ipsilesional cortical brain structure (Fig. 2). As clearly shown in Figure 2, BOLD-fMRI signal amplitudes were higher for exercise groups than no-exercise groups: Increased BOLD signal activation was observed mostly in S1FL and M1 in the contralesional hemisphere for right forepaw stimulation (intact side) compared to the sham operation group. For the left forepaw stimulation, the BOLD-signal activation was also observed in the contralesional hemisphere mostly in S1ULp and S1BF with lower signal amplitude compared to the intact hemisphere.Conclusions
In this study, we show the brain activation map by BOLD-fMRI for the HI injured rat brains that were subjected to physical exercise after the injury. Enhanced neuroplasticity with physical exercise after the injury was clearly demonstrated on the intact side as well as the damaged side: Increased inter-hemispheric transfer and intra-hemispheric extension of activated areas were observed with physical exercise covering the widespread sensory-motor related areas in the contralesional hemisphere.Acknowledgements
No acknowledgement found.References
1. Johnston MV, Trescher WH, Ishida A, Nakajima W., et al. Neurobiology of hypoxic-ischemic injury in the developing brain. Pediatr Res. 2001 Jun;49(6):735-41
2. Jung WB, Im GH, Chung JJ, et al. Neuroplasticity for spontaneous functional recovery after neonatal hypoxic ischemic brain injury in rats observed by functional MRI and Diffusion tensor imaging. Neuroimage. 2016 Feb 1; 126:140-50
3. Jhonston MV, et al. Plasticity in the developing brain: implications for rehabilitation. Dev Disabil Res Rev. 2009;15(2):94-101
4. Hou TT, Yang XY, Xia P, et al. Exercise promotes motor functional recovery in rats with corticospinal tract injury: anti-apoptosis mechanism. Neural Regan Res. 2015 Apr; 10(4) 644-50
Figures