1889
Measurement of lung perfusion using optimized pseudo-continuous arterial spin labeling of pulmonary arteries and fast True-FISP imaging at 3 Tesla1Section on Experimental Radiology, University of Tübingen, Tübingen, Germany, 2Max Planck Institute for Biological Cybernetics, Tübingen, Germany, 3Institute of Signal Processing and System Theory, University of Stuttgart, Stuttgart, Germany, 4Department of Diagnostic and Interventional Radiology, University of Tübingen, Tübingen, Germany, 5Department of Paediatric Cardiology, University of Tübingen, Tübingen, Germany, 6Department of Biomedical Magnetic Resonance, University of Tübingen, Tübingen, Germany
Synopsis
Pseudo-continuous-arterial-spin-labeling (pCASL) has been successfully applied in the brain and kidney providing high signal-to-noise-ratio. The aim of this study was to optimize pCASL for measurement of lung perfusion by optimized labeling of pulmonary arteries and fast signal acquisition. Effective labeling of pulmonary arteries was possible by ECG triggering and an appropriate orientation of the labeling plane. Sufficient signal from lung parenchyma was acquired by True-FISP imaging with TE=1ms. The presented method provides high quality perfusion images of the lung without applying intravenous contrast agents and offers diagnostic imaging of lung diseases such as pulmonary embolism and bronchial carcinoma.
Introduction and Purpose
The spatial distribution of lung perfusion is important for diagnosing lung diseases such as pulmonary embolism, pneumoconiosis, and bronchial carcinoma. Arterial spin labeling (ASL) has been shown to be a promising non-invasive approach for perfusion measurements of the lung.1,2 Pseudo-continuous ASL (pCASL) has been successfully applied in the brain3 as well as kidney4 and provides in general higher signal-to-noise-ratio than pulsed ASL approaches.5 Even though the lung is an organ with very high perfusion, perfusion imaging is still a challenging task. First, due to the complex vessel anatomy and highly pulsatile nature of pulmonary circulation, the vessel identification for accurate blood labeling is a tough choice. Recently Greer et al. measured pulmonary perfusion using pCASL of the inferior vena cava.6 However, the non-labeled blood from the superior vena cava hampers the accurate quantification of lung perfusion. Another important issue in lung imaging is the detection of sufficiently high and reproducible signal intensity from the lung parenchyma, which is generally hampered by its low proton density and extremely short T2* values.7
In this work, both issues are addressed: 1) Optimization of pCASL to measure the lung perfusion by directly labeling the pulmonary arteries (PA). For accurate and effective labeling, the tagging duration was limited to the systolic period using ECG-triggering. 2) To minimize the signal loss in the lung, fast True-FISP acquisition with short echo time was utilized.
Methods
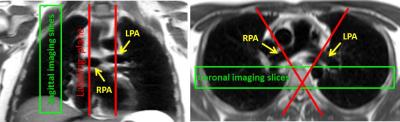
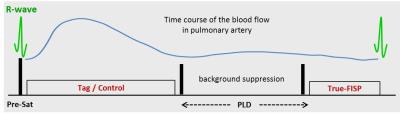
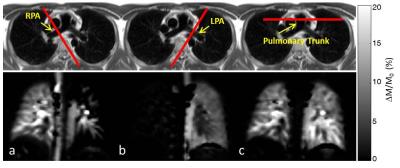
Measurements were performed on a 3T MR scanner (MAGNETOM Prisma, Siemens Healthcare, Erlangen, Germany). The lung perfusion of four healthy volunteers was measured using a pCASL sequence8 with background suppression (BS) and True-FISP data acquisition module. The True-FISP sequence was adapted to achieve short TE/TR (1.0/2.3ms) using following parameters: four slices with thickness/gap, 10/3mm; in-plane resolution, 4×4mm2; partial-Fourier, 0.75; acquisition matrix, 45×64/87×96 (sag/cor); readout bandwidth, 1240Hz/Pixel. Tagging duration (TD) was varied from 100 up to 500ms and post-labeling duration (PLD) was in the range of 500-1200ms. For labeling, a flip angle (FA) of 25°/40° and a gradient strength of 7mT/m were used. BS for lung tissue (T1~1400ms) was utilized using a double inversion approach. Forty label-control image pairs were acquired with a repetition delay of 4s by employing a timed breathing protocol. The overall scan time was in the range of 3:30-5:00min. A proton-density weighted image was also acquired at the start of the sequence. The tagging plane was placed nearly perpendicular to the right or left pulmonary artery (RPA and LPA) allowing separate perfusion measurements of the right and left lung (Figure 1). In one volunteer the pulmonary trunk was labeled allowing simultaneous perfusion imaging of the right and left lung. Since blood supply to the lung is mainly present during the systolic period, the tagging duration was limited to this time, avoiding unnecessary RF power deposition in the body. The data acquisition was performed in diastole in the same (Figure 2) or the next cardiac cycle. Images which showed strong motion artifacts were discarded from subsequent analysis using MATLAB®. Perfusion was quantified in manually drawn regions of interest (ROIs) based on a one-compartment perfusion model.5,9 ROIs were carefully placed in the lung periphery in order to avoid contribution of macroscopic vessels.Results
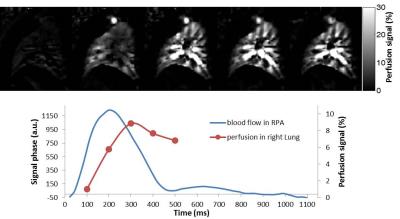
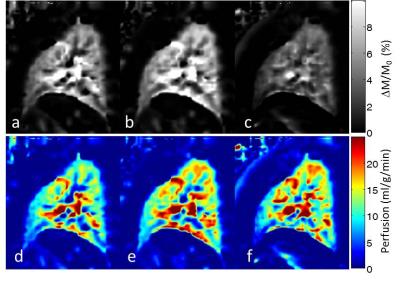
Figure 3a shows perfusion-weighted images measured at various TD by tagging the RPA of a healthy volunteer with an R-R interval of about 1100ms. The corresponding signal time curve is depicted in Figure 3b. Although the signal in RPA increases for long TD, the signal in lung parenchyma achieved a maximum at TD=300ms. In Figure 4 sagittal perfusion images of the right lung are shown. Figure 5 shows coronal perfusion images of a volunteer, which are acquired with labelling of RPA, LPA and pulmonary trunk.Discussion
The presented work demonstrates significant benefits of ECG triggering for pCASL imaging of the lung. The ECG triggering allows an effective labeling of the lung arteries only during systolic blood supply (approx. 300ms) reducing dramatically the RF power deposition in the body. This reduces the heating of the subject during the relatively long pCASL scan time. The calculated perfusion values are significantly larger than the values reported for human lung (approx. 5ml/g/min) mainly due to the overestimation of the labeling efficiency which was assumed 95%.
The combination of the pCASL sequence with fast True-FISP imaging provides high-quality perfusion images of the lung without administration of contrast media. The presented method offers flexible labeling of the lung arteries: left and right lung can be examined separately or both parts simultaneously. This unique feature could be useful for perfusion evaluation of various lung diseases.
Acknowledgements
No acknowledgement found.References
1. Mai VM, Berr SS. MR perfusion imaging of pulmonary parenchyma using pulsed arterial spin labeling techniques: FAIRER and FAIR. J Magn Reson Imaging 1999;9:483–487.
2. Roberts DA et al. Pulmonary perfusion: respiratory-triggered three-dimensional MR imaging with arterial spin tagging–preliminary results in healthy volunteers. Radiology 1999;212:890–895
3. Dai W et al. Continuous flow-driven inversion for arterial spin labeling using pulsed radio frequency and gradient fields. Magn Reson Med. 2008;60:1488-1497.
4. Robson PM et al. Strategies for reducing respiratory motion artifacts in renal perfusion imaging with arterial spin labeling. Magn Reson Med 2009;61:1374–1387.
5. Alsop DC and Detre JA. Reduced transit-time sensitivity in noninvasive magnetic resonance imaging of human cerebral blood flow. J Cereb Blood Flow Metab 1996;16:1236–1249.
6. Greer JS et al. Non-contrast pulmonary perfusion using pseudo-continuous arterial spin labeling of the inferior vena cava. In Proc. Intl Soc Mag Reson Med 2015, p. 534.
7. Martirosian P et al. Quantitative lung perfusion mapping at 0.2 T using FAIR True-FISP MRI. Magn ResonMed 2006;55:1065–1074.
8. Pohmann R et al. Theoretical and experimental evaluation of continuous arterial spin labeling techniques. Magn Reson Med 2010;63:438–446.
9. Buxton RB et al. A general kinetic model for quantitative perfusion imaging with arterial spin labeling. Magn Reson Med 1998;40:383–396.
Figures