1469
Brain activity alteration during the training period of the Hybrid Assistive Limb® (HAL) for chronic spinal cord injuries: a task-based fMRI case report1Center for Cybernics Reseach, University of Tsukuba, Tsukuba, Japan, 2Graduate School of Comprehensive Human Science Majors of Medical Sciences, University of Tsukuba, Tsukuba, Japan, 3Department of Neurosurgery, Faculty of Medicine, University of Tsukuba, Tsukuba, Japan, 4Department of Neurosurgery, Ibaraki Prefectural University of Health Sciences, Inashiki, Japan, 5Faculty of Engineering, Information and Systems, University of Tsukuba, Tsukuba, Japan, 6Department of Radiology, Faculty of Medicine, University of Tsukuba, Tsukuba, Japan, 7Department of Orthopaedic Surgery, Faculty of Medicine, University of Tsukuba, Tsukuba, Japan
Synopsis
We previously developed the novel brain phantom showing image contrast and construction similar to those of in vivo MRI. This phantom has the potential to quantitatively assess the capability of the motion-corrected PROPELLER technique, which has been never approached. In the current study, we investigated the rotational frequency dependencies of the different two motion-corrected PROPELLER techniques by using our brain phantom. Our findings allow to quantitatively assess the capability of the Motion-Correction in PROPELLER.
INTRODUCTION
Hybrid Assistive Limb (HAL; Cyberdyne Inc, Japan) is a new rehabilitation robotic approach (Fig 1a).1 The feasibility of HAL training for chronic spinal cord injury (SCI) should be evaluated.2 This is the first report on changes in brain activation patterns in the primary motor cortex (M1) during training for HAL for complete SCI by using motor imagery task-based functional magnetic resonance imaging (fMRI) 3 times.CASE PRESENTATION
A 19-year-old man who was injured during judo 4 years previously was diagnosed as having a cervical vertebral fracture-dislocation (C3/C4). Complete paralysis and serious sensory dysfunction inferior to the C5 level occurred at the onset of injury. He received additional HAL training 2 times per week for 5 weeks (10 sessions, 40 minutes per session) with standard physical and occupational therapies (Fig 1b). Walking distance and time, and modified Ashworth scale (mAs) score were measured after HAL training.fMRI ACQUISITION
fMRI was performed 3 times during training for HAL (sessions 4, 6, and 10), using a 3-T Philips Ingenia MRI system with echo planar imaging (2.5×2.5 mm in-plane resolution, TR/TE=2500/35 ms, 40 axial slices). Three-dimensional T1-weighted sequence was obtained (TR/TE=7.6/3.6 ms, 0.8×0.8 mm in-plane resolution). The patient performed a motor imagery task of the right ankle movement with a block paradigm (Fig 2). Preprocessing included slice-time correction, realignment, de-spiking, and spatial normalization to MNI space using SPM12. A first-level fMRI model was constructed for the motor imagery sequences, and contrast modeled the linear increase in BOLD signal due to increasing sequence difficulty levels. Region-of-interest analysis was applied to focus on the motor function of the precentral gyrus (PrG). We used the mask Right-Left PrG with SPM12 and a normalized index (laterality index [LI]) to quantify any shift in the cortical activation balance in the PrG during the training period of HAL.3 We calculated LI for the PrG, excluding the face motor area in 3 sessions. LI is defined as LI=(L−R)/(L+R), where L and R are the active voxel count for the specified region in the left and right hemispheres, respectively. Then, we evaluated the correlation between LI and total mAs score by using the Spearman correlation coefficient (R2).RESULTS
Walking distance and time increased from 25.2 m/7.6 min to 148.3 m/7.6 min. The total mAs score (0–144; number of joints: 36) was 15.13±2.80 points before HAL training and 5.75±2.38 points after HAL training. Figure 3 shows that the activation maps for the right and left PrG masked the ROIs at each HAL session. Decreased signal was present in the right side (ipsilateral) but not in the left side (contralateral). The LIs for the PrG regions at the HAL sessions were 0.44, 0.52, and 0.73, respectively (Fig 4). LI significantly correlated with total mAs score (R2=0.938, P<0.01; Fig 5).DISCUSSION
Some studies focused on the changes in measures related to motor ability to clarify the clinical potential of HAL for chronic SCIs.4,5 However, the effectiveness of HAL training for recovery of brain function after chronic SCI remains to be clarified. Our major finding is that fMRI results demonstrated rearrangement of the cortical activation pattern, inducing cerebral lateralization in M1 toward the contralateral hemisphere. Although generalization from the results of a single-case study should be made with caution, this suggests that HAL training could drive neuroplasticity in the cerebral cortex in patients with complete SCIs. LI negatively correlated with total mAs score, which suggests that temporally releasing spasticity due to long-lasting locomotor activity using HAL improved the ability of attempted voluntary ankle movement and subserved the perception of spatial position of the right ankle, allowing a shift to the contralateral hemisphere in activation balance. Considering that all fMRI acquisitions were performed after training, these fMRI results might show a longitudinal change in the immediate effect due to HAL. More cases and further controlled studies are required to clarify the changes in brain function associated with HAL training for chronic SCIs.CONCLUSION
Motor imagery task-based fMRIs during the training period for HAL for patients with complete C4 quadriplegia and chronic SCI were performed. Long-lasting locomotor activity using a HAL device could drive neuroplasticity in the cerebral cortex for patients with complete SCI.Acknowledgements
This study was supported by the Industrial Disease Clinical Research Grants of the Ministry of Health Labour and Welfare, Japan (14060101-01) and funded by ImPACT Program of Council for Science, Technology and Innovation (Cabinet Office, Government of Japan).References
1. Kawamoto H, Sankai Y. Power assist method based on Phase Sequence and muscle force condition for HAL. Adv Robot. 2005;19:717-34.
2. Wall A, Borg J, Palmcrantz S. Clinical application of the Hybrid Assistive Limb (HAL) for gait training-a sustematic review. Front Syst Neurosci. 2014;9:48.
3. Cramer SC, Nelles G, Benson RR, et al. A Functional MRI Study of Subjects Recovered From Hemiparetic Stroke. Stroke. 1997;28:2518-2527.
4. Fujii K, Abe T, Yamazaki M, et al. The voluntary driven exoskeleton Hybrid Assistive Limb (HAL) for postoperative training of thoracic ossification of the posterior longitudinal ligament: a case report. J Spinal Cord Med. 2016;Feb 9:1-7.
5. Ikumi A, Kubota S, Yamazaki M, et al. Decrease of spasticity after hybrid assistive limb training for a chronic spinal cord injury patient with C4 quadriplegia. J Spinal Cord Med. 2016;Oct 20:1-6.
Figures

The Hybrid Assistive Limb (HAL) robot suit (Cyberdyne Inc, Japan).
a: HAL is an exoskeleton with a hybrid system that allows both voluntary and autonomous modes of action to support gait training.
b: Gait training using the HAL robot suit with a body weight support system (BIODEX). Two physicians and 1 therapist support the subject in balance and weight bearing.

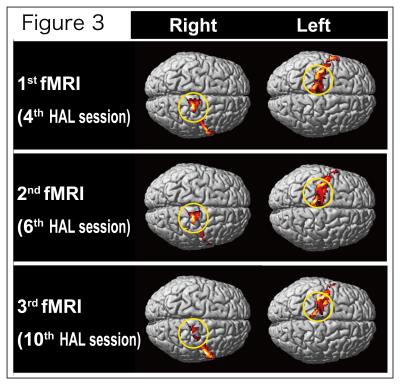
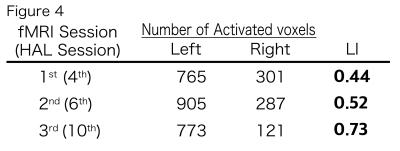
Laterality index (LI) for the PrG region during the motor imagery of the right ankle dorsal-plantar flexion at HAL sessions 4, 6, and 10.The LI at the tenth HAL session shows a higher score than those at the fourth and sixth session.
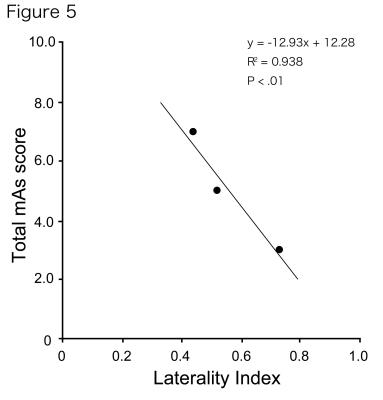
Correlations between the laterality index (LI) and the total mAs score. A significant correlation can be observed between the LI and the total mAs score (R2=0.938, p<0.01).