1041
Flexibility and Robustness of Cones UTE for Pediatric Abdominal and Chest Imaging1Department of Radiology, Stanford University, Stanford, CA, United States, 2Department of Bioengineering, University of California, Berkeley, CA, 3Applied Science Laboratory, GE Healthcare, San Diego, CA, 4Department of Electrical Engineering and Computer Sciences, University of California, Berkeley, CA, United States
Synopsis
Three-dimensional cones k-space sampling trajectory in UTE imaging is ideal for pediatric abdominal and chest MRI which are often hindered by patient motion and lengthy scan durations. The scan-time efficiency of the trajectory reduces scan durations with little to no subsampling. With the repeated sampling and oversampling of the k-space center, the acquisition is robust to motion and can flexibly retrospectively tradeoff spatial and temporal resolutions. Lastly, the method enables retrospective delineation of physiological dynamics. These features of cones UTE enable flexibility and robustness that is ideal for pediatric MRI.
Purpose
A major obstacle in abdomen and chest MRI is patient motion which often necessitates repeated acquisitions. Corruption from motion results in prolonged examinations and prolonged anesthesia if needed. To address these issues, data acquisition should ideally be robust to motion and be able to leverage advanced reconstruction algorithms for scan time reductions. Three-dimensional cones imaging1,2 has such characteristics along with high sampling efficiency. The goal of this work is to develop a cones ultra-short-echo (UTE) technique for pediatric abdominal and chest imaging that enables motion correction and dynamic imaging in respiratory and contrast-enhancement dimensions.Methods
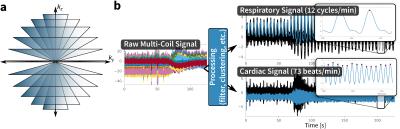
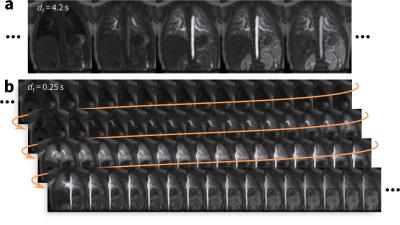
An RF-spoiled GRE sequence with UTE is designed with a 3D cones k-space sampling trajectory1,2 (Figure 1a). The data collection of each cone interleaf is ordered using the golden-ratio increment3. Since each cone interleaf starts at the k-space center, a simple DC navigator4,5 is directly extracted from the data with no added modifications (Figure 1b). The pseudo-random ordering ensures that any effects from gradient hysteresis or eddy currents that may corrupt the signal can be suppressed using a low-pass filter. A low-pass filter (cutoff at 0.67 Hz) is applied to extract respiratory signal, and a band-pass filter (passband from 0.67 to 3 Hz) is applied to extract cardiac signal. Each element from the coil-array receiver produces a spatially-localized motion waveform; the dominant motion signal is extracted using a clustering algorithm6. These physiological signals are used to either suppress motion effects through soft-gating7,8 or to resolve motion dynamics as extra dimensions9.
With informed consent and IRB approval, subjects referred for contrast-enhanced chest 3T MRI (GE MR750) were recruited and scanned using the cones UTE sequence with a 32-channel cardiac coil. All images were acquired with subjects freely breathing. Scan parameters include a flip angle of 15°, bandwidth of ±125 kHz, echo time of 32 us, repetition times of 4.7–5.8 ms, and scan durations of 4.5–5.8 min. To evaluate the scan efficiency of the cones trajectory, a volumetric radial UTE acquisition was obtained with the same scan duration. To evaluate the ability of the acquisition to provide intrinsic motion information for respiratory motion suppression, another acquisition was reconstructed using soft-gating. The self-gating signal was also used to resolve motion dynamics; thus, one acquisition was used to resolve respiratory motion. Lastly, the cones trajectory was used to acquire dynamic-contrast-enhanced (DCE) images with gadolinium-based contrast administration. All datasets were reconstructed using BART10. Parallel imaging and compressed sensing were used for subsampled or soft-gated datasets.
Results
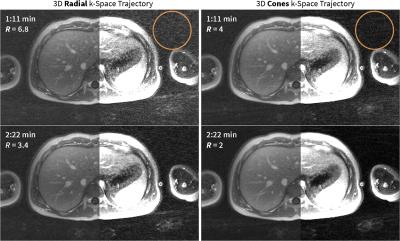
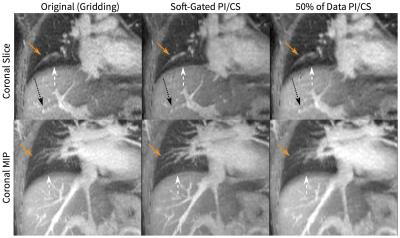
With a longer readout (by 36%) and a spiral twist, the cones k-space trajectory demonstrated a higher sampling efficiency compared to the radial trajectory: cones imaging achieved a lower reduction factor (which results in less aliasing artifacts) compared to radial imaging (Figure 2). The oversampling of the k-space center in the cones trajectory resulted in images that are robust to motion (Figure 3). The sampling trajectory enabled the extraction of a respiratory motion signal (Figure 1) that allowed for soft-gating to suppress respiratory-motion effects (Figure 3). The same technique to extract respiratory waveforms was used to resolve respiratory motion (Figure 4). The respiratory-resolved reconstruction also reduced image artifacts from respiratory motion and enabled the assessment of lung function. Lastly, the oversampling of the k-space center also provided for flexibility in the reconstruction of DCE images. As shown in Figure 5, the single acquisition was retrospectively reconstructed with different spatial and temporal resolutions.
Discussion
The robustness of the acquisition of the cones UTE sequence was demonstrated by the resulting images acquired with all subjects freely breathing and with no retrospective motion compensation. No noticeable artifacts (e.g. ghosting) were observed, and contrast-enhanced structures were well visualized. However, image blurring was still apparent, but this blurring can be mitigated through motion compensation techniques. The nature of the sampling trajectory allowed for intrinsic motion monitoring and for retrospective reconstructions that either suppresses artifacts through soft-gating or resolving respiratory dynamics.
Oversampling the k-space center and pseudo-random ordering the acquisition made for an ideal setup to apply auto-calibrated parallel imaging and compressed sensing techniques. With these reconstruction methods, the acquisition can be readily motion corrected and accelerated through subsampling. Additionally, the trajectory features enabled flexibility in reconstructing DCE images with the potential to push both spatial and temporal resolutions with more advanced reconstructions. However, the computation burden of performing a full volumetric reconstruction with non-Cartesian imaging must be considered. Optimizing the reconstruction will be future work.
Conclusion
The 3D cones k-space trajectory provided motion-robust imaging with the ability to retrospectively suppress residual motion artifacts and to resolve motion dynamics and contrast-enhancement dynamics.Acknowledgements
NIH R01-EB009690, NIH R01-EB019241, NIH P41-EB015891, Tashia and John Morgridge Faculty Scholars Fund, and GE Healthcare.References
[1] Gurney PT., Hargreaves BA, Nishimura DG. Design and analysis of a practical 3D cones trajectory. Magn Reson Med 2006;55:575–582.
[2] Carl M., Bydder GM., Du J. UTE imaging with simultaneous water and fat signal suppression using a time-efficient multispoke inversion recovery pulse sequence. Magn Reson Med 2016;76:577–582.
[3] Malavé MO., Addy NO., Ingle RR., Cheng JY., Nishimura DG. 3D Cones Reordering Design Methods for Whole-Heart Coronary MR Angiography. In: Proceedings of the 23rd Annual Meeting of ISMRM, Toronto, Ontario, Canada, 2015. (abstract 652).
[4] Brau AC., Brittain JH. Generalized self-navigated motion detection technique: preliminary investigation in abdominal imaging. Magn Reson Med 2006;55:263–270.
[5] Larson AC., White RD., Laub G., McVeigh ER., Li D., Simonetti OP. Self-gated cardiac cine MRI. Magn Reson Med 2004;51:93–102.
[6] Zhang T., Cheng JY., Chen Y., Nishimura DG., Pauly JM., Vasanawala SS. Robust self-navigated body MRI using dense coil arrays. Magn Reson Med 2016;76:197–205.
[7] Johnson KM., Block WF., Reeder SB., Samsonov A. Improved least squares MR image reconstruction using estimates of k-Space data consistency. Magn Reson Med 2012;67:1600–1608.
[8] Cheng JY., Zhang T., Ruangwattanapaisarn N., Alley MT., Uecker M., Pauly JM., et al. Free-breathing pediatric MRI with nonrigid motion correction and acceleration. J Magn Reson Imaging 2015;42:407–420.
[9] Feng L., Axel L., Chandarana H., Block KT., Sodickson DK., Otazo R. XD-GRASP: Golden-angle radial MRI with reconstruction of extra motion-state dimensions using compressed sensing. Magn Reson Med 2016;75:775–788.
[10] Uecker M., Ong F., Tamir JI., Bahri D., Virtue P., Cheng JY., et al. Berkeley Advanced Reconstruction Toolbox. In: Proceedings of the 23rd Annual Meeting of ISMRM, Toronto, Ontario, Canada, 2015. (abstract 2486).
Figures