0542
In Vivo Hyperpolarized MRS Study Showing Improved Cardiac Metabolism in Type 1 Diabetes with Daily L-Carnitine Treatment.1University of Oxford, Oxford, United Kingdom
Synopsis
Carnitine acts as a buffer of acetyl-CoA units in the mitochondria, as well as facilitating transport of fatty acids, and carnitine levels are decreased in the diabetic heart. The purpose of this study was to investigate the effect of L-Carnitine supplementation on cardiac function and metabolism in the diabetic rat heart. We show that daily injections of L-Carnitine can alter cardiac metabolism in the in vivo diabetic rat heart, and can increase flux through pyruvate dehydrogenase. Such studies will allow a better understanding of the interactions between metabolism and function in the diabetic heart and may provide new insight into novel therapeutics.
Introduction
Diabetes increases the incidence of myocardial infarction and heart failure, which are leading causes of mortality in diabetic patients. Improving our understanding of the metabolic changes that occur in the diabetic heart may be key to understanding the pathophysiology of the cardiac effects of diabetes. Several studies have shown that L-carnitine levels are decreased in the diseased state and that L-carnitine supplementation protects cardiac function in disease [1,2]. Carnitine acts as a transporter for fatty acids as well as acting, via acetyl-carnitine, as a buffer of excess acetyl-CoA units in the mitochondria. It may therefore have multiple effects on the balance of the energy sources used in the diabetic heart[3]. The purpose of this study was to explore the metabolic and functional effects of daily L-carnitine supplementation on the diabetic rodent heart.Method
22 male Wistar rats(~200g) were split into two groups; rats injected with streptozotocin (STZ, 55 mg/Kg) to induce a model of Type 1 diabetes and rats injected with citrate buffer (CTR) to act as controls. Two weeks after injection, daily intra-peritoneal injections of either 600μl of saline or L-carnitine (3g/kg/day) diluted in saline were initiated in the STZ animals and continued for three weeks. Control animals all received daily saline injections. Following treatment the animals were anesthetized with isoflurane and 13C MR pulse-acquire cardiac spectra were acquired over 60s following an injection of hyperpolarised [1-13C]pyruvate (repetition time 1s; excitation flip angle 15°; sweep width 13,021Hz; acquired points 2,048; frequency centred on the C1 pyruvate resonance) and spectra were summed over 30s from the first appearance of pyruvate and analysed with JMRUI[4] for metabolic assessment of the heart. In addition, eight-ten short-axis slices (slice thickness:1.6mm, matrix size:128×128, TE/TR:1.67/4.6ms, flip angle:15°, number of averages:4) were acquired with a CINE-FLASH sequence and analysed with ImageJ for assessment of cardiac function.Results
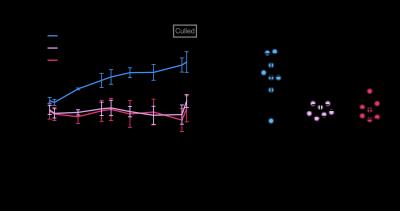
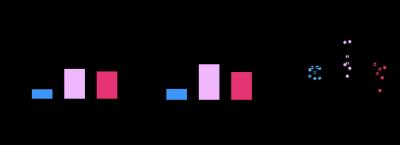
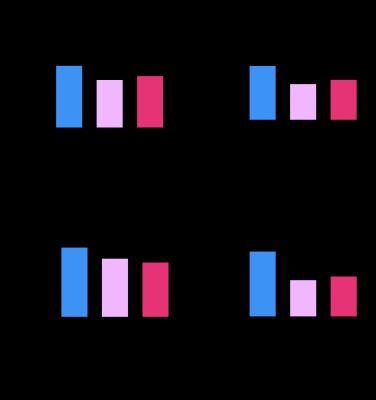
All animals gained weight over the treatment period. The control animals gained significantly more weight (35±18g) than the diabetic animals (STZ saline:8±5g, STZ L-carnitine:8±8g, Fig. 1AB). Blood glucose levels were elevated in the diabetic groups at both 2 and 5 weeks post STZ injection (Fig. 2AB) with the saline treated STZ group showing a significant and progressive increase in hyperglycaemia over the three weeks of treatment (Fig. 2C). Whilst the L-carnitine treated STZ group showed elevated blood glucose at 2 weeks, there was no further increase over the duration of the treatment (Fig. 2C). Average left ventricular mass, stroke volume, ejection fraction and cardiac output were all depressed in the two STZ groups (Fig. 4). Flux through pyruvate dehydrogenase (PDH), as indicated by the bicarbonate to pyruvate ratio, was significantly reduced in the saline and L-carnitine treated STZ animals compared to the control animals (Fig. 5A). However, despite the reduction in PDH flux in the L-carnitine treated STZ animals, PDH flux was significantly higher when compared to the saline treated STZ animals indicating an enhancement of PDH flux caused by the carnitine treatment. Incorporation of the 13C label from pyruvate into both lactate and alanine was increased in the two STZ groups compared to the control animals, with incorporation into lactate significantly elevated in the L-carnitine treated STZ animals, when compared to the saline treated STZ animals (Fig. 5BC).Discussion and Conclusion
This study suggests potentially beneficial effects of L-carnitine supplementation in diabetic animals. Despite demonstrating no differences in cardiac function or weight gain, L-carnitine treatment slowed the progression of hyperglycaemia and elevated flux through the key regulatory enzyme pyruvate dehydrogenase when compared to saline treatment. This metabolic improvement implies that L-carnitine offers an alternative route for the disposal of excess glucose through increased glucose oxidation, as well as via conversion to lactate. It is possible that increased buffering of acetyl-CoA units into acetylcarnitine offers a relief of PDH inhibition caused by excessive fatty acid oxidation in the diabetic heart. Previous studies have shown a beneficial effect of L-carnitine treatment on cardiac function [1]. However, this functional improvement was only seen after six weeks of treatment with metabolic changes preceding the functional changes. It is therefore possible that cardiac function may be improved over a longer period of treatment. This study has showed the potential for L-carnitine treatment to increase flux through PDH and slow down the progression of blood glucose elevation in a model of Type 1 diabetes. Such studies will allow a better understanding of the interactions between metabolism and function in the diabetic heart and may provide new insights into novel therapeutics.Acknowledgements
No acknowledgement found.References
[1] B. Rodrigues, H. Xiang, and J. H. Mcneill, “C 1358,” vol. 37, no. October, 1988.
[2] A. J. Liedtke, T. C. Vary, S. H. Nellis, and C. W. Fultz, “Properties of L-carnitine Incorporation in Working Swine Hearts Effects of Coronary Flow , Ischemia , and Excess Fatty Acids,” 1981.
[3] T. Doenst, T. D. Nguyen, and E. D. Abel, “Cardiac metabolism in heart failure: implications beyond ATP production.,” Circ. Res., vol. 113, no. 6, pp. 709–24, Aug. 2013.
[4] Vanhamme L, van den Boogaart A, Van Huffel S. Improved method for accurate and efficient quantification of MRS data with use of prior knowledge. J Magn Reson 1997;129:35-43
Figures